Abstract
Purpose
A real-time 3D image guidance system is needed to facilitate treatment of liver masses using radiofrequency ablation, for example. This study investigates the feasibility and accuracy of using an electromagnetically tracked flexible needle inserted into the liver to track liver motion and deformation.
Methods
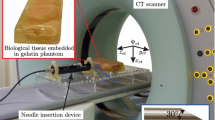
This proof-of-principle study was conducted both ex vivo and in vivo with a CT scanner taking the place of an electromagnetic tracking system as the spatial tracker. Deformations of excised livers were artificially created by altering the shape of the stage on which the excised livers rested. Free breathing or controlled ventilation created deformations of live swine livers. The positions of the needle and test targets were determined through CT scans. The shape of the needle was reconstructed using data simulating multiple embedded electromagnetic sensors. Displacement of liver tissues in the vicinity of the needle was derived from the change in the reconstructed shape of the needle.
Results
The needle shape was successfully reconstructed with tracking information of two on-needle points. Within 30 mm of the needle, the registration error of implanted test targets was 2.4 ± 1.0 mm ex vivo and 2.8 ± 1.5 mm in vivo.
Conclusion
A practical approach was developed to measure the motion and deformation of the liver in real time within a region of interest. The approach relies on redesigning the often-used seeker needle to include embedded electromagnetic tracking sensors. With the nonrigid motion and deformation information of the tracked needle, a single- or multimodality 3D image of the intraprocedural liver, now clinically obtained with some delay, can be updated continuously to monitor intraprocedural changes in hepatic anatomy. This capability may be useful in radiofrequency ablation and other percutaneous ablative procedures.
Similar content being viewed by others
References
Tateishi R, Shiina S, Teratani T, Obi S, Sato S, Koike Y, Fujishima T, Yoshida H, Kawabe T, Omata M (2005) Percutaneous radiofrequency ablation for hepatocellular carcinoma: an analysis of 1000 cases. Cancer 103: 1201–1209. doi:10.1002/cncr.20892
Vogl TJ, Muller PK, Mack MG, Straub R, Engelmann K, Neuhaus P (1999) Liver metastases: interventional therapeutic techniques and results, state of the art. Eur Radiol 9: 675–684
Sutherland LM, Williams JA, Padbury RT, Gotley DC, Stokes B, Maddern GJ (2006) Radiofrequency ablation of liver tumors: a systematic review. Arch Surg 141: 181–190. doi:10.1001/archsurg.141.2.181
Antoch G, Kuehl H, Vogt FM, Debatin JF, Stattaus J (2002) Value of CT volume imaging for optimal placement of radiofrequency ablation probes in liver lesions. J Vasc Interv Radiol 13: 1155–1161
Baegert C, Villard C, Schreck P, Soler L, Gangi A (2007) Trajectory optimization for the planning of percutaneous radiofrequency ablation of hepatic tumors. Comput Aided Surg 12: 82–90. doi:10.1080/10929080701312000
Sandrasegaran K, Rydberg J (2007) Fundamentals of multislice CT scanners. In: Boiselle P, White C (eds) New techniques in cardiothoracic imaging. Informa Healthcare, New York, p 1
Lei P, Dandekar O, Widlus D, Shekhar R (2009) Incorporation of preprocedural PET into CT-guided radiofrequency ablation of hepatic metastases: a nonrigid image registration validation study. J Digit Imaging (in press). doi:10.1007/s10278-009-9204-x
Das M, Sauer F, Schoepf UJ, Khamene A, Vogt SK, Schaller S, Kikinis R, vanSonnenberg E, Silverman SG (2006) Augmented reality visualization for CT-guided interventions: system description, feasibility, and initial evaluation in an abdominal phantom. Radiology 240: 230–235. doi:10.1148/radiol.2401040018
Giesel FL, Mehndiratta A, Locklin J, McAuliffe MJ, White S, Choyke PL, Knopp MV, Wood BJ, Haberkorn U, von Tengg-Kobligk H (2009) Image fusion using CT, MRI and PET for treatment planning, navigation and follow up in percutaneous RFA. Exp Oncol 31: 106–114
Wein W, Brunke S, Khamene A, Callstrom MR, Navab N (2008) Automatic CT-ultrasound registration for diagnostic imaging and image-guided intervention. Med Image Anal 12: 577–585. doi:S1361-8415(08)00063-7
Wein W, Khamene A, Clevert DA, Kutter O, Navab N (2007) Simulation and fully automatic multimodal registration of medical ultrasound. Med Image Comput Comput Assist Interv 10: 136–143
Elhawary H, Oguro S, Tuncali K, Paul RM, Shyn BP, Tatli S, Silverman GS, Hata N (2009) Intra-operative multimodal non-rigid registration of the liver for navigated tumor ablation. Med Image Comput Comput Assist Interv 12: 837–844
Hawkes DJ, Barratt D, Blackall JM, Chan C, Edwards PJ, Rhode K, Penney GP, McClelland J, Hill DL (2005) Tissue deformation and shape models in image-guided interventions: a discussion paper. Med Image Anal 9: 163–175. doi:10.1016/j.media.2004.11.007
Lu W, Chen ML, Olivera GH, Ruchala KJ, Mackie TR (2004) Fast free-form deformable registration via calculus of variations. Phys Med Biol 49: 3067–3087
Sdika M (2008) A fast nonrigid image registration with constraints on the Jacobian using large scale constrained optimization. IEEE Trans Med Imaging 27: 271–281. doi:10.1109/Tmi.2007.905820
Slomka PJ, Dey D, Przetak C, Aladl UE, Baum RP (2003) Automated 3-dimensional registration of stand-alone (18)F-FDG whole-body PET with CT. J Nucl Med 44: 1156–1167
de Baere T, Rehim MA, Teriitheau C, Deschamps F, Lapeyre M, Dromain C, Boige V, Ducreux M, Elias D (2006) Usefulness of guiding needles for radiofrequency ablative treatment of liver tumors. Cardiovasc Intervent Radiol 29: 650–654. doi:10.1007/s00270-005-0187-9
Tomiyama N, Mihara N, Maeda M, Johkoh T, Kozuka T, Honda O, Hamada S, Yoshida S, Nakamura H (2000) CT-guided needle biopsy of small pulmonary nodules: value of respiratory gating. Radiology 217: 907–910
Levy EB, Zhang H, Lindisch D, Wood BJ, Cleary K (2007) Electromagnetic tracking-guided percutaneous intrahepatic portosystemic shunt creation in a swine model. J Vasc Interv Radiol 18: 303–307. doi:10.1016/j.jvir.2006.12.716
Levy EB, Tang J, Lindisch D, Glossop N, Banovac F, Cleary K (2007) Implementation of an electromagnetic tracking system for accurate intrahepatic puncture needle guidance: accuracy results in an in vitro model. Acad Radiol 14: 344–354. doi:10.1016/j.acra.2006.12.004
Wong K, Tang J, Zhang H, Varghese E, Cleary RK (2005) Prediction of 3D internal organ position from skin surface motion: results from electromagnetic tracking studies. Proc SPIE 5744: 879–887
Borgert J, Kruger S, Timinger H, Krucker J, Glossop N, Durrani A, Viswanathan A, Wood BJ (2006) Respiratory motion compensation with tracked internal and external sensors during CT-guided procedures. Comput Aided Surg 11: 119–125. doi:10.3109/10929080600740871
Khamene A, Warzelhan JK, Vogt S, Elgort D, Chefd’Hotel C, Duerk JL, Lewin J, Wacker FK, Sauer F (2004) Characterization of internal organ motion using skin marker positions. In: Medical image computing and computer-assisted intervention—MICCAI 2004, Pt 2, proceedings, vol 3217, pp 526–533
Kothary N, Dieterich S, Louie JD, Chang DT, Hofmann LV, Sze DY (2009) Percutaneous implantation of fiducial markers for imaging-guided radiation therapy. AJR Am J Roentgenol 192: 1090–1096. doi:10.2214/AJR.08.1399
Kothary N, Heit JJ, Louie JD, Kuo WT, Loo BW Jr, Koong A, Chang DT, Hovsepian D, Sze DY, Hofmann LV (2009) Safety and efficacy of percutaneous fiducial marker implantation for image-guided radiation therapy. J Vasc Interv Radiol 20: 235–239. doi:10.1016/j.jvir.2008.09.026
Bricault I, DiMaio S, Clatz O, Pujol S, Vosburgh K, Kikinis R (2005) Computer-assisted interventions on liver feasibility of the “anchor needle” technique. Surgetica 2005—computer-aided medical interventions: tools and applications, pp 348–352
Krücker J, Xu S, Glossop N, Pritchard FW, Karanian J, Chiesa A, Wood BJ (2008) Evaluation of motion compensation approaches for soft tissue navigation. Proc SPIE 6918: 691814
Maier-Hein L, Muller SA, Pianka F, Worz S, Muller-Stich BP, Seitel A, Rohr K, Meinzer HP, Schmied BM, Wolf I (2008) Respiratory motion compensation for CT-guided interventions in the liver. Comput Aided Surg 13: 125–138. doi:10.3109/10929080802091099
Maier-Hein L, Tekbas A, Franz AM, Tetzlaff R, Muller SA, Pianka F, Wolf I, Kauczor HU, Schmied BM, Meinzer HP (2008) On combining internal and external fiducials for liver motion compensation. Comput Aided Surg 13: 369–376. doi:10.3109/10929080802610674
Orozco M, Gorges S, Pescatore J (2008) Respiratory liver motion tracking during transcatheter procedures using guidewire detection. Int J CARS 3: 79–83. doi:10.1007/s11548-008-0214-9
Mortenson M (1985) Geometric modeling. Wiley, New York
Buchholz RH (1992) Perfect pyramids. Bull Austral Math Soc 45: 353–368
Shirabe K, Takenaka K, Gion T, Fujiwara Y, Shimada M, Yanaga K, Maeda T, Kajiyama K, Sugimachi K (1997) Analysis of prognostic risk factors in hepatic resection for metastatic colorectal carcinoma with special reference to the surgical margin. Br J Surg 84: 1077–1080
Elias D, Cavalcanti A, Sabourin JC, Lassau N, Pignon JP, Ducreux M, Coyle C, Lasser P (1998) Resection of liver metastases from colorectal cancer: the real impact of the surgical margin. Eur J Surg Oncol 24: 174–179
Hara W, Soltys SG, Gibbs IC (2007) CyberKnife robotic radiosurgery system for tumor treatment. Expert Rev Anticancer Ther 7: 1507–1515. doi:10.1586/14737140.7.11.1507
Rohlfing T, Maurer CR Jr, O’Dell WG, Zhong J (2004) Modeling liver motion and deformation during the respiratory cycle using intensity-based nonrigid registration of gated MR images. Med Phys 31: 427–432
Yaniv Z, Wilson E, Lindisch D, Cleary K (2009) Electromagnetic tracking in the clinical environment. Med Phys 36: 876–892
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lei, P., Moeslein, F., Wood, B.J. et al. Real-time tracking of liver motion and deformation using a flexible needle. Int J CARS 6, 435–446 (2011). https://doi.org/10.1007/s11548-010-0523-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11548-010-0523-7