Abstract
Background
Leukemia stem cells (LSCs) are considered to be the cause of treatment failure and relapse in acute myeloid leukemia (AML). Overexpression of the Bcl-2 family of anti-apoptotic proteins such as Bcl-2, Bcl-xl, and Mcl-1 accounts for survival and self-renewal of LSCs. AT101 binds to the BH3 motif of all Bcl-2 family anti-apoptotic proteins and demonstrates anti-tumor activity in multiple types of tumor. Thus, we hypothesized that this agent might have the potential to deplete LSCs.
Objective
The present study aims to investigate if and by what mechanism AT101 is able to target AML stem-like cells.
Methods
As LSCs and hematopoietic stem cells (HSCs) are enriched in CD34+CD38− populations, CD34+CD38− cells from KG1α and Kasumi-1 cell lines as well as CD34+ blasts from AML patients were used as LSC models, while CD34+ cells from healthy donors were used as normal hematopoietic cells. Cell proliferation and apoptosis were assessed by a cell counting kit-8 (CCK8) assay and an Annexin V/PI assay using flow cytometry, respectively. Colony-forming units experiments were performed to monitor the stemness features of AML cells. Western blot and quantitative real-time polymerase chain reaction (qPCR) analysis were performed to examine the levels of proteins and mRNAs related to either the intrinsic apoptotic pathway or DNA damage response.
Results
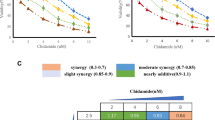
AT101 inhibited proliferation and induced apoptosis in CD34+CD38− KG1α and Kasumi-1 cells in a dose- and time-dependent manner. Exposure to AT101 for 24 h resulted in apoptosis in primary CD34+ AML blasts (EC50 [concentration needed for a 50% maximal effect] = 2.45–76.00 μmol/L), while it only had a modest effect on normal CD34+ hematopoietic cells. Mechanistically, AT101 activated the intrinsic apoptotic pathway by inhibition of Bcl-2 anti-apoptotic proteins, reflected by a decrease in mitochondrial membrane potential. Moreover, AT101 caused DNA damage (e.g., increased γH2AX phosphorylation), which might also contribute to its anti-leukemic effects. Interestingly, the ex vivo efficacy of AT101 in primary AML samples significantly correlated to hyperleukocytosis and FLT3-ITD mutations. AT101 was also effective against CD34+ blasts isolated from elderly patients and patients who did not achieve complete remission after induction therapy.
Conclusions
AT101 effectively eliminates LSCs in vitro through the induction of DNA damage and activation of the intrinsic apoptotic pathway. AT101 is effective towards leukemic cells from patients with adverse prognostic factors, suggesting that AT101 could have the potential as an alternative salvage therapy for the treatment of relapsed and refractory AML.






Similar content being viewed by others
References
Klco JM, Miller CA, Griffith M, et al. Association between mutation clearance after induction therapy and outcomes in acute myeloid leukemia. JAMA. 2015;314:811–22.
Walter RB, Kantarjian HM, Huang X, et al. Effect of complete remission and responses less than complete remission on survival in acute myeloid leukemia: a combined eastern cooperative oncology group, southwest oncology group, and M. D. Anderson cancer center study. J. Clin Oncol. 2010;28:1766–71.
Hu Y, Li S. Survival regulation of leukemia stem cells. Cell Mol Life Sci. 2016;5:1039–50.
Vo T, Ryan J, Carrasco R, et al. Relative mitochondrial priming of myeloblasts and normal HSCs determines chemotherapeutic success in AML. Cell. 2012;151:344–55.
Guzman ML, Allan JN. Concise review: leukemia stem cells in personalized medicine. Stem Cells. 2014;32:844–51.
Goff DJ, Recart AC, Sadarangani A, et al. A pan-BCL2 inhibitor renders bone-marrow-resident human leukemia stem cells sensitive to tyrosine kinase inhibition. Cell Stem Cell. 2013;12:316–28.
Domingo-Domenech J, Vidal SJ, Rodriguez-Bravo V, et al. Suppression of acquired docetaxel resistance in prostate cancer through depletion of notch- and hedgehog-dependent tumor-initiating cells. Cancer Cell. 2012;22:373–88.
Colak S, Zimberlin CD, Fessler E, et al. Decreased mitochondrial priming determines chemoresistance of colon cancer stem cells. Cell Death Differ. 2014;21:1170–7.
Lagadinou ED, Sach A, Callahan K, et al. BCL-2 inhibition targets oxidative phosphorylation and selectively eradicates quiescent human leukemia stem cells. Cell Stem Cell. 2013;12:329–41.
Vela L, Marzo I. Bcl-2 family of proteins as drug targets for cancer chemotherapy: the long way of BH3 mimetics from bench to bedside. Curr Opin Pharmacol. 2015;23:74–81.
Balakrishnan K, Burger JA, Wierda WG, et al. AT-101 induces apoptosis in CLL B cells and overcomes stromal cell-mediated mcl-1 induction and drug resistance. Blood. 2009;113:149–53.
Paulus A, Advani P, Laplant BR, et al. Phase I/II clinical trial of Lenalidomide in combination with AT101 for the treatment of relapsed B-cell chronic lymphocytic leukemia (B-CLL). Blood. 2015;126:5299.
Stein MN, Hussain M, Stadler WM, et al. A phase II study of AT-101 to overcome Bcl-2 mediated resistance to androgen deprivation therapy in patients with newly diagnosed castration sensitive metastatic prostate cancer. Clin Genitourin Canc. 2015;14:22–7.
Baggstrom MQ, Qi Y, Koczywas M, et al. A phase II study of AT-101 (gossypol) in chemotherapy-sensitive recurrent extensive stage small cell lung cancer (ES-SCLC). J Thorac Oncol. 2011;6:1757.
Kaza N, Kohli L, Graham CD, et al. BNIP3 regulates AT101 [(−)-gossypol] induced death in malignant peripheral nerve sheath tumor cells. PLoS One. 2014;9:e96733.
Hu W, Wang F, Tang J, et al. Proapoptotic protein Smac mediates apoptosis in cisplatin-resistant ovarian cancer cells when treated with the anti-tumor agent AT101. J Biol Chem. 2012;287:68–80.
Li G, Liu L, Shan C, et al. RhoA/ROCK/PTEN signaling is involved in AT-101-mediated apoptosis in human leukemia cells in vitro and in vivo. Cell Death Disease. 2014;5:e998.
Reinisch A, Chan SM, Thomas D, et al. Biology and clinical relevance of acute myeloid leukemia stem cells. Semin Hematol. 2015;52:150–64.
Zeijlemaker W, Kelder A, Oussoren-Brockhoff YJM, et al. A simple one-tube assay for immunophenotypical quantification of leukemic stem cells in acute myeloid leukemia. Leukemia. 2016;30:439.
Rao J, Xu DR, Zheng FM, et al. Curcumin reduces expression of Bcl-2, leading to apoptosis in daunorubicin-insensitive CD34+ acute myeloid leukemia cell lines and primary sorted CD34+ acute myeloid leukemia cells. J Transl Med. 2011;9:71.
She M, Niu X, Chen X, et al. Resistance of leukemic stem-like cells in AML cell line KG1a to natural killer cell-mediated cytotoxicity. Cancer Lett. 2012;318:173–9.
Liu Y, Chen F, Wang S, et al. Low-dose triptolide in combination with idarubicin induces apoptosis in AML leukemic stem-like KG1a cell line by modulation of the intrinsic and extrinsic factors. Cell Death Disease. 2013;4:e948.
Li Y, Chen K, Zhou Y, et al. A new strategy to target acute myeloid leukemia stem and progenitor cells using Chidamide, a histone deacetylase inhibitor. Curr Cancer Drug Targets. 2015;15:493–503.
Quotti TL, Canovas NS, Brancalion A, et al. Protein kinase CK2 regulates AKT, NF-kappaB and STAT3 activation, stem cell viability and proliferation in acute myeloid leukemia. Leukemia. 2017;31:292–300.
Ozguner M, Azik MF, Tavil B, et al. Do two different stem cell grafts: G-CSF stimulated and unstimulated bone marrow differ according to hematopoietic colony forming capacity? Transfus Apher Sci. 2014;50:467–72.
Ahmed T, Holwerda S, Klepin HD, et al. High dose cytarabine, mitoxantrone and l-asparaginase (HAMA) salvage for relapsed or refractory acute myeloid leukemia (AML) in the elderly. Leukemia Res. 2015;39:945–9.
Colin DJ, Hain KO, Allan LA, et al. Cellular responses to a prolonged delay in mitosis are determined by a DNA damage response controlled by Bcl-2 family proteins. Open Biol. 2015;5:140156.
Jamil S, Stoica C, Hackett T, et al. MCL-1 localizes to sites of DNA damage and regulates DNA damage response. Cell Cycle. 2014;9:2915–27.
Pan M, Peng G, Hung W, et al. Monoubiquitination of H2AX protein regulates DNA damage response signaling. J Biol Chem. 2011;286:28599–607.
Ichim G, Lopez J, Ahmed SU, et al. Limited mitochondrial Permeabilization causes DNA damage and genomic instability in the absence of cell death. Mol Cell. 2015;57:860–72.
Delbridge AR, Grabow S, Strasser A, et al. Thirty years of BCL-2: translating cell death discoveries into novel cancer therapies. Nat Rev Cancer. 2016;16:99–109.
Bogenberger JM, Kornblau SM, Pierceall WE, et al. BCL-2 family proteins as 5-Azacytidine-sensitizing targets and determinants of response in myeloid malignancies. Leukemia. 2014;28:1657–65.
Soderquist R, Bates DJP, Danilov AV, et al. Gossypol overcomes stroma-mediated resistance to the BCL2 inhibitor ABT-737 in chronic lymphocytic leukemia cells ex vivo. Leukemia. 2013;27:2262–4.
Pan R, Ruvolo VR, Wei J, et al. Inhibition of mcl-1 with the pan-Bcl-2 family inhibitor (−)BI97D6 overcomes ABT-737 resistance in acute myeloid leukemia. Blood. 2015;126:363–72.
Yoshimoto G, Miyamoto T, Jabbarzadeh-Tabrizi S, et al. FLT3-ITD up-regulates MCL-1 to promote survival of stem cells in acute myeloid leukemia via FLT3-ITD-specific STAT5 activation. Blood. 2009;114:5034–43.
Frohling S, Scholl C, Levine RL, et al. Identification of driver and passenger mutations of FLT3 by high-throughput DNA sequence analysis and functional assessment of candidate alleles. Cancer Cell. 2007;12:501–13.
Walter RB, Estey EH. Management of older or unfit patients with acute myeloid leukemia. Leukemia. 2014;29:770–5.
Sadahira K, Sagawa M, Nakazato T, et al. Gossypol induces apoptosis in multiple myeloma cells by inhibition of interleukin-6 signaling and Bcl-2/mcl-1 pathway. Int J Oncol. 2014;45:2278–86.
Meng Y, Tang W, Dai Y, et al. Natural BH3 mimetic (−)-gossypol chemosensitizes human prostate cancer via Bcl-xL inhibition accompanied by increase of puma and Noxa. Mol Cancer Ther. 2008;7:2192–202.
Weiss C, Ito K. DNA damage: a sensible mediator of the differentiation decision in hematopoietic stem cells and in leukemia. Int J Mol Sci. 2015;16:6183–201.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Funding
This work is supported by the National Natural Science Foundation of China (No. 81570156, No. 81428003 and No. 81400104), the Natural Science Foundation of Guangdong Province(No. 2014A030313355), and the Guangdong Provincial Basic Research Program, PR China (No. 2015B020227003).
Conflict of Interest
The authors declare that they have no conflicts of interest.
Electronic supplementary material
ESM 1
(PDF 109 kb)
Rights and permissions
About this article
Cite this article
Zhang, L., Zhou, Y., Chen, K. et al. The pan-Bcl2 Inhibitor AT101 Activates the Intrinsic Apoptotic Pathway and Causes DNA Damage in Acute Myeloid Leukemia Stem-Like Cells. Targ Oncol 12, 677–687 (2017). https://doi.org/10.1007/s11523-017-0509-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11523-017-0509-2