Abstract
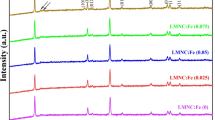
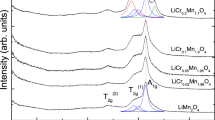
Lithium nickel cobalt oxide materials doped with Al, Mg, and Fe were synthesized by solid-state reaction at 800 °C for 18 h to study the effects of adding transition and nontransition metals to the structure. Crystalline compounds were obtained as revealed by powder X-ray diffraction (XRD). Energy dispersive analysis of X-rays (EDAX) was used to determine the elemental ratio of all the samples. Impedance measurements showed that all samples have decreasing conductivities at higher temperatures and gave negative activation energies. The addition of nontransition metals actually decreased the conductivities of the materials.














Similar content being viewed by others
References
Dahn JR, von Sacken U, Michal CA (1990) Solid State Ionics 44:87–97
Ohzuku T, Ueda A, Nagayama M (1993) J Electrochem Soc 140:1862–1870
Brousseley M, Planchat JP, Rigobert G, Virey D, Sarre G (1997) J Power Sources 68:8–12
Ohzuku T, Ueda A (1994) Solid State Ionics 69:201–211
Dahn JR, Sacken UV, Juzkow MW, Al-Anaby H (1991) J Electrochem Soc 138:2207–2211
Broussely M, Perton F, Labat J, Staniewicz RJ, Romero A (1993) J Power Sources 43–44:209–216
Saadoune I, Delmas C (1998) J Solid State Chem 136:8–15
Fujita Y, Amine K, Maruta J, Yasuda H (2000) J Power Sources 90:82–88
Madhavi S, Subba Rao GV, Chowdari BVR, Li SFY (2002) Solid State Ionics 152–153:199–205
Guilmard M, Pouillerie C, Croguennec L, Delmas C (2003) Solid State Ionics 160:39–50
Delmas C, Saadoune I (1992) Solid State Ionics 53–56:370–375
Delmas C, Saadoune I, Rougier A (1993) J Power Sources 4344:595–602
Zhecheva E, Stoyanova R (1993) Solid State Ionics 66:143–149
Ueda A, Ohzuku T (1994) J Electrochem Soc 141(8):2010–2014
Rougier A, Saadoune I, Gravereau P, Willmann P, Delmas C (1996) Solid State Ionics 90:83–90
Madhavi S, Subba Rao GV, Chowdari BVR, Li SFY (2001) J Power Sources 93:156–162
Lee KK, Yoon WS, Kim KB, Lee KY, Hong ST (2001) J Power Sources 97–98:308–312
Weaving JS, Cowar F, Teagle DA, Cullen J, Dass V, Bindin P, Green R, Macklin WJ (2001) J Power Sources 97–98:733–735
Dahn JR, Von Sacken V, Micheal CA (1990) Solid State Ionics 44:87–97
Ooms FGB, Kelder EM, Schoonman J, Wagemaker M, Mulder FM (2002) Solid State Ionics 152–153:143–153
Chang CC, Kim JY, Kumta PN (2000) J Power Sources 89:56–63
Antolini E (2004) Solid State Ionics 170:159–171
Imanishi N, Fujiyoshi M, Takeda Y, Yamamoto O, Tabuchi T (1999) Solid State Ionics 118:121–128
Levasseur S, Menetrier M, Suard E, Delmas C (2000) Solid State Ionics 128:11–24
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sethuprakhash, V., Basirun, W.J. Structural and electrochemical investigation of LiNi0.8Co0.2 − x M x O2 (M = Al, Al+Mg, Al+Mg+Fe) synthesized by solid-state method. Ionics 14, 501–507 (2008). https://doi.org/10.1007/s11581-008-0207-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11581-008-0207-2