Abstract
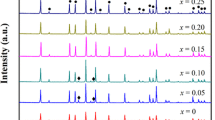
Garnet-type Li7La3Zr2O12 (LLZO) is considered as a promising solid electrolyte. However, the synthesis of LLZO often requires a high temperature, which may lead to the evaporation of the lithium and result in a decrease in ionic conductivity. Therefore, it is an important issue how to reduce the synthesis temperature of LLZO and simultaneously increase its ionic conductivity. Herein, we synthesized garnet-type solid electrolytes of Li6.1Ga0.3La3Zr2O12 (LLZO-Ga) with x wt% CuO (x = 0, 0.2, 0.5, 1, 2) by the traditional solid-state reaction method, in which CuO was introduced as a sintering aid to reduce the sintering temperature of LLZO-Ga and increase its Li-ion conductivity. It is found that adding a small amount of CuO as an additive can reduce the sintering temperature from 1100 ℃ to about 1000 ℃. As a result, when the amount of CuO is 0.5 wt%, LLZO-Ga shows the highest room-temperature ionic conductivity and the lowest activation energy, which are 1.111 mS/cm and 0.27 eV, respectively.







Similar content being viewed by others
References
Armand AM, Tarascon JM (2001) Issues and challenges facing rechargeable lithium batteries. Nature Nature 414:359–367
Nishi Y (2001) Lithium ion secondary batteries; past 10 years and the future. J Power Sources 100:101–106
Quartarone E, Mustarelli P (2011) Electrolytes for solid-state lithium rechargeable batteries: recent advances and perspectives. Chem Soc Rev 40:2525–2540
Wu JF, Pang WK, Peterson VK, Wei L, Guo X (2017) Garnet-type fast Li-ion conductors with high ionic conductivities for all-solid-state batteries. ACS Appl Mater Interfaces 9:12461–12468
Thangadurai V, Narayanan S, Pinzaru D (2014) Garnet-type solid-state fast Li ion conductors for Li batteries: critical review. Chem Soc Rev 43:4714–4727
Pervez SA, Vinayan BP, Cambaz MA, Melinte G, Diemant T, Braun T, Karkera G, Behm RJ, Fichtner M (2020) Electrochemical and compositional characterization of solid interphase layers in an interface-modified solid-state Li–sulfur battery. J Mater Chem A 8:16451–16462
Zhang W, Nie J, Li F, Wang ZL, Sun C (2018) A durable and safe solid-state lithium battery with a hybrid electrolyte membrane. Nano Energy 45:413–419
Nguyen QH, Luu VT, Nguyen HL, Lee YW, Cho Y, Kim SY, Jun YS, Ahn W (2020) Li7La3Zr2O12 garnet solid polymer electrolyte for highly stable all-solid-state batteries. Front Chem 8:619832
Zhao M, Li BQ, Peng HJ, Yuan H, Wei JY, Huang JQ (2020) Lithium-sulfur batteries under lean electrolyte conditions: challenges and opportunities. Angew Chem Int Ed Engl 59:12636–12652
Li Y, Chen X, Dolocan A, Cui Z, Xin S, Xue L, Xu H, Park K, Goodenough JB (2018) Garnet electrolyte with an ultralow interfacial resistance for Li-metal batteries. J Am Chem Soc 140:6448–6455
Thangadurai V, Pinzaru D, Narayanan S, Baral AK (2015) Fast solid-state Li ion conducting garnet-type structure metal oxides for energy storage. J Phys Chem Lett 6:292–299
Samson AJ, Hofstetter K, Bag S, Thangadurai V (2019) A bird’s-eye view of Li-stuffed garnet-type Li7La3Zr2O12 ceramic electrolytes for advanced all-solid-state Li batteries. Energy Environ Sci 12:2957–2975
Alexander GV, Indu MS, Murugan R (2021) Review on the critical issues for the realization of all-solid-state lithium metal batteries with garnet electrolyte: interfacial chemistry, dendrite growth, and critical current densities. Ionics 27:4105–4126
Xu S, Hu L (2020) Towards a high-performance garnet-based solid-state Li metal battery: a perspective on recent advances. J Power Sources 472:228571
Wang CW, Fu K, Kammampata SP, McOwen DW, Samson AJ, Zhang L, Hitz GT, Nolan AM, Wachsman ED, Mo YF, Thangadurai V, Hu LB (2020) Garnet-type solid-state electrolytes: materials, interfaces, and batteries. Chem Rev 120:4257–4300
Murugan R, Thangadurai V, Weppner W (2007) Fast lithium ion conduction in garnet-type Li7La3Zr2O12. Angewandte Chemie-International Edition 46:7778–7781
Liu Q, Geng Z, Han C, Fu Y, Li S, Y-b He F, Kang BL (2018) Challenges and perspectives of garnet solid electrolytes for all solid-state lithium batteries. J Power Sources 389:120–134
Buschmann H, Dolle J, Berendts S, Kuhn A, Bottke P, Wilkening M, Heitjans P, Senyshyn A, Ehrenberg H, Lotnyk A, Duppel V, Kienle L, Janek J (2011) Structure and dynamics of the fast lithium ion conductor “Li7La3Zr2O12.” Phys Chem Chem Phys 13:19378
Geiger CA, Alekseev E, Lazic B, Fisch M, Armbruster T, Langner R, Fechtelkord M, Kim N, Pettke T, Weppner W (2011) Crystal chemistry and stability of “Li7La3Zr2O12” garnet: a fast lithium-ion conductor. Inorg Chem 50:1089–1097
Chen Y, Rangasamy E, CR dela Cruz, C Liang, K An, (2015) A study of suppressed formation of low-conductivity phases in doped Li7La3Zr2O12 garnets by in situ neutron diffraction. J Mater Chem A 3:22868–22876
Abrha LH, Hagos TT, Nikodimos Y, Bezabh HK, Berhe GB, Hagos TM, Huang CJ, Tegegne WA, Jiang SK, Weldeyohannes HH, Wu SH, Su WN, Hwang BJ (2020) Dual-doped cubic garnet solid electrolytes with superior air stability. ACS Appl Mater Interfaces 12:25709–25717
Kim M, Kim G, Lee H (2021) Tri-doping of sol-gel synthesized garnet-type oxide solid-state electrolyte. Micromachines (Basel) 12(2):134
Botros M, Djenadic R, Clemens O, Moller M, Hahn H (2016) Field assisted sintering of fine-grained Li7-3xLa3Zr2AlxO12 solid electrolyte and the influence of the microstructure on the electrochemical performance. J Power Sources 309:108–115
He LC, Oh JAS, Chua JJJ, Zhou HH (2021) Solid-state electrolytes: advances and perspectives. Funct Mater Lett 14:2130001
Mukhopadhyay S, Thompson T, Sakamoto J, Huq A, Wolfenstine J, Allen JL, Bernstein N, Stewart DA, Johannes MD (2015) Structure and stoichiometry in supervalent doped Li7La3Zr2O12. Chem Mater 27:3658–3665
Hu Z, Liu H, Ruan H, Hu R, Su Y, Zhang L (2016) High Li-ion conductivity of Al-doped Li7La3Zr2O12 synthesized by solid-state reaction. Ceram Int 42:12156–12160
Gai J, Zhao E, Ma F, Sun D, Ma X, Jin Y, Wu Q, Cui Y (2018) Improving the Li-ion conductivity and air stability of cubic Li7La3Zr2O12 by the co-doping of Nb, Y on the Zr site. J Eur Ceram Soc 38:1673–1678
Shen L, Wang L, Wang Z, Jin C, Peng L, Pan X, Sun J, Yang R (2019) Preparation and characterization of Ga and Sr co-doped Li7La3Zr2O12 garnet-type solid electrolyte. Solid State Ionics 339:114992
Kobi S, Amardeep AV, Bhargava P, Mukhopadhyay A (2020) Al and Mg Co-doping towards development of air-stable and Li-ion conducting Li-La-zirconate based solid electrolyte exhibiting low electrode/electrolyte interfacial resistance. J Electrochem Soc 167:120519
Cao Z, Li Y, Su J, Zhao J, Li Y, Yan S, Liu Q, He T, Zhang H, Li G-R (2021) Y and Sb co-doped Li7La3Zr2O12 electrolyte for all solid-state lithium batteries. Ionics 27:1861–1870
Mishra M, Hsu C-W, Chandra Rath P, Patra J, Lai H-Z, Chang T-L, Wang C-Y, Wu T-Y, Lee T-C, Chang J-K (2020) Ga-doped lithium lanthanum zirconium oxide electrolyte for solid-state Li batteries. Electrochim Acta 353:136536
Chen C, Sun Y, He LC, Kotobuki M, Hanc E, Chen Y, Zeng KY, Lu L (2020) Microstructural and electrochemical properties of Al- and Ga-Doped Li7La3Zr2O12 garnet solid electrolytes. ACS Applied Energy Materials 3:4708–4719
Jalem R, Yamamoto Y, Shiiba H, Nakayama M, Munakata H, Kasuga T, Kanamura K (2013) Concerted migration mechanism in the Li ion dynamics of garnet-type Li7La3Zr2O12. Chem Mater 25:425–430
Rosero-Navarro NC, Yamashita T, Miura A, Higuchi M, Tadanaga K, Stevenson JW (2017) Effect of sintering additives on relative density and Li-ion conductivity of Nb-doped Li7La3ZrO12 solid electrolyte. J Am Ceram Soc 100:276–285
Fagg DP, Kharton VV, Frade JR (2002) P-type electronic transport in Ce0.8Gd0.2O2-delta: the effect of transition metal oxide sintering aids. J Electroceram 9:199–207
Qin S, Zhu X, Jiang Y, Me Ling ZHu, Zhu J (2018) Growth of self-textured Ga3+-substituted Li7La3Zr2O12 ceramics by solid state reaction and their significant enhancement in ionic conductivity. Appl Phys Lett 112:113901
Jiang Y, Zhu X, Qin S, Me Ling J, Zhu, (2017) Investigation of Mg2+, Sc3+ and Zn2+ doping effects on densification and ionic conductivity of low-temperature sintered Li7La3Zr2O12 garnets. Solid State Ionics 300:73–77
Ashuri M, Golmohammad M, Soleimany Mehranjani A, Faghihi Sani M (2021) Al-doped Li7La3Zr2O12 garnet-type solid electrolytes for solid-state Li-Ion batteries. J Mater Sci: Mater Electron 32:6369–6378
Zhang Y, Luo D, Luo W, Du S, Deng Y, Deng J (2020) High-purity and high-density cubic phase of Li7La3Zr2O12 solid electrolytes by controlling surface/volume ratio and sintering pressure. Electrochim Acta 359:136965
Funding
This work was financially supported by the Ministry of Science and Technology of China (MOST) (Grant No. 2013CB934700), Sichuan Science and Technology Program (Grant No. 2020YFH0047), and the Fundamental Research Funds for Central Universities.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liu, X., Jiang, Y., Cheng, X. et al. Reduced synthesis temperature and significantly enhanced ionic conductivity for Li6.1Ga0.3La3Zr2O12 electrolyte prepared with sintering aid CuO. Ionics 28, 5071–5080 (2022). https://doi.org/10.1007/s11581-022-04761-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11581-022-04761-7