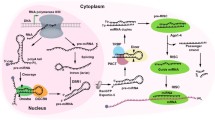
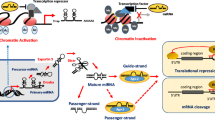
Abstract
MicroRNAs (miRNAs) constitute a family of small non-coding RNA molecules 22–25 nucleotides in length. miRNAs control the expression of target genes at the post-transcriptional level by inhibiting translation or by degrading target mRNA through binding to complementary sequences in the 3′-untranslated regions. Recent studies have demonstrated that miRNAs are intimately involved in processes leading to nasopharyngeal carcinoma, such as the Epstein-Barr virus-encoded latent membrane protein 1 activated signal transduction pathways, gene-regulatory networks, mitosis, tumor angiogenesis, invasion and migration. Exploring the relationship between miRNAs and the development of nasopharyngeal carcinoma, will further the understanding of this cancer and provide new avenues for diagnosis and treatment.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
George A C, Carlo M C. MicroRNA signatures in human cancers. Nat Rev Cancer, 2006, 6: 857–866
Chin D, Boyle G M, Porceddu S, et al. Head and neck cancer: Past, present and future. Expert Rev Anticancer Ther, 2006, 6: 1111–1118
Deyrup A T. Epstein-Barr virus-associated epithelial and mesenchymal neoplasms. Hum Pathol, 2008, 39: 473–483
Zhu J Y, Pfuhl T, Motsch N, et al. Identification of novel Epstein-Barr virus microRNA genes from nasopharyngeal carcinomas. J Virol, 2009, 83: 3333–3341
Georgia S. Emerging roles of microRNAs as molecular switches in the integrated circuit of the cancer cell. RNA, 2009, 15: 1443–1461
Filipowicz W, Bhattacharyya S N, Sonenberg N. Mechanisms of post-transcriptional regulation by microRNAs: Are the answers in sight? Nat Rev Genet, 2008, 9: 102–114
Israel A, Sharan R, Ruppin E, et al. Increased microRNA activity in human cancers. PLoS One, 2009, 4: e6045
Olson P, Lu J, Zhang H, et al. MicroRNA dynamics in the stages of tumorigenesis correlate with hallmark capabilities of cancer. Genes Dev, 2009, 23: 2152–2165
Zhang B, Pan X, Cobb G P, et al. MicroRNAs as oncogenes and tumor suppressors. Dev Biol, 2007, 302: 1–12
Wu M, Jolicoeur N, Li Z, et al. Genetic variations of microRNAs in human cancer and their effects on the expression of miRNAs. Carcinogenesis, 2008, 29: 1710–1716
Kumar M S, Lu J, Mercer K L, et al. Impaired microRNA processing enhances cellular transformation and tumorigenesis. Nature Genet, 2007, 39: 673–677
Liu C G, Calin G A, Volinia S, et al. MicroRNA expression profiling using microarrays. Nat Protoc, 2008, 3: 563–578
Hatfield S, Ruohola-Baker H. MicroRNA and stem cell function. Cell Tissue Res, 2008, 331: 57–66
Bushati N, Cohen S M. MicroRNA functions. Annu Rev Cell Dev Biol, 2007, 23: 175–205
Finoux A L, Chartrand P. Oncogenic and tumour suppressor microRNAs. Med Sci (Paris), 2008, 24: 1049–1054
Wang X, Tang S, Le S Y, et al. Aberrant expression of oncogenic and tumor-suppressive microRNAs in cervical cancer is required for cancer cell growth. PLoS One, 2008, 3: e2557
Aurora E K, Frank J S. Oncomirs-microRNAs with a role in cancer. Nat Rev Cancer, 2006, 6: 259–269
Rai D, Karanti S, Jung I, et al. Coordinated expression of microRNA-155 and predicted target genes in diffuse large B-cell lymphoma. Cancer Genet Cytogenet, 2008, 181: 8–15
Takakura S, Mitsutake N, Nakashima M, et al. Oncogenic role of miR-17-92 cluster in anaplastic thyroid cancer cells. Cancer Sci, 2008, 99: 1147–1154
Welch C, Chen Y, Stallings R L. MicroRNA-34a functions as a potential tumor suppressor by inducing apoptosis in neuroblastoma cells. Oncogene, 2007, 26: 5017–5022
Douglas R H. Metastamir: The field of metastasis-regulatory microRNA is spreading. Cancer Res, 2009, 69: 7495–7498
Cosmopoulos K, Pegtel M, Hawkins J, et al. Comprehensive profiling of Epstein-Barr virus microRNAs in nasopharyngeal carcinoma. J Virol, 2009, 83: 2357–2367
Griffiths-Jones S, Saini H K, van Dongen S, et al. miRBase: Tools for microRNA genomics. Nucleic Acids Res, 2008, 36: D154–D158
Gourzones C, Gelin A, Bombik I, et al. Extra-cellular release and blood diffusion of BART viral micro-RNAs produced by EBV-infected nasopharyngeal carcinoma cells. Virol J, 2010, 7: 271
Jing Y Z, Wang Y, Jia Y P, et al. Polymorphisms of Epstein-Barr virus BHRF1 gene, a homologue of bcl-2. Chin J Cancer, 2010, 29: 1000–1005
Edwards R H, Marquitz A R, Raab-Traub N. Epstein-Barr virus BART microRNAs are produced from a large intron prior to splicing. J Virol, 2008, 82: 9094–9106
Ghosh Z, Mallick B, Chakrabarti J. Cellular versus viral microRNAs in host-virus interaction. Nucleic Acids Res, 2009, 37: 1035–1048
Cullen B R. Viral and cellular messenger RNA targets of viral microRNAs. Nature, 2009, 457: 421–425
Xia T, O’Hara A, Araujo I, et al. EBV microRNAs in primary lymphomas and targeting of CXCL-11 by ebv-mir-BHRF1-3. Cancer Res, 2008, 68: 1436–1442
Choy E Y, Siu K L, Kok K H, et al. An Epstein-Barr virus-encoded microRNA targets PUMA to promote host cell survival. J Exp Med, 2008, 205: 2551–2560
Iizasa H, Wulff B E, Alla N R, et al. Editing of Epstein-Barr virus-encoded BART6 microRNAs controls their dicer targeting and consequently affects viral latency. J Biol Chem, 2010, 285: 33358–33370
Chang L K, Chuang J Y, Nakao M, et al. MCAF1 and synergistic activation of the transcription of Epstein-Barr virus lytic genes by Rta and Zta. Nucleic Acids Res, 2010, 38: 4687–4700
Hariwiyanto B, Sastrowiyoto S, Mubarika S, et al. LMP1 and LMP2 may be prognostic factors for outcome of therapy in nasopharyngeal cancers in Indonesia. Asian Pac J Cancer Prev, 2010, 11: 763–766
Natalie M, Thorsten P, Jan M, et al. Epstein-Barr virus-encoded latent membrane protein 1 (LMP1) induces the expression of the cellular microRNA miR-146a. RNA Biol, 2007, 4: 131–137
Middeldorp J M, Pegtel D M. Multiple roles of LMP1 in Epstein-Barr virus induced immune escape. Semin Cancer Biol, 2008, 18: 388–396
Graziana G, Annalisa R, Daniela R, et al. Epstein-Barr virus latent membrane protein 1 trans-activates miR-155 transcription through the NF-κB pathway. Nucleic Acids Res, 2008, 36: 6608–6619
Lo A K, To K F, Lo K W, et al. Modulation of LMP1 protein expression by EBV-encoded microRNAs. Proc Natl Acad Sci USA, 2007, 104: 16164–16169
Zheng H, Li L L, Hu D S, et al. Role of Epstein-Barr virus encoded latent membrane protein 1 in the carcinogenesis of nasopharyngeal carcinoma. Cell Mol Immunol, 2007, 4: 185–196
Liu Q, Fu H, Sun F, et al. miR-16 family induces cell cycle arrest by regulating multiple cell cycle genes. Nucleic Acids Res, 2008, 36: 5391–5404
Chen H C, Chen G H, Chen Y H, et al. MicroRNA deregulation and pathway alterations in nasopharyngeal carcinoma. Br J Cancer, 2009, 100: 1002–1011
Li Y H, Hu C F, Shao Q, et al. Elevated expressions of survivin and VEGF protein are strong independent predictors of survival in advanced nasopharyngeal carcinoma. J Transl Med, 2008, 6: 1
Pan J, Kong L, Lin S, et al. The clinical significance of coexpression of cyclooxygenases-2, vascular endothelial growth factors, and epidermal growth factor receptor in nasopharyngeal carcinoma. Laryngoscope, 2008, 118: 1970–1975
Zeng Z Y, Zhou Y H, Zhang W L, et al. Gene expression profiling of nasopharyngeal carcinoma reveals the abnormally regulated Wnt signaling pathway. Hum Pathol, 2007, 38: 120–133
Zhang L M, Deng T, Li X Y, et al. MicroRNA-141 is involved in a nasopharyngeal carcinoma-related genes network. Carcinogenesis, 2010, 31: 559–566
Zhou H D, Li X L, Li G Y, et al. Effect of SPLUNC1 protein on the Pseudomonas aeruginosa and Epstein-Barr virus. Mol Cell Biochem, 2008, 309: 191–197
Nakada C, Matsuura K, Tsukamoto Y, et al. Genome-wide microRNA expression profiling in renal cell carcinoma: Significant down-regulation of miR-141 and miR-200c. J Pathol, 2008, 216: 418–427
Xiao B, Fan S, Zeng Z, et al. Purification of novel UBAP1 protein and its decreased expression on nasopharyngeal carcinoma tissue microarray. Protein Expr Purif, 2006, 47: 60–67
Chalhoub N, Baker S J. PTEN and the PI3-kinase pathway in cancer. Annu Rev Pathol, 2009, 4: 127–150
Shen G P, Pan Q H, Hong M H, et al. Human genetic variants of homologous recombination repair genes first found to be associated with Epstein-Barr virus antibody titers in healthy cantonese. Int J Cancer, 2010, doi: 10.1002/ijc.25759
Ran Y, Wu S, You Y. Demethylation of E-cadherin gene in nasopharyngeal carcinoma could serve as a potential therapeutic strategy. J Biochem, 2010, doi: 10.1093/jb/mvq128
Guo Y, Chen J X, Yang S, et al. Selection of reliable reference genes for gene expression study in nasopharyngeal carcinoma. Acta Pharmacol Sin, 2010, 31: 1487–1494
Xu Y F, Liu W L, Dong J Q, et al. Sequencing of DC-SIGN promoter indicates an association between promoter variation and risk of nasopharyngeal carcinoma in cantonese. BMC Med Genet, 2010, doi: 10.1186/1471-2350-11-161
Wang S, Xiao X, Zhou X, et al. TFPI-2 is a putative tumor suppressor gene frequently inactivated by promoter hypermethylation in nasopharyngeal carcinoma. BMC Cancer, 2010, doi: 10.1186/1471-2407-10-617
Strebhardt K, Ullrich A. Targeting Polo-like kinase 1 for cancer therapy. Nat Rev Cancer, 2006, 6: 321–330
Wei S, Nehad M A, Carlo B, et al. Significance of Plk1 regulation by miR-100 in human nasopharyngeal cancer. Int J Cancer, 2010, 126: 2036–2048
Liu X, Lei M, Erikson R L. Normal cells, but not cancer cells, survive severe Plk1 depletion. Mol Cell Biol, 2006, 26: 2093–2108
Jang Y J, Ji J H, Choi Y C, et al. Regulation of Polo-like kinase 1 by DNA damage in mitosis: Inhibition of mitotic PLK-1 by protein phosphatase 2A. J Biol Chem, 2007, 282: 2473–2482
Sun F, Mikuni S, Kinjo M. Monitoring the caspase cascade in single apoptotic cells using a three-color fluorescent protein substrate. Biochem Biophys Res Commun, 2010, doi: 10.1016/j.bbrc.2010.12.047
Syljuasen R G, Jensen S, Bartek J, et al. Adaptation to the ionizing radiation-induced G2 checkpoint occurs in human cells and depends on checkpoint kinase 1 and Polo-like kinase 1 kinases. Cancer Res, 2006, 66: 10253–10257
Spankuch B, Kurunci-Csacsko E, Kaufmann M, et al. Rational combinations of siRNAs targeting Plk1 with breast cancer drugs. Oncogene, 2007, 26: 5793–5807
Su S F, Han F, Zhao C, et al. Long-term outcomes of early-stage nasopharyngeal carcinoma patients treated with intensity-modulated radiotherapy alone. Int J Radiat Oncol Biol Phys, 2010, doi: 10.1016/j.ijrobp.2010.09.011
Zhao L, Wan Q, Zhou Y, et al. The role of replanning in fractionated intensity modulated radiotherapy for nasopharyngeal carcinoma. Radiother Oncol, 2010, doi: 10.1016/j.radonc.2010.10.009
Shai E, Varon D. Development, cell differentiation, angiogenesis—Microparticles and their roles in angiogenesis. Arterioscler Thromb Vasc Biol, 2011, 31: 10–14
Lv X, Xiang Y Q, Cao S M, et al. Prospective validation of the prognostic value of elevated serum vascular endothelial growth factor in patients with nasopharyngeal carcinoma: More distant metastases and shorter overall survival after treatment. Head Neck, 2010, doi: 10.1002/hed.21541
Hua Z, Lv Q, Ye W B, et al. MiRNA-directed regulation of VEGF and other angiogenic factors under hypoxia. PLoS One, 2006, 1: e116
Olsson A K, Dimberg A, Kreuger J, et al. VEGF receptor signalling—in control of vascular function. Nat Rev Mol Cell Biol, 2006, 7: 359–371
Li G, Wu Z R, Peng Y, et al. MicroRNA-10b induced by Epstein-Barr virus-encoded latent membrane protein-1 promotes the metastasis of human nasopharyngeal carcinoma cells. Cancer Lett, 2010, 299: 29–36
Chew M M, Gan S Y, Khoo A S, et al. Interleukins, laminin and Epstein-Barr virus latent membrane protein 1 (EBV LMP1) promote metastatic phenotype in nasopharyngeal carcinoma. BMC Cancer, 2010, 10: 574
Kim T J, Lee Y S, Kang J H, et al. Prognostic significance of expression of vegf and cox-2 in nasopharyngeal carcinoma and its association with expression of C-erbB2 and EGFR. J Surg Oncol, 2010, doi: 10.1002/jso.21767
Sikumar S, Johan A B, Chen I H, et al. MicroRNA 29c is down-regulated in nasopharyngeal carcinomas, up-regulating mRNAs encoding extracelluar matrix proteins. Proc Natl Acad Sci USA, 2008, 105: 5874–5878
Xia H, Ng S S, Jiang S, et al. MiR-200a-mediated downregulation of ZEB2 and CTNNB1 differentially inhibits nasopharyngeal carcinoma cell growth, migration and invasion. Biochem Biophys Res Commun, 2010, 391: 535–541
Chen L C, Chen C C, Liang Y, et al. A novel role for TNFAIP2: Its correlation with invasion and metastasis in nasopharyngeal carcinoma. Mod Pathol, 2010, doi: 10.1038/modpathol.2010.193
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at Springerlink.com
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Zhao, L., Chen, X. & Cao, Y. miRNA and nasopharyngeal carcinoma. Chin. Sci. Bull. 56, 722–728 (2011). https://doi.org/10.1007/s11434-010-4330-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11434-010-4330-x