Abstract
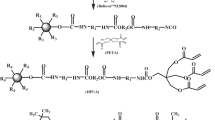
A novel UV-curable prepolymer hexanediol diglycidyl ether diacrylate (HDGEA) was synthesized by utilizing hexanediol diglycidyl ether (HDGE) and acrylic acid (AA) as starting materials, N, N-dimethylbenzylamine as catalyst and p-hydroxyanisole as inhibitor. The optimal synthetic conditions were that the concentration of N, N-dimethylbenzylamine was 0.80 wt% of reactants, the concentration of p-hydroxyanisole was 0.3 wt% of reactants, the reaction temperature was 90–110 °C, and the molar ratio of HDGE to AA was 1: 2.2. Meanwhile, 1-hydroxycyclohexyl phenyl ketone of a UV-curing initiator was added to the synthesized HDGEA to prepare a kind of UV-curing coating. The mechanical properties of the UV-cured films were determined, giving 31.87 MPa of tensile strength, 871.88 MPa of Young’s modulus and 6.77% of elongation at tear.
Similar content being viewed by others
References
Masamitsu Shirai. Reworkable UV Curing Materials[J]. Prog. Org. Coat, 2007, 58: 158–165
M sangermano, N lak, G Malucelli, et al. UV-curing and Characterization of Polymer-clay Nanocoatings by Dispersion of Acrylate-funtionalized Organoclays[J]. Prog. Org. Coat, 2008, 61: 89–94
Dietliker K, Crivello J V. Chemistry and Technology of UV and EB Formulation for Coatings, Inks and Paints[M]. London: SITA Technology Ltd., 1991: 1–9
Davidson R S. Exploring the Science Technology and Application of UV and EB Curing[M]. London: SITA Technology Ltd., 1999: 5–12
Takimoto Y. Radiation Curing in Polymer Science and Technology[M]. London: Elsevier, 1993: 26–29
C Decker, K Moussa. Kinetic Study of Light-induced Polymerization by Real-time UV and IR Spectroscopy[J]. J. Polym. Sci., Polym. Chem., 1993, 30(5): 913–928
Pappas S P. UV-curing Science and Technology[M]. New York: Plenum Press, 1992: 64–71
Crivello J V, Ortiz R A. Synthesis of Epoxy Monomers that Undergo Synergistic Photopolymerization by A Radical- induced Cationic Mechanism[J]. J. Polym. Chem., 2001, 39(20): 3 578–3 592
Decker C, Thi Viet T N, Thi H P. Photoinitiated Cationic Polymerization of Epoxides[J]. Polym. Int., 2001, 50(9): 986–997
Kumar V, Bhardwaj Y K, Goel N K, et al. Coating Characteristics of Electron Beam Cured Bisphenol A Diglycidyl Ether Diacrylate-co-aliphatic Urethane Diacrylate Resins[J]. Surf Coat Technol., 2008, 202(21): 5 202–5 209
Young-Jun Park, Dong-Hyuk Lim, Hyun-Joong Kim, et al. UV- and Thermal-curing Behaviors of Dual-curable Adhesives Based on Epoxy Acrylate Oligomers[J]. Int. J. Adhes., 2009, 29: 710–717
Crivello J V, Ortiz R A. Design and Synthesis of Highly Reactive Photopolymerizable Epoxy Monomers[J]. J. Polym. Sci. Polym. Chem., 2001, 39(14): 2 385–2 395
Sangermano M, Malucelli G, Morel F, et al. Cationic Photopolymerization of Vinyl Ether Systems[J]. Eur. Polym. J., 1999, 35(4): 639–645
Decker C, Bianchi C, Decker D, et al. Photoinitiated Polymerization of Vinyl Ether-based Systems[J]. Prog. Org. Coat, 2001, 42(3): 253–266
Y C Zheng, W J Wei, F Chen. Study on Synthesis and UV-curing Behaviors of ADGDE[J]. Thermosetting Resin, 2003, 18(1): 14–17
W J Wei, Y C Zheng, G Lu. Synthesis and Characteristics of Acrylate Epoxy Oligomer Used for Light-curing[J]. Chemical World, 2002, 12: 638–640
B W Huang, B F Huang, G P Du, et al. Synthesis of a Novel UV-curable Prepolymer Polypropyleneglycol Diglycidyl Ether Diacrylate[J]. Journal of Wuhan University of Technology- Mater. Sci.Ed., 2008, 23(4): 495–498
Sperling L H, Mishra V. Polymer Materials Encyclopedia[M]. New York: CRC Press, 1996: 3 252–3 276
ASTM D882-91. The Method for Tensile Properties of Thin Plastic Sheeting[S]. Agencies of the Department of Defense, 1993
Author information
Authors and Affiliations
Corresponding author
Additional information
Funded by the Natural Science Foundation of Jiangxi Province (No.2008GZC0021) and the National “863” Hi-tech Foundation of China (No.2002AA6Z3083)
Rights and permissions
About this article
Cite this article
Huang, B., Chen, W., Yang, Z. et al. Synthesis of a novel UV-curable prepolymer hexanediol diglycidyl ether diacrylate and its cured film tensile property. J. Wuhan Univ. Technol.-Mat. Sci. Edit. 26, 4–9 (2011). https://doi.org/10.1007/s11595-011-0156-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11595-011-0156-x