Abstract
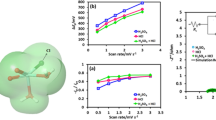
The composition of electrolyte affects to a great extent the electrochemical performance of vanadium redox flow batteries (VRB). The effects of Cr3+ concentration in the anolyte on the electrode process of V(V)/V(IV) couple have been investigated by cyclic voltammetry (CV) and electrochemical impedance spectroscopy (EIS). It was found that Cr3+ causes no side reactions, but affects the electrochemical performance of V(V)/V(IV) redox reaction, including the reaction activity, the reversibility of electrode reaction, the diffusivity of vanadium ions, the interface film impedance, and the electrode reaction impedance. The experimental results show that Cr3+ within a certain concentration range can improve the reversibility of electrode reaction and the diffusion of vanadium ions. With the Cr3+ concentration increasing from 0 to 0.30 g L−1, the reversibility of V(V)/V(IV) reaction increases, while the diffusion resistance decreases. Correspondingly, the diffusion coefficient of vanadium ions increases from (5.48–6.77) × 10−7 to (6.82–8.44) × 10−7 cm2 s−1, an increase of ∼24%. However, the diffusion resistance increases and the diffusion coefficient decreases when Cr3+ concentration is over 0.30 g L−1, while the impedances of the interface, the film as well as the charge transfer increase continuously. As a result, Cr3+ with a certain concentration improves the diffusion and mass transfer process, but the resistances of the film, the interface, and the charge transfer rise. Furthermore, Cr3+ concentration of no more than 0.10 g L−1 has few effect on the electrode reaction process, and that of no more than 0.30 g L−1 is favorable to the diffusion of vanadium ions.
Article PDF
Similar content being viewed by others
References
Li X F, Zhang H M, Mai Z S, et al. Ion exchange membranes for vanadium redox flow battery (VRB) applications. Energy Environ Sci, 2011, 4: 1147–1160
Skyllas-Kazacos M, Sylvania H, Miron R. All-vanadium redox battery. US Patent, 4786567, 1988-11-22
Huang K L, Li X G, Liu S Q, et al. Research progress of vanadium redox flow battery for energy storage in China. Renew Energy, 2008, 33: 186–192
Huang F, Wang G X, Yan K P, et al. Influence of Mn2+ concentration on the electrochemical behavior of the anolyte for vanadium redox flow batteries. Chin J Inorg Chem, 2012, 28: 898–904
Liu Q H, Shinkle A A, Li Y D, et al. Non-aqueous chromium acetylacetonate electrolyte for redox flow batteries. Electrochem Commun, 2010, 12: 1634–1637
Gaku O, Yasushi K, Takashi M. Investigation on V(IV)/V(V) species in a vanadium redox flow battery. Electrochim Acta, 2004, 49: 3091–3095
Tang A, Bao J, Skyllas-Kazacos M. Dynamic modelling of the effects of ion diffusion and side reactions on the capacity loss for vanadium redox flow battery. J Power Sources, 2011, 196: 10737–10747
Kear G, Shah A A, Walsh F C. Development of the all-vanadium redox flow battery for energy storage: A review of technological, financial and policy aspects. Int J Energy Res, 2012, 36: 1105–1120
Qiu G, Abhijit S J, Dennison C R, et al. 3-D pore-scale resolved model for coupled species/charge/uid transport in a redox ow battery vanadium. Electrochim Acta, 2012, 64: 46–64
González Z, Sánchez A, Blanco C, et al. Enhanced performance of a Bi-modified graphite felt as the positive electrode of a vanadium redox flow battery. Electrochem Commun, 2011, 13: 1379–1382
Chang F, Hu C W, Liu X J. Coulter dispersant as positive electrolyte additive for the vanadium redox flow battery. Electrochim Acta, 2012, 60: 334–338
Wang C, Chen X M, Hei P X, et al. Digital simulation of cyclic voltammetry for reversible, quasi-reversible and irreversible surface reaction. Chem J Chin Univ, 1991, 12: 875–878
Bard A J, Faulkner L R. Electrochemical Methods—Fundamentals and Applications. New York: Wiley, 2001. 228–238
Faizur R, Maria S-K. Vanadium redox battery: Positive half-cell electrolyte studies. J Power Sources, 2009, 189: 1212–1219
Weber A Z, Mench M M, Meyers J P, et al. Redox flow batteries: A review. J Appl Electrochem, 2011, 41: 1137–1164
Sun C X, Chen J, Zhang H M, et al. Investigations on transfer of water and vanadium ions across Nafion membrane in an operating vanadium redox flow battery. J Power Sources, 2010, 195: 890–897
Kim H S. Electrochemical properties of graphite-based electrodes for redox flow batteries. Korean Chem Soc, 2011, 32: 571–575
Marina P, Tadeusz H J. Electrochemistry of vanadium electrodes. Part 1. Cyclic voltammetry in aqueous solutions. Electro Anal Chem, 1995, 38: 137–144
Zhuang Q C, Tian L L, Wei G Z, et al. Two- and three-electrode impedance spectroscopic studies of graphite electrode in the first lithiation. Chin Sci Bull, 2009, 54: 1233–1237
Tang C, Zhou D B. Methanesulfonic acid solution as supporting electrolyte for zinc-vanadium redox battery. Electrochim Acta, 2012, 65: 179–184
Skyllas-Kazacos M, Kazacos M. State of charge monitoring methods for vanadium redox flow battery control. J Power Sources, 2011, 196: 8822–8827
Shao Y Y, Wang X Q, Engelhard M, et al. Nitrogen-doped mesoporous carbon for energy storage in vanadium redox flow batteries. J Power Sources, 2010, 195: 4375–4379
Han P X, Wang H B, Liu Z H, et al. Graphene oxide nanoplatelets as excellent electrochemical active materials for VO2+/VO2 + and V2+/V3+ redox couples for a vanadium redox flow battery. Carbon, 2011, 49: 693–700
Li W W, You W S, Han X M, et al. Synthesis, crystal structure and electrochemical properties of 3D coordinated polymers based on {VO3}n −n and {M(bpp)(H2O)2}n 2n+ chains (in Chinese). Chin Sci Bull (Chin Ver), 2011, 56: 919–925
Zhang J L, Li L Y, Nie Z M, et al. Effects of additives on the stability of electrolytes for all-vanadium redox flow batteries. J Appl Electrochem, 2011, 41: 1215–1221
Vijayakumar M, Burton S D, Huang C, et al. Nuclear magnetic resonance studies on vanadium (IV) electrolyte solutions for vanadium redox flow battery. J Power Sources, 2010, 195: 7709–7717
Araujo T S, Souza S O, Sousa E M B, et al. Effect of Zn2+, Fe3+ and Cr3+ addition to hydroxyapatite for its application as an active constituent of sunscreens. J Physics: Conference Series, 2010, 249: 012012
Dudnikova V B, Zharikov E V, Eremin N N, et al. Vanadium distribution between forsterite and its melt: The structural and oxidation state of vanadium. Geochem Internat, 2001, 39: 667–675
Hao L, Zhang S X, Dong J H, et al. Evolution of corrosion of MnCuP weathering steel submitted to wet/dry cyclic tests in a simulated coastal atmosphere. Corros Sci, 2012, 58: 175–180
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at Springerlink.com
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Huang, F., Zhao, Q., Luo, C. et al. Influence of Cr3+ concentration on the electrochemical behavior of the anolyte for vanadium redox flow batteries. Chin. Sci. Bull. 57, 4237–4243 (2012). https://doi.org/10.1007/s11434-012-5302-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11434-012-5302-0