Abstract
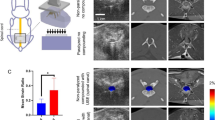
Many studies have shown that strategies of nerve regeneration and cell-based transplantation are valid based on animal models of spinal cord injury (SCI). To apply these strategies and bridge spinal cord defects, the identification and precise localization of lesions during spinal cord surgery is necessary. The aim of the present experiment was to evaluate the capabilities of ultrasound backscatter microscopy (UBM) in identifying morphologic changes after SCI. After laminectomy, high-resolution ultrasound images of the spinal cord were obtained in one normal and seven spinal cord-injured adult Wistar rats using a UBM system with a 55-MHz center frequency scanner. Comparison between histoanatomic and UBM images was also performed. The results showed that UBM can identify cysts after the experimental SCI is removed in adult rats. In addition, the glial scar formed in secondary injury showed obvious hyperechoic speckle in the UBM image and correlated with the histoanatomic image. UBM has obvious clinical value in nerve regeneration and cell-based transplantation strategies in injured spinal cords.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Ramon-Cueto A, Cordero M I, Santos-Benito F F, et al. Functional recovery of paraplegic rats and motor axon regeneration in their spinal cords by olfactory ensheathing glia. Neuron, 2000, 25: 425–435
Teng Y D, Lavik E B. Functional recovery following traumatic spinal cord injury mediated by a unique polymer scaffold seeded with neural stem cells. Proc Natl Acad Sci USA, 2002, 99: 3024–3029
Li X G, Yang Z Y, Yang Y. Morphological and electrophysiological evidence for regeneration of transected spinal cord fibers and restoration of motor functions in adult rats. Chin Sci Bull, 2006, 51: 918–926
Fawcett J W. Overcoming inhibition in the damaged spinal cord. J Neurotrauma, 2006, 23: 371–383
Stichel C C, Hans W M. Experimental strategies to promote axonal regeneration after traumatic central nervous system injury. Prog Neurobiol, 1998, 56: 119–148
John W M, Cristina S. Spinal-cord injury. Lancet, 2002, 359: 417–425
Rasouli A, Bhatia N, Dinh P, et al. Resection of glial scar following spinal cord injury. J Orthop Res, 2009, 27: 931–936
Fitch M T, Doller C, Combs C K. Cellular and molecular mechanisms of glial scarring and progressive cavitation: In vivo and in vitro analysis of inflammation-induced secondary injury after CNS trauma. J Neurol Sci, 1999, 19: 8182–8198
Wang S S, Hu Y Y, Luo Z J. Quantity analysis of local cavity and scar in rat spinal cord injury. Orthop J Chin, 2005, 6: 450–452
Weber T, Vroemen M, Behr V, et al. In vivo high-resolution MR imaging of neuropathologic changes in the injured rat spinal cord. Am J Neuroradiol, 2006, 27: 598–604
Kane R A. Intraoperative ultrasonography history, current state of the art, and future directions. J Ultrasound Med, 2004, 23: 1407–1420
Sosna J, Barth M M, Kruskal J B, et al. Intraoperative sonography for neurosurgery. J Ultrasound Med, 2005, 24: 1671–1682
Lammertse D, Dungan D, Dreisbach J, et al. Neuroimaging in traumatic spinal cord injury: An evidence-based review for clinical practice and research. J Spinal Cord Med, 2007, 30: 205–214
Wang M Y, Levi A D, Green B A. Intradural spinal arachnoid cysts in adults. Surg Neurol, 2003, 60: 49–55
Zeidman S M, Tomlin J M, Rodriguez-Cruz L. Intraoperative spinal sonography techniques in neurosurgery. Tech Neurosurg, 2003, 8: 27–39
Kolstad F, Rygh O, MSelbekk T, et al. Three-dimensional ultrasonography navigation in spinal cord tumor surgery. J Neurosurg Spine, 2006, 5: 264–270
Foster F S, Pavlin C J, Lockwood G R, et al. Principles and applications of ultrasound backscatter microscopy. IEEE Trans Ultrason Ferroelec Freq Contr, 1993, 40: 608–617
Razuvaev A, Lund K, Roy J, et al. Noninvasive real-time imaging of intima thickness after rat carotid artery balloon injury using ultrasound biomicroscopy. Atherosclerosis, 2008, 199: 310–316
Sherar M D, Noss M B, Foster E S. Ultrasound backscatter microscopy images the internal structure of living tumour spheroids. Nature, 1987, 330: 493–495
Pavlin C J, Harasiewicz K, Sherar M D, et al. Clinical use of ultrasound biomicroscopy. Ophthalmology, 1991, 98: 287–295
Goessling W, North T E, Zon L I. Ultrasound biomicroscopy permits in vivo characterization of zebrafish liver tumors. Nat Methods, 2007, 4: 551–553
Li X G, Yang Z Y, Yang Y. Studies on repairing of hemisected thoracic spinal cord of adult rats by using a chitosan tube filled with alginate fibers. Prog Nat Sci, 2006, 16: 1051–1055
Quencer R M, Morse B M, Green B A, et al. Intraoperative spinal sonography: Adjunct to metrizamide CT in the assessment and surgical decompression of posttraumatic spinal cord cysts. Am J Roentgenol, 1984, 142: 593–601
Knake J E, Gabrielsen T O, Chandler W F, et al. Real-time sonography during spinal surgery. Radiology, 1984, 151: 461–465
Gooding G A, Berger M S, Linkowski G D, et al. Transducer frequency considerations in intraoperative US of the spine. Radiology, 1986, 160: 272–273
Author information
Authors and Affiliations
Corresponding authors
Additional information
This article is published with open access at Springerlink.com
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Niu, H., Li, L., Liao, K. et al. Preliminary animal studies on observation of injured spinal cord with intraoperative ultrasound backscatter microscopy. Chin. Sci. Bull. 57, 2280–2284 (2012). https://doi.org/10.1007/s11434-012-5219-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11434-012-5219-7