Abstract
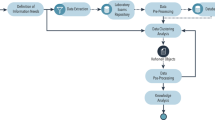
Pulmonary tuberculosis (PTB) remains a worldwide public health problem. Diagnostic algorithms to identify the best combination of diagnostic tests for PTB in each setting are needed for resource optimization. We developed one artificial neural network model for classification (multilayer perceptron—MLP) and another risk group assignment (self-organizing map—SOM) for PTB in hospitalized patients in a high complexity hospital in Rio de Janeiro City, using clinical and radiologic data collected from 315 presumed PTB cases admitted to isolation rooms from March 2003 to December 2004 (TB prevalence = 21.5 %). The MLP model included 7 variables—radiologic classification, age, gender, cough, night sweats, weight loss and anorexia. The sensitivity of the MLP model was 96.0 % (95 % CI ±2.0), the specificity was 89.0 % (95 % CI ±2.0), the positive predictive value was 72.5 % (95 % CI ±3.5) and the negative predictive value was 98.5 % (95 % CI ±0.5). The variable with the highest discriminative power was the radiologic classification. The high negative predictive value found in the MLP model suggests that the use of this model at the moment of hospital admission is safe. SOM model was able to correctly assign high-, medium- and low-risk groups to patients. If prospective validation in other series is confirmed, these models can become a tool for decision-making in tertiary health facilities in countries with limited resources.





Similar content being viewed by others
References
Adams ST, Leveson SH (2012) Clinical prediction rules. BMJ 16(344):d8312. doi:10.1136/bmj.d8312
Aguiar FS, Almeida LL, Ruffino-Netto A, Kritski AL, Mello FC, Werneck GL (2012) Classification and regression tree (CART) model to predict pulmonary tuberculosis in hospitalized patients. BMC Pulm Med. 7(12):40. doi:10.1186/1471-2466-12-40
Barnes PF, Verdegem TD, Vachon LA, Leedom JM, Overturf GD (1988) Chest roentgenogram in pulmonary tuberculosis. New data on an old test. Chest 94(2):316–320
Bock NN, McGowan JE Jr, Ahn J, Tapia J, Blumberg HM (1996) Clinical predictors of tuberculosis as a guide for a respiratory isolation policy. Am J Respir Crit Care Med 154(5):1468–1472
Centers for Disease Control. Guidelines for Preventing the Transmission of Mycobacterium tuberculosis in Health-Care Settings, 2005. MMWR December 30, 2005/Vol 54(RR17); pp 1–141
da Costa PA, Trajman A, Mello FC, Goudinho S, Silva MA, Garret D, Ruffino-Netto A, Kritski AL (2009) Administrative measures for preventing Mycobacterium tuberculosis infection among healthcare workers in a teaching hospital in Rio de Janeiro, Brazil. J Hosp Infect 72(1):57–64. doi:10.1016/j.jhin.2009.01.016 (Epub 2009 Mar 17)
El-Solh A, Mylotte J, Sherif S, Serghani J, Grant BJB (1997) Validity of a decision tree for predicting active pulmonary tuberculosis. Am J Respir Crit Care Med 155:1711–1716
El-Solh AA, Hsiao CB, Goodnough S, Serghani J, Grant BJ (1999) Predicting active pulmonary tuberculosis using an artificial neural network. Chest 116(4):968–973
Elveren E, Yumusak N (2011) Tuberculosis disease diagnosis using artificial neural network trained with genetic algorithm. J Med Syst 35(3):329–332. doi:10.1007/s10916-009-9369-3 (Epub 2009 Aug 28)
Er O, Termutas F, Tanrikulu AC (2010) Tuberculosis disease diagnosis using artificial neural networks. J Med Syst 34(3):299–302
Faber V (1994) Clustering and the Continuous k-means Algorithm Los Alamos. Science 22:138–144
Harries AD, Kamenya A, Subramanyam VR, Maher D, Squire SB, Wirima JJ, Nyangulu DS, Nunn P (1997) Screening pulmonary tuberculosis suspects in Malawi: testing different strategies. Trans R Soc Trop Med Hyg 91(4):416–419
Haykin S (2008) Neural networks and learning machines, 3rd edn, ISBN 2008
Hogeweg L, Mol C, de Jong PA, Dawson R, Ayles H, van Ginneken B (2010) Fusion of local and global detection systems to detect tuberculosis in chest radiographs. Med Image Comput Comput Assist Interv. 13(Pt 3):650–657
Keeler E, Perkins MD, Small P, Hanson C, Reed S, Cunningham J, Aledort JE, Hillborne L, Rafael ME, Girosi F, Dye C (2006) Reducing the global burden of tuberculosis: the contribution of improved diagnostics. Nature 23(444 Suppl 1):49–57
Kohonen T (2000) Self-organizing maps. Springer
Lagrange-X’elot M, Porcher R, Gallien S, Wargnier A, Pavie J, de Castro N, Molina J-M (2010) Prevalence and clinical predictors of pulmonary tuberculosis among isolated inpatients: a prospective study. Clin Microbiol Infect 17(4):610–614. doi:10.1111/j.1469-0691.2010.03259.x
Liao L, Mark DB (2003) Clinical prediction models: are we building better mousetraps? J Am Coll Cardiol 42(5):851–853
Luna JA. A tuberculosis guide for specialist physicians. International Union Against Tuberculosis and Lung Disease (IUATLD), 2004. http://www.uphs.upenn.edu/bugdrug/antibiotic_manual/TBGuideForSpecialistPhysicians-complete.pdf. Accessed 20 June 2015
Maduskar P, Muyoyeta M, Ayles H, Hogeweg L, Peters-Bax L, van Ginneken B (2013) Detection of tuberculosis using digital chest radiography: automated reading versus interpretation by clinical officers. Int J Tuberc Lung Dis. 17(12):1613–1620. doi:10.5588/ijtld.13.0325
Mello FC, Bastos LG, Soares SL, Rezende VM, Conde MB, Chaisson RE, Kritski AL, Ruffino-Netto A, Werneck GL (2006) Predicting smear negative pulmonary tuberculosis with classification trees and logistic regression: a cross-sectional study. BMC Public Health. 6:43
Pai M, Minion J, Steingart K, Ramsay A (2010) New and improved tuberculosis diagnostics: evidence, policy, practice, and impact. Curr Opin Pulm Med. 16(3):271–284. doi:10.1097/MCP.0b013e328338094f
Riedmiller M, Braun H (1993) A direct adaptive method for faster back- propagation learning: the RPROP algorithm. In: Proceedings of the IEEE international conference on neural networks, San Francisco, USA, pp. 586–591
Rutjes AW, Reitsma JB, Di Nisio M, Smidt N, van Rijn JC, Bossuyt PM (2006) Evidence of bias and variation in diagnostic accuracy studies. CMAJ 174(4):469–476
Santos AM, Pereira BB, Seixas JM, Mello FCQ, Kritski AL (2006) Neural networks: an application for predicting smear negative pulmonary tuberculosis. In: Jean-Louis A, Balakrishnan N, Mounir M, Geert M (eds) Advances in statistical methods for the health sciences applications to cancer and AIDS studies, genome sequence analysis, and survival analysis. Birkhäuser, Boston, pp 279–292
Seixas JM, Calôba LP, Delpino I (1996) Relevance criteria for variable selection in classifier design. In: International conference on engineering applications of neural networks, London, England, pp 451–454
Seixas JM, Faria J, Souza-Filho JB, Vieira AF, Kritski A, Trajman A (2013) Artificial neural network models to support the diagnosis of pleural tuberculosis in adult patients. Int J Tuberc Lung Dis. 17(5):682–686. doi:10.5588/ijtld.12.0829
Sester M, Giehl C, McNerney R, Kampmann B, Walzl G, Cuchí P, Wingfield C, Migliori GB, Kritski AL, Meyerhans A (2010) Challenges and perspectives for improved management of HIV/TB coinfection. Eur Respir J 36:1242–1247
Solari L, Acuna-Villaorduna C, Soto A, van der Stuyft P (2011) Evaluation of clinical prediction rules for respiratory isolation of inpatients with suspected pulmonary tuberculosis. Clin Infect Dis 52(5):595–603
Steingart KR, Schiller I, Horne DJ, Pai M, Boehme CC, Dendukuri N (2014) Xpert® MTB/RIF assay for pulmonary tuberculosis and rifampicin resistance in adults. Cochrane Database Syst Rev 1:CD009593. doi:10.1002/14651858.CD009593.pub3
Telles MA, Kritski AL. Biosafety and hospital control. In: Palomino-Leão-Ritacco. Tuberculosis 2007. From basic science to patient care. Available at http://www.pneumo-iasi.ro/student/tuberculosis2007.pdf. Accessed 20 June 2015
Tuberculosis Chemotherapy Centre (1959) Madras. A concurrent comparison of home and sanatorium treatment of pulmonary tuberculosis in South India. Bull World Health Organ 21(1):51–144
Uplekar Mukund, Raviglione Mario (2012) Hospital or home? Scripting a high point in the history of TB care and control. WHO South-East Asia J Public Health 1(2):220–223
Wang CS, Chen HC, Chong IW, Hwang MS (2008) Predictors for identifying the most infectious pulmonary tuberculosis patient. J Formos Med Assoc 107(1):13–20. doi:10.1016/S0929-6646(08)60003-0
Wisnivesky JP, Kaplan J, Henschke C, McGinn TG, Crystal RG (2000) Evaluation of clinical parameters predicts Mycobacterium tuberculosis in inpatients. Arch Intern Med 160:2471–2476
Wisnivesky JP, Henschke C, Balentine J, Willner C, Deloire AM, McGinn TG (2005) Prospective validation of a prediction model for isolating inpatients with suspected pulmonary tuberculosis. Arch Intern Med 165:453–457
World Health Organization. Global Tuberculosis Report 2015. World Health Organization. ISBN 978 92 4 156 5059
Acknowledgments
This work was supported by Fundação de Amparo à Pesquisa do Estado do Rio de Janeiro (FAPERJ) Process: Nº E: 26/111608/2008. AK, JMS and FCQM are recipients of a career award from Conselho Nacional de Desenvolvimento CIentífico e Tecnológico (CNPq) (produtividade em pesquisa) and FAPERJ (Cientistas do Nosso Estado). The funders had no role in study design, data collection and analysis, the decision to publish, or the preparation of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors report no conflicts of interest relevant to this article
Rights and permissions
About this article
Cite this article
Aguiar, F.S., Torres, R.C., Pinto, J.V.F. et al. Development of two artificial neural network models to support the diagnosis of pulmonary tuberculosis in hospitalized patients in Rio de Janeiro, Brazil. Med Biol Eng Comput 54, 1751–1759 (2016). https://doi.org/10.1007/s11517-016-1465-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11517-016-1465-1