Summary
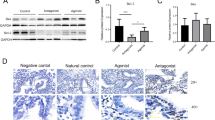
The debate exists whether or not gonadotropin-releasing hormone (GnRH) analogs used in controlled ovarian hyperstimulation (COH) impair endometrial receptivity. Homeobox A11 (Hoxa11), Meis homeobox 1 (Meis1), cadherin 1 (Cdh1), and catenin beta 1 (Ctnnb1) are well known to be involved in successful implantation. In this study, the endometrial expression of Hoxa11, Meis1, Cdh1, and Ctnnb1 during the peri-implantation period was investigated in an in vitro fertilization (IVF) mouse model by real-time RT-PCR and Western blot to evaluate the relationship between Hoxa11, Meis1, Cdh1, and Ctnnb1 expression and the impact of the COH on endometrial receptivity. The mimic COH protocols included GnRH agonist plus human menopausal gonadotropin (HMG) (GnRH agonist group), GnRH antagonist plus HMG (GnRH antagonist group), and HMG alone (HMG group). The expression levels of Hoxa11, Meis1, Cdh1, and Ctnnb1 mRNA and protein were decreased in all of the COH groups. The expression levels of Hoxa11 and Ctnnb1 were the lowest in the GnRH agonist group, and those of Meis1 and Cdh1 were lower in the GnRH analog groups than the HMG group. There were positive correlations between the expression of Hoxa11 and Ctnnb1, as well as the expression of Meis1 and Cdh1 among all the groups. In conclusion, the COH protocols, particularly with GnRH analogs, suppressed Hoxa11, Meis1, Ctnnb1 and Cdh1 expression, in mouse endometrium during the peri-implantation period. Our data reveal a novel molecular mechanism by which the COH protocols might impair endometrial receptivity.
Similar content being viewed by others
References
Santos MA, Kuijk EW, Macklon NS. The impact of ovarian stimulation for IVF on the developing embryo. Reproduction, 2010,139(1):23–34
Smitz J, Devroey P, Camus M, et al. The luteal phase and early pregnancy after combined GnRH-agonist/HMG treatment for superovulation in IVF or GIFT. Hum Reprod, 1988,3(5):585–590
Griesinger G, Felberbaum R, Diedrich K. GnRH-antag onists in reproductive medicine. Arch Gynecol Obstet, 2005,273(2):71–78
Hayden C. GnRH analogues: applications in assisted reproductive techniques. Eur J Endocrinol, 2008, 159(Suppl 1):S17–S25
Kolibianakis EM, Kalogeropoulou L, Griesinger G, et al. Among patients treated with FSH and GnRH analogues for in vitro fertilization, is the addition of recombinant LH associated with the probability of live birth? A systematic review and meta-analysis. Hum Reprod Update, 2007,13:(5):445–452
Tarlatzis BC, Kolibianakis EM. GnRH agonists vs antagonists. Best Pract Res Clin Obstet Gynaecol, 2007, 21(1):57–65
Andersen AN, Gianaroli L, Felberbaum R, et al. Assisted reproductive technology in Europe, 2001. Results generated from European registers by ESHRE. Hum Reprod, 2005,20(5):1158–1176
Nygren KG, Andersen AN. Assisted reproductive technology in Europe, 1997. Results generated from European registers by ESHRE. European IVF-Monitoring Programme (EIM), for the European Society of Human Reproduction and Embryology (ESHRE). Hum Reprod, 2001,16(2):384–391
Diedrich K, Fauser BC, Devroey P, et al. The role of the endometrium and embryo in human implantation. Hum Reprod Update, 2007,13(4):365–377
Check JH, Nowroozi K, Chase J, et al. Comparison of pregnancy rates following in vitro fertilization-embryo transfer between the donors and the recipients in a donor oocyte program. J Assist Reprod Genet, 1992,9(3): 248–250
Stafford-Bell MA, Copeland CM. Surrogacy in Australia: implantation rates have implications for embryo quality and uterine receptivity. Reprod Fertil Dev, 2001,13(1): 99–104
Behrsin RF, da Silva Junior CT, Gabetto JM, et al. Lung volume reduction surgery (LVRS): criteria for selecting patient in Antonio Pedro Hospital, Federal Fluminense University, Niter i city, Rio de Janeiro, Brazil. Rev Assoc Med Bras, 2003,49(3):274–277
Fauser BC, Devroey P. Reproductive biology and IVF: ovarian stimulation and luteal phase consequences. Trends Endocrinol Metab, 2003,14(5):236–242
Simon C, Cano F, Valbuena D, et al. Clinical evidence for a detrimental effect on uterine receptivity of high serum oestradiol concentrations in high and normal responder patients. Hum Reprod, 1995,10(9):2432–2437
van der Gaast MH, Classen-Linke I, Krusche CA, et al. Impact of ovarian stimulation on mid-luteal endometrial tissue and secretion markers of receptivity. Reprod Biomed Online, 2008,17(4):553–563
Haouzi D, Assou S, Dechanet C, et al. Controlled ovarian hyperstimulation for in vitro fertilization alters endometrial receptivity in humans: protocol effects. Biol Reprod, 2010,82(4):679–686
Koler M, Achache H, Tsafrir A, et al. Disrupted gene pattern in patients with repeated in vitro fertilization (IVF) failure. Hum Reprod, 2009,24(10):2541–2548
Mansouri-Attia N, Aubert J, Reinaud P, et al. Gene expression profiles of bovine caruncular and intercaruncular endometrium at implantation. Physiol Genomics, 2009, 39(1):14–27
Ruan HC, Zhu XM, Luo Q, et al. Ovarian stimulation with GnRH agonist, but not GnRH antagonist, partially restores the expression of endometrial integrin beta3 and leukaemia-inhibitory factor and improves uterine receptivity in mice. Hum Reprod, 2006,21(10):2521–2529
Van Vaerenbergh I, Van Lommel L, Ghislain V, et al. In GnRH antagonist/rec-FSH stimulated cycles, advanced endometrial maturation on the day of oocyte retrieval correlates with altered gene expression. Hum Reprod, 2009,24(5):1085–1091
Zhang X, Bocca S, Franchi A, et al. Do GnRH analogues directly affect human endometrial epithelial cell gene expression? Mol Hum Reprod, 2010,16(5):347–60
McGinnis W, Krumlauf R. Homeobox genes and axial patterning. Cell, 1992,68(2):283–302
Taylor HS, Vanden Heuvel GB, Igarashi P. A conserved Hox axis in the mouse and human female reproductive system: late establishment and persistent adult expression of the Hoxa cluster genes. Biol Reprod, 1997,57(6): 1338–1345
Taylor HS. A regulatory element of the empty spiracles homeobox gene is composed of three distinct conserved regions that bind regulatory proteins. Mol Reprod Dev, 1998,49(3):246–253
Taylor HS, Igarashi P, Olive DL, et al. Sex steroids mediate HOXA11 expression in the human peri-implantation endometrium. J Clin Endocr Metab, 1999,84(3):1129–1135
Gendron RL, Paradis H, Hsieh-Li HM, et al. Abnormal uterine stromal and glandular function associated with maternal reproductive defects in Hoxa-11 null mice. Biol Reprod, 1997,56(5):1097–1105
Taylor HS. The role of HOX genes in the development and function of the female reproductive tract. Semin Reprod Med, 2000,18(1):81–89
Hsieh-Li HM, Witte DP, Weinstein M, et al. Hoxa 11 structure, extensive antisense transcription, and function in male and female fertility. Development, 1995,121(5): 1373–1385
Argiropoulos B, Yung E, Humphries RK. Unraveling the crucial roles of Meis1 in leukemogenesis and normal hematopoiesis. Genes Dev, 2007,21(22):2845–2849
Sarno JL, Kliman HJ, Taylor HS. HOXA10, Pbx2, and Meis1 protein expression in the human endometrium: Formation of multimeric complexes on HOXA10 target genes. J Clin Endocr Metab, 2005,90(1):522–528
Xu B, Geerts D, Qian K, et al. Myeloid ecotropic viral integration site 1 (MEIS) 1 involvement in embryonic implantation. Hum Reprod, 2008,23(6):1394–1406
Achache H, Revel A. Endometrial receptivity markers, the journey to successful embryo implantation. Hum Reprod Update, 2006,12(6):731–746
Ozawa M, Engel J, Kemler R. Single amino acid substitutions in one Ca2+ binding site of uvomorulin abolish the adhesive function. Cell, 1990,63(5):1033–1038
Ozawa M, Kemler R. Molecular organization of the uvomorulin-catenin complex. J Cell Biol, 1992,116(4):989–996
Kemler R. From cadherins to catenins: cytoplasmic protein interactions and regulation of cell adhesion. Trends Genet, 1993,9(9):317–321
Rosales C, O’Brien V, Kornberg L, et al. Signal transduction by cell adhesion receptors. Biochim Biophys Acta, 1995,1242(1):77–98
Fujimoto J, Sakaguchi H, Hirose R, et al. Significance of sex steroids in roles of cadherin subfamily and its related proteins in the uterine endometrium and placenta. Horm Res, 1998,50(Suppl 2):S30–S36
Larue L, Ohsugi M, Hirchenhain J, et al. E-cadherin null mutant embryos fail to form a trophectoderm epithelium. Proc Natl Acad Sci USA, 1994,91(17):8263–8267
Ohsugi M, Butz S, Kemler R. Beta-catenin is a major tyrosine-phosphorylated protein during mouse oocyte maturation and preimplantation development. Dev Dyn, 1999,216(2):168–176
Paria BC, Zhao XM, Das SK, et al. Zonula occludens-1 and E-cadherin are coordinately expressed in the mouse uterus with the initiation of implantation and decidualization. Dev Biol, 1999,208(2):488–501
Jeong JW, Lee HS, Franco HL, et al. beta-catenin mediates glandular formation and dysregulation of beta-catenin induces hyperplasia formation in the murine uterus. Oncogene, 2009,28(1):31–40
Deutscher E, Yao HH-C. Essential roles of mesenchyme-derived Beta-catenin in mouse Müllerian duct morphogenesis. Dev Biol, 2007,307(2):227–236
Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods, 2001,25(4):402–408
Chegini N, Rong H, Dou Q, et al. Gonadotropin-releasing hormone (GnRH) and GnRH receptor gene expression in human myometrium and leiomyomata and the direct action of GnRH analogs on myometrial smooth muscle cells and interaction with ovarian steroids in vitro. J Clin Endocrinol Metab, 1996,81(9):3215–3221
Cheon KW, Lee HS, Parhar IS, et al. Expression of the second isoform of gonadotrophin-releasing hormone (GnRH-II) in human endometrium throughout the menstrual cycle. Mol Hum Reprod, 2001,7(5):447–452
Grundker C, Gunthert AR, Millar RP, et al. Expression of gonadotropin-releasing hormone II (GnRH-II) receptor in human endometrial and ovarian cancer cells and effects of GnRH-II on tumor cell proliferation. J Clin Endocrinol Metab, 2002,87(3):1427–1430
Raga F, Casan EM, Kruessel JS, et al. Quantitative gonadotropin-releasing hormone gene expression and immunohistochemical localization in human endometrium throughout the menstrual cycle. Bio Reprod, 1998,59(3): 661–669
Dey SK. Focus on implantation. Reproduction, 2004, 128(6):655–656
Cakmak H, Taylor HS. Molecular mechanisms of treatment resistance in endometriosis: The role of progesterone-Hox gene interactions. Semin Reprod Med, 2010, 28(1):69–74
Sarno J, Schatz F, Huang SJ, et al. Thrombin and interleukin-1beta decrease HOX gene expression in human first trimester decidual cells: implications for pregnancy loss. Mol Hum Reprod, 2009,15(7):451–457
Ertzeid G, Storeng R. The impact of ovarian stimulation on implantation and fetal development in mice. Hum Reprod, 2001,16(2):221–225
Fossum GT, Davidson A, Paulson RJ. Ovarian hyperstimulation inhibits embryo implantation in the mouse. J In Vitro Fert Embryo Transf, 1989,6(1):7–10
Singh H, Aplin JD. Adhesion molecules in endometrial epithelium: tissue integrity and embryo implantation. J Anat, 2009,215(1):3–13
Wang X, Matsumoto H, Zhao X, et al. Embryonic signals direct the formation of tight junctional permeability barrier in the decidualizing stroma during embryo implantation. J Cell Sci, 2004,117(Pt 1):53–62
Konac E, Alp E, Onen HI, et al. Endometrial mRNA expression of matrix metalloproteinases, their tissue inhibitors and cell adhesion molecules in unexplained infertility and implantation failure patients. Reprod Biomed Online, 2009,19(3):391–397
Author information
Authors and Affiliations
Corresponding author
Additional information
This project was supported by a grant from the Major State Basic Research Development Program of China (973 Program) (No. 2007-CB948100).
Rights and permissions
About this article
Cite this article
Xiong, M., Zhang, H., Jin, L. et al. Association of controlled ovarian hyperstimulation treatment with down-regulation of key regulators involved in embryonic implantation in mice. J. Huazhong Univ. Sci. Technol. [Med. Sci.] 31, 535–542 (2011). https://doi.org/10.1007/s11596-011-0486-0
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-011-0486-0