Abstract
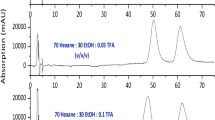
Separation of betaxolol, bevantolol, metoprolol, bisoprolol with new bonded chiral stationary (3,5-dimethylcabamate cellulose) Chiralpak IB column was investigated. The factors, such as mobile phase composition and the ratio, column temperature, flow rate and the proportion of chiral additives, were also investigated. The optimal mobile phase compositions of betaxolol, bevantolol, metoprolol, bisoprolol were n-hexane/ethanol (95/5, v/v; 0.2% DEA); n-hexane/ethanol (90/10, v/v; 0.1% DEA); hexane/ethanol (60/40, v/v; 0.1% DEA); n-hexane/ethanol (95/5, v/v; 0.2% DEA), respectively. The optimal temperature was 30°C with a flow rate of 0.8 mL min−1. The detection wavelength, by 1200VWD UV detector, of betaxolol, bevantolol, metoprolol, bisoprolol was 259, 274, 224 and 223 nm, respectively. Experimental results demonstrated that baseline separation (R s>1.5) of the betaxolol, bevantolol, metoprolol, bisoprolol enantiomers were obtained with new bonded Chiralpak IB column. This method can be used for analysis and detection of these four drugs.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Gronefeld G C, Bansch D. Antiarrhythmische therapie mit β-rezeptor- antagonisten. Herzschrittmacherther Elektrophys, 2010, 21: 222–227
Chu Y B. Separation of nine chiral drugs by non-aqueous capillary electrophoresis. Anal Chem, 2003, 21: 138–142
Li G B, Lin X L, Zhu C F, et al. Separation of enantiomers by capillary electrophoresis using L-glutamine as chiral selecter. Anal Chem, 2000, 28: 1287–1290
Chen Z Y, Xia Z L, Hu C Q, et al. Separation of bisoprolol, atenolol, clenbuterol and terbutaline. Anal Chem Research Report, 2007, 35: 181–186
Ding G S, Liu Y, Cong R, et al. Study on the separation of β2-receptor blockers and its analogue using norvancomycin bonded chiral stationary phase. Chin J Chromatogr, 2004, 22: 386–389
Yu L S, Yao T W, Wang X J. Separation of β2-receptor blockers and its analogue by chiral stationary phase chiral derivatization method. J Zhejiang Univ (Medical Sciences), 2002, 31: 414–428
Zhang Y H, Zou X R, Yun Z H. Separation of β2-receptor blockers enantiomers by amide chiral stationary phase. J Instr Anal, 1999, 18: 75–77
Ceccato A, Hubert P, Crommen J. Direct liquid chromatographic enantioseparation of sotaloland other β-blockers using an β1-glycoprotein- based chiral stationary phase. Chromatogr A, 1997, 760: 193–203
Petersen P V, Ekelund J, Olsen L, et al. Chiral separations of β- blocking drug substancesusing the Pirkle-type α-Burke 1 chiral stationary phase. Chromatogr A, 1997, 757: 65–71
Yang G C, Chen J D. Chemical ferment catalysis separation of two kinds of betaxolol enantiomers. Foreign pharmaceutical·Synthetic drugs-Biochemical drugs. Preparation Fascicule, 1996, 17: 318
Darmon A, Thenot J P. Determination of betaxolol enantiomers by high-performance liquid Chromatography. Chromatogr B, 1986, 374: 321–328
Krstulovic A M, Fouchet M H, Burke J T, et al. Driect enantiomeric separation of betaxolol with applications to analysis of bulk drug and biological samples. Chromatogr A, 1988, 452: 477–483
Ekelund J, Arkens A, Bronnum-Hansen K, et al. Chiral separations of β-blocking drug substances using chiral stationary phases. Chromatogr A, 1995, 708: 253–261
Zhang C, Jin L, Zhou S, et al. Chiral separation of neonicotinoid insecticides by polysaccharide-type stationary phases using high-performance liquid chromatography and supercritical fluid chromatography. Chirality, 2011, 23: 215–221
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at Springerlink.com
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Zhou, J., Pei, W., Su, N. et al. Separation of four -lol drugs by HPLC with new bonded chiral stationary column. Chin. Sci. Bull. 58, 1267–1273 (2013). https://doi.org/10.1007/s11434-012-5636-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11434-012-5636-7