Abstract
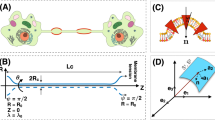
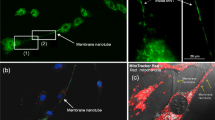
Tunneling nanotubes (TNTs) have previosly been observed as long and thin transient structures forming between cells and intercellular protein transfer through them has been experimentally verified. It is hypothesized that this may be a physiologically important means of cell–cell communication. This paper attempts to give a simple model for the rates of transfer of molecules across these TNTs at different distances. We describe the transfer of both cytosolic and membrane bound molecules between neighboring populations of cells and argue how the lifetime of the TNT, the diffusion rate, distance between cells, and the size of the molecules may affect their transfer. The model described makes certain predictions and opens a number of questions to be explored experimentally.




Similar content being viewed by others
References
Abou-Khalil, R., et al. (2009). Autocrine and paracrine angiopoietin 1/Tie-2 signaling promotes muscle satellite cell self-renewal. Cell Stem Cell, 5(3), 298–309.
Agnati, L. F., et al. (2011). Possible new targets for GPCR modulation: allosteric interactions, plasma membrane domains, intercellular transfer and epigenetic mechanisms. J. Recept. Signal Transduct. Res., 31(5), 315–331.
Ahmed, K. A., & Xiang, J. (2011). Mechanisms of cellular communication through intercellular protein transfer. J. Cell. Mol. Med., 15(7), 1458–1473.
Al-Nedawi, K., et al. (2008). Intercellular transfer of the oncogenic receptor EGFRvIII by microvesicles derived from tumour cells. Nat. Cell Biol., 10(5), 619–624.
Ambudkar, S. V., Sauna, Z. E., Gottesman, M. M., & Szakacs, G. (2005). A novel way to spread drug resistance in tumor cells: functional intercellular transfer of P-glycoprotein (ABCB1). Trends Pharmacol. Sci., 26(8), 385–387.
Baba, E., et al. (2001). Functional CD4 T cells after intercellular molecular transfer of 0X40 ligand. J. Immunol., 167(2), 875–883.
Behnke, B. J., Armstrong, R. B., & Delp, M. D. (2011). Adrenergic control of vascular resistance varies in muscles composed of different fiber types: influence of the vascular endothelium. Am. J. Physiol., Regul. Integr. Comp. Physiol., 301(3), R783–790.
Bosenberg, M. W., & Massague, J. (1993). Juxtacrine cell signaling molecules. Curr. Opin. Cell Biol., 5(5), 832–838.
Bukoreshtliev, N. V., et al. (2009). Selective block of tunneling nanotube (TNT) formation inhibits intercellular organelle transfer between PC12 cells. FEBS Lett., 583(9), 1481–1488.
Camussi, G., Deregibus, M. C., Bruno, S., Cantaluppi, V., & Biancone, L. (2010). Exosomes/microvesicles as a mechanism of cell-to-cell communication. Kidney Int., 78(9), 838–848.
Carlin, L. M., Eleme, K., McCann, F. E., & Davis, D. M. (2001). Intercellular transfer and supramolecular organization of human leukocyte antigen C at inhibitory natural killer cell immune synapses. J. Exp. Med., 194(10), 1507–1517.
Davis, D. M. (2007). Intercellular transfer of cell-surface proteins is common and can affect many stages of an immune response. Nat. Rev. Immunol., 7(3), 238–243.
Denzer, K., Kleijmeer, M. J., Heijnen, H. F., Stoorvogel, W., & Geuze, H. J. (2000). Exosome: from internal vesicle of the multivesicular body to intercellular signaling device. J. Cell Sci., 113(19), 3365–3374.
Driesen, R. B., et al. (2005). Partial cell fusion: a newly recognized type of communication between dedifferentiating cardiomyocytes and fibroblasts. Cardiovasc. Res., 68(1), 37–46.
Eugenina, E. A., Gaskilla, P. J., & Bermana, J. W. (2009). Tunneling nanotubes (TNT) are induced by HIV-infection of macrophages: A potential mechanism for intercellular HIV trafficking. Cell. Immunol., 254(2), 142–148.
Gregor, T., Bialek, W., de Ruyter van Steveninck, R. R., Tank, T. D., & Wieschaus, E. F. (2005). Diffusion and scaling during early embryonic pattern formation. Proc. Natl. Acad. Sci. USA, 102(51), 18403–18407.
Groebe, K., Erz, S., & Mueller-Klieser, W. (1994). Glucose diffusion coefficients determined from concentration profiles in EMT6 tumor spheroids incubated in radioactively labeled L-glucose. Adv. Exp. Med. Biol., 361, 619–625.
Guescini, M., et al. (2012). Microvesicle and tunneling nanotube mediated intercellular transfer of g-protein coupled receptors in cell cultures. In Experimental cell research.
Gurke, S., et al. (2008). Tunneling nanotube (TNT)-like structures facilitate a constitutive, actomyosin-dependent exchange of endocytic organelles between normal rat kidney cells. Exp. Cell Res., 314(20), 3669–3683.
Levchenko, A., et al. (2005). Intercellular transfer of P-glycoprotein mediates acquired multidrug resistance in tumor cells. Proc. Natl. Acad. Sci. USA, 102(6), 1933–1938.
Li, M., et al. (2010). Intercellular transfer of proteins as identified by stable isotope labeling of amino acids in cell culture. J. Biol. Chem., 285(9), 6285–6297.
Lichtenberger, B. M., et al. (2010). Autocrine VEGF signaling synergizes with EGFR in tumor cells to promote epithelial cancer development. Cell, 140(2), 268–279.
Lou, E., et al. (2012). Tunneling nanotubes provide a unique conduit for intercellular transfer of cellular contents in human malignant pleural mesothelioma. PLoS ONE, 7(3), e33093.
Luby-Phelps, K., Taylor, D. L., & Lanni, F. (1986). Probing the structure of cytoplasm. J. Cell Biol., 102(6), 2015–2022.
Mack, M., et al. (2000). Transfer of the chemokine receptor CCR5 between cells by membrane-derived microparticles: a mechanism for cellular human immunodeficiency virus 1 infection. Nat. Med., 6(7), 769–775.
Marzo, L., Gousset, K., & Zurzolo, C. (2012). Multifaceted roles of tunneling nanotubes in intercellular communication. Front. Physiol., 3, 72.
Nicholson, C., & Tao, L. (1993). Hindered diffusion of high molecular weight compounds in brain extracellular microenvironment measured with integrative optical imaging. Biophys. J., 65(6), 2277–2290.
Niu, X., Gupta, K., Yang, J. T., Shamblott, M. J., & Levchenko, A. (2009). Physical transfer of membrane and cytoplasmic components as a general mechanism of cell–cell communication. J. Cell Sci., 122(5), 600–610.
Pap, E., Pallinger, E., Pasztoi, M., & Falus, A. (2009). Highlights of a new type of intercellular communication: microvesicle-based information transfer. Inflamm. Res., 58(1), 1–8.
Phillips, G. N. Jr. (1997). Structure and dynamics of green fluorescent protein. Curr. Opin. Struct. Biol., 7(6), 821–827.
Prochiantz, A. (2011). Homeoprotein intercellular transfer, the hidden face of cell-penetrating peptides. Methods Mol. Biol., 683, 249–257.
Qin, L., Bromberg-White, J. L., & Qian, C. N. (2012). Opportunities and challenges in tumor angiogenesis research: back and forth between bench and bed. Adv. Cancer Res., 113, 191–239.
Quah, B. J., et al. (2008). Bystander B cells rapidly acquire antigen receptors from activated B cells by membrane transfer. Proc. Natl. Acad. Sci. USA, 105(11), 4259–4264.
Rustom, A., Saffrich, R., Markovic, I., Walther, P., & Gerdes, H. H. (2004). Nanotubular highways for intercellular organelle transport. Science, 303(5660), 1007–1010.
Singh, A. B., & Harris, R. C. (2005). Autocrine, paracrine and juxtacrine signaling by EGFR ligands. Cell. Signal., 17(10), 1183–1193.
Stine, M. J., et al. (2011). Integration of genotypic and phenotypic screening reveals molecular mediators of melanoma-stromal interaction. Cancer Res., 71(7), 2433–2444.
Twiss, J. L., & Fainzilber, M. (2009). Ribosomes in axons—scrounging from the neighbors? Trends Cell Biol., 19(5), 236–243.
Valadi, H., et al. (2007). Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat. Cell Biol., 9(6), 654–659.
Vikne, H., Gundersen, K., Liestol, K., Maelen, J., & Vollestad, N. (2012). Intermuscular relationship of human muscle fiber type proportions: slow leg muscles predict slow neck muscles. Muscle Nerve, 45(4), 527–535.
Weis, S. M., & Cheresh, D. A. (2011). Tumor angiogenesis: molecular pathways and therapeutic targets. Nat. Med., 17(11), 1359–1370.
Author information
Authors and Affiliations
Corresponding author
Additional information
Y. Suhail and Kshitiz contributed equally to the manuscript.
Rights and permissions
About this article
Cite this article
Suhail, Y., Kshitiz, Lee, J. et al. Modeling Intercellular Transfer of Biomolecules Through Tunneling Nanotubes. Bull Math Biol 75, 1400–1416 (2013). https://doi.org/10.1007/s11538-013-9819-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11538-013-9819-4