Abstract
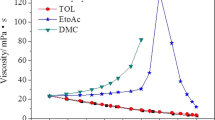
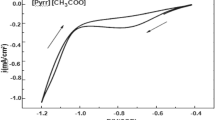
Four typical chloroaluminate ionic liquids (ILs) with different cations, namely 1-butyl-3-methylimidazoliumchloride [Bmim]Cl/AlCl3 (33.3/66.7 mol%), 1-butyl-3-methylpyridinium chloride [BMPyri]Cl/AlCl3 (33.3/66.7 mol%), 1-butyl-1-methylpyrrolidiniumchloride [Py1,4]Cl/AlCl3 (33.3/66.7 mol%), and trimethylphenylammonium chloride [TMPA]Cl/AlCl3 (33.3/66.7 mol%), were employed to deposit Al. The viscosity and ionic conductivity of these ILs were measured. The results showed that [Bmim]Cl/AlCl3 (33.3/66.7 mol%) had the lowest viscosity and the highest conductivity. Raman spectra showed that Al2Cl7 − was the main anion in the four systems. Cyclic voltammetry indicated that [Bmim]Cl/AlCl3 (33.3/66.7 mol%) had the highest reduction current for Al deposition in the four ILs. By comparing the quality of the Al coatings prepared at the same current density and temperature, it was found that compact and smooth Al deposits could be obtained from [Bmim]Cl/AlCl3 (33.3/66.7 mol%) at 303 K, while the temperature needed was higher than 333 K for the other three ILs to obtain Al deposits with the same quality. Based on the density functional theory (DFT) calculations, the differences in these properties were attributed to the different molecular structure and cation-anion interaction induced by the cations of these ILs.








Similar content being viewed by others
References
Zhang S, Sun J, Zhang X, Xin J, Miao Q, Wang J (2014) Ionic liquid-based green processes for energy production. Chem Soc Rev 43:7838–7869
Abbott AP, Frisch G, Ryder KS (2013) Electroplating using ionic liquids. Annu Rev Mater Res 43:335–358
SZE A, Polleth M, Meiss SA, Janek J, Endres F (2007) Ionic liquids as green electrolytes for the electrodeposition of nanomaterials. Green Chem 9:549–553
Zhao Y, Vander Noot TJ (1997) Review: electrodeposition of aluminium from nonaqueous organic electrolytic systems and room temperature molten salts. Electrochim Acta 42:3–13
Endo A, Miyake M, Hirato T (2014) Electrodeposition of aluminum from 1,3-dimethyl-2-imidazolidinone/AlCl3 baths. Electrochim Acta 137:470–475
Abbott AP, KJ MK (2006) Application of ionic liquids to the electrodeposition of metals. Phys Chem Chem Phys 8:4265–4279
Abbott AP, Qiu F, Abood HM, Ali MR, Ryder KS (2010) Double layer, diluent and anode effects upon the electrodeposition of aluminium from chloroaluminate based ionic liquids. Phys ChemChem Phys 12:1862–1872
Barchi L, Bardi U, Caporali S, Fantini M, Scriv Scrivani A (2010) Electroplated bright aluminium coatings for anticorrosion and decorative purposes. Prog Org Coat 67:146–151
Chang JK, Chen SY, Tsai WT, Deng MJ, Sun IW (2007) Electrodeposition of aluminum on magnesium alloy in aluminum chloride (AlCl3)-1-ethyl-3-methylimidazolium chloride (EMIC) ionic liquid and its corrosion behavior. Electrochem Commun 9:1602–1606
Endres F, Bukowski M, Hempelmann R, Natter H (2003) Electrodeposition of nanocrystalline metals and alloys from ionic liquids. Angew Chem Int Edit 42:3428–3430
Yue G, Zhang S, Zhu Y, Lu X, Li C, Li Z (2009) A promising method for electrodeposition of aluminium on stainless steel in ionic liquid. AIChE 55:783–796
Zhang Q, Wang Q, Zhang S, Lu X (2014) Effect of nicotinamide on electrodeposition of Al from aluminium chloride (AlCl3)-1-butyl-3-methylimidazolium chloride (BmimCl) ionic liquids. J Solid State Electr 18:257–267
Wang Q, Chen B, Zhang Q, Lu X, Zhang S (2015) Aluminum deposition from lewis acidic 1-butyl-3-methylimidazolium chloroaluminate ionic liquid (BmimCl/AlCl3) modified with methyl nicotinate. Chem Electro Chem 2:1794–1798
Wang Q, Zhang Q, Chen B, Lu X, Zhang S (2015) Electrodeposition of bright Al coatings from 1-butyl-3-methylimidazolium chloroaluminate ionic liquids with specific additives. J Electrochem Soc 162:D320–D324
Abbott AP, Harris RC, Hsieh YT, Ryder KS, Sun IW (2014) Aluminium electrodeposition under ambient conditions. Phys ChemChem Phys 16:14675–14681
Estager J, Holbrey JD, Swadzba Kwasny M (2014) Halometallate ionic liquids. Chem Soc Rev 43:847–886
Robinson J, Osteryoung RA (1980) The electrochemical behavior of aluminum in the low temperature molten salt system n-butyl pyridinium chloride: aluminum chloride and mixtures of this molten salt with benzene. J Electrochem Soc 127:122–128
Chang JK, Chen SY, Tsai WT, Deng MJ, Sun IW (2008) Improved corrosion resistance of magnesium alloy with a surface aluminum coating electrodeposited in ionic liquid. J Electrochem Soc 155:C112–C116
Giridhar P, Zein El AS, Endres F (2012) Electrodeposition of aluminium from 1-butyl-1-methylpyrrolidinium chloride/AlCl3 and mixtures with 1-ethyl-3-methylimidazolium chloride/AlCl3. Electrochim Acta 70:210–214
Jiang T, Chollier Brym MJ, Dubé G, Lasia A, Brisard GM (2006) Electrodeposition of aluminium from ionic liquids: part II—studies on the electrodeposition of aluminum from aluminum chloride (AlCl3)-trimethylphenylammonium chloride (TMPAC) ionic liquids. Surf Coat Tech 201:10–18
Eiden P, Liu Q, SZE A, Endres F, Krossing I (2009) An experimental and theoretical study of the aluminium species present in mixtures of AlCl3 with the ionic liquids BMPTf2N and EMImTf2N. Chem-Eur J 15:3426–3434
Schwabe T, Grimme S (2007) Double-hybrid density functionals with long-range dispersion corrections: higher accuracy and extended applicability. Phys Chem Chem Phys 9:3397–3406
Zheng Y, Dong K, Wang Q, Zhang J, Lu X (2013) Density, viscosity, and conductivity of lewis acidic 1-butyl- and 1-hydrogen-3-methylimidazolium chloroaluminate ionic liquids. J Chem Eng Data 58:32–42
Zhang X, Huo F, Liu X, Dong K, He H, Yao X, Zhang S (2015) Influence of microstructure and interaction on viscosity of ionic liquids. Ind Eng Chem Res 54:3505–3514
Acknowledgements
The authors gratefully acknowledge the financial support from the General Program Youth of National Natural Science Foundation of China (51404230, 21406002), National Basic Research Program of China (2013CB632606), and CAS Province Cooperation Program (2014JZ0012).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, Q., Zhang, Q., Lu, X. et al. Electrodeposition of Al from chloroaluminate ionic liquids with different cations. Ionics 23, 2449–2455 (2017). https://doi.org/10.1007/s11581-017-2074-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11581-017-2074-1