Summary
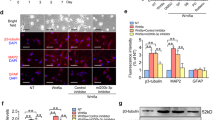
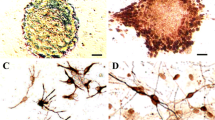
The effects of E-cadherin-transfected neural stem cells (NSCs) transplantation for spinal cord injury (SCI) in rats were investigated. Sixty SD rats were randomly divided into model control group, NSCs group, empty plasmid group and E-cadherin overexpression group (n=15 each). The animal SCI model was established by using the modified Allen’s method. NSCs were cultured. Rats in NSCs group were subjected to NSCs transplantation. E-cadherin gene eucaryotic expression vector and pcDNA3.1-E-cadherin were respectively transfected into cultured NSCs, serving as empty plasmid group and E-cadherin overexpression group respectively. At 7th day after transplantation, neurological function of all rats was assessed by Tarlov score. After rats were sacrificed in each group, the number of BrdU and Nestin positive cells was counted by immunohistochemistry. Immumofluorescence method was used to detect the expression of neurofilament protein (NF) and glial fibrillary acidic protein (GFAP). As compared with model control group, the Tarlov score and the number of of BrdU and Nestin positive cells, and the expression of NF and GFAP in NSCs group, empty plasmid group, and E-cadherin overexpression group were increased significantly (P<0.05), and those in the E-cadherin overexpression group were increased more significantly than the other transplantation groups (P<0.05). It was suggested that E-cadherin could be conductive to nerve regeneration and repair probably by promoting the proliferation and differentiation of NSCs.
Similar content being viewed by others
References
Kim BG, Kang YM, Phi JH, et al. Implantation of polymer scaffolds seeded with neural stem cells in a canine spinal cord injury model. Cytotherapy, 2010,12(6):841–845
Gaspard N, Vanderhaeghen P. From stem cells to neural networks: recent advances and perspectives for neurodevelopmental disorders. Dev Med Child Neurol, 2011,53(1):13–17
Karpowicz P, Willaime-Morawek S, Balenci L, et al. E-cadherin regulates neural stem cell self-renewal. J Neurosci, 2009,29(12):3885–3896
Cioczyk-Wierzbicka D, Gil D, Laidler P. The inhibition of cell proliferation using silencing of N-cadherin gene by siRNA process in human melanoma cell lines. Curr Med Chem, 2012,19(1):145–151
Lee DH, Phi JH, Kim SK, et al. Enhanced reclosure of surgically induced spinal open neural tube defects in chick embryos by injecting human bone marrow stem cells into the amniotic cavity. Neurosurgery, 2010,67(1):129–135
Zhang N, Wimmer J, Qian SJ, et al. Stem cells: Current approach and future prospects in spinal cord injury repair. Anat Rec (Hoboken), 2010,293(3):519–530
Chen S, Kaneko S, Ma X, et al. Lissencephaly-1 controls germline stem cell self-renewal through modulating bone morphogenetic protein signaling and niche adhesion. Proc Natl Acad Sci, 2010,107(466):19 939–19 944
Li D, Zhou J, Wang L, et al. Integrated biochemical and mechanical signals regulate multifaceted human embryonic stem cell functions. J Cell Biol, 2010,191(3):631–644
Spencer H, Keramari M, Ward CM. Using cadherin expression to assess spontaneous differentiation of embryonic stem cells. Methods Mol Biol, 2010,690:81–94
Nasonkin I, Mahairaki V, Xu L, et al. Long-term, stable differentiation of human embryonic stem cell-derived neural precursors grafted into the adult mammalian neostriatum. Stem Cells, 2009,27(10):2414–2426
Carbajal KS, Schaumburg C, Strieter R, et al. Migration of engrafted neural stem cells is mediated by CXCL12 signaling through CXCR4 in a viral model of multiple sclerosis. Proc Natl Acad Sci U S A. 2010,107(24):11068–11073
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, C., Tu, F., Zhang, Jy. et al. E-cadherin-transfected neural stem cells transplantation for spinal cord injury in rats. J. Huazhong Univ. Sci. Technol. [Med. Sci.] 34, 554–558 (2014). https://doi.org/10.1007/s11596-014-1314-0
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-014-1314-0