Abstract
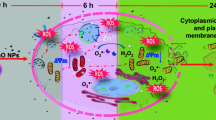
Owing to the rapidly increasing output of nano-scale titanium dioxide (TiO2) particles, their potential risk for central nerve system (CNS) has elicited much concern recently. Microglia is the resident macrophage in CNS and essential for the homeostasis of the CNS microenvironment. They are supposed to response to nanoparticles depositing in the brain tissues. Therefore, we investigated the cytotoxic effects of TiO2 NPs on microglia N9 cells in vitro. Results of propidium iodide/fluorescein diacetate (PI/FDA) double staining and MTT test clearly showed that TiO2 NPs more efficiently affected the viability of microglia N9 cells. Further Hoechst 33258 staining and flow cytometric analysis proved that nano-scale but not normal scale TiO2 induced apoptosis in vitro. These data suggest that TiO2 NPs can elicit apoptosis of N9 cells in vitro and thus present a potential risk for CNS.
Similar content being viewed by others
References
Park E J, Yi J, Chung K H, et al. Oxidative stress and apoptosis induced by titanium dioxide nanoparticles in cultured BEAS-2B cells. Toxicol Lett, 2008, 180: 222–229
Rahman Q, Lohani M, Dopp E, et al. Evidence that ultrafine titanium dioxide induces micronuclei and apoptosis in Syrian hamster embryo fibroblasts. Environ Health Perspect, 2002, 110: 797–800
Yu Y, Ren W, Ren B. Nanosize titanium dioxide cause neuronal apoptosis: A potential linkage between nanoparticle exposure and neural disorder. Neurol Res, 2008, doi: 10.1179/174313208X 305391
Bermudez E, Mangum J B, Wong B A, et al. Pulmonary responses of mice, rats, and hamsters to subchronic inhalation of ultrafine titanium dioxide particles. Toxicol Sci, 2004, 77: 347–357
Wang J, Liu Y, Jiao F, et al. Time-dependent translocation and potential impairment on central nervous system by intranasally instilled TiO(2) nanoparticles. Toxicology, 2008, 254: 82–90
Sharma S H. Nanoneuroscience: Emerging concepts onnanoneuro toxicity and nanoneuroprotection. Nanomed, 2007, 2: 753–758
Elder A, Gelein R, Silva V, et al. Translocation of inhaled ultrafine manganese oxide particles to the central nervous system. Environ Health Perspect, 2006, 114: 1172–1178
Wang J, Chen C, Liu Y, et al. Potential neurological lesion after nasal instillation of TiO(2) nanoparticles in the anatase and rutile crystal phases. Toxicol Lett, 2008, 183: 72–80
Kim Y S, Joh T H. Microglia, major player in the brain inflammation: Their roles in the pathogenesis of Parkinson’s disease. Exp Mol Med, 2006, 38: 333–347
Sharma S H, Sharma A. Nanoparticles aggravate heat stress induced cognitive deficits, blood-brain barrier disruption, edema formation and brain pathology. Prog Brain Res, 2007, 162: 245–273
Wang J J, Sanderson B J, Wang H. Cyto- and genotoxicity of ultrafine TiO2 particles in cultured human lymphoblastoid cells. Mutat Res, 2007, 628: 99–106
Kreuter J. Nanoparticles—a historical perspective. Int J Pharm, 2007, 331: 1–10
Schroder U, Sabel B A. Nanoparticles, a drug carrier system to pass the blood-brain barrier, permit central analgesic effects of i.v. dalargin injections. Brain Res, 1996, 710: 121–124
Olivier J C. Drug transport to brain with targeted nanoparticles. NeuroRx, 2005, 2: 108–119
Federici G, Shaw B J, Handy R D. Toxicity of titanium dioxide nanoparticles to rainbow trout (Oncorhynchus mykiss): gill injury, oxidative stress, and other physiological effects. Aquat Toxicol, 2007, 84: 415–430
Gremo F, Sogos V, Ennas M G, et al. Features and functions of human microglia cells. Adv Exp Med Biol, 1997, 429: 79–97
Vamanu C I, Cimpan M R, Hol P I, et al. Induction of cell death by TiO(2) nanoparticles: Studies on a human monoblastoid cell line. Toxicol In Vitro, 2008, 22: 1689–1696
Ayoub A E, Salm A K. Increased morphological diversity of microglia in the activated hypothalamic supraoptic nucleus. J Neurosci, 2003, 23: 7759–7766
Armour A D, Powell H M, Boyce S T. Fluorescein diacetate for determination of cell viability in tissue-engineered skin. Tissue Eng Part C Methods, 2008, 14: 89–96
Gonzalez-Scarano F, Baltuch G. Microglia as mediators of inflammatory and degenerative diseases. Annu Rev Neurosci, 1999, 22: 219–240
Hickman S E, Allison E K, El Khoury J. Microglial dysfunction and defective beta-amyloid clearance pathways in aging Alzheimer’s disease mice. J Neurosci, 2008, 28: 8354–8360
Sasaki A, Kawarabayashi T, Murakami T, et al. Microglial activation in brain lesions with tau deposits: Comparison of human tauopathies and tau transgenic mice TgTauP301L. Brain Res, 2008, 1214: 159–168
Lomer M C, Thompson R P, Powell J J. Fine and ultrafine particles of the diet: Influence on the mucosal immune response and association with Crohn’s disease. Proc Nutr Soc, 2002, 61: 123–130
Gelis C, Girard S, Mavon A, et al. Assessment of the skin photoprotective capacities of an organo-mineral broad-spectrum sunblock on two ex vivo skin models. Photodermatol Photoimmunol Photomed, 2003, 19: 242–253
Wang B, Feng W, Zhu M, et al. Neurotoxicity of low-dose repeatedly intranasal instillation of nano- and submicron-sized ferric oxide particles in mice. J Nanoparticle Res, 2008, 11: 41–53
Wang M L, Tuli R, Manner P A, et al. Direct and indirect induction of apoptosis in human mesenchymal stem cells in response to titanium particles. J Orthop Res, 2003, 21: 697–707
Osano E, Kishi J, Takahashi Y. Phagocytosis of titanium particles and necrosis in TNF-alpha-resistant mouse sarcoma L929 cells. Toxicol In Vitro, 2003, 17: 41–47
Darzynkiewicz Z, Juan G, Li X, et al. Cytometry in cell necro-biology: analysis of apoptosis and accidental cell death (necrosis). Cytometry, 1997, 27: 1–20
Cimpan M R, Cressey L I, Skaug N, et al. Patterns of cell death induced by eluates from denture base acrylic resins in U-937 human monoblastoid cells. Eur J Oral Sci, 2000, 108: 59–69
Wang J, Zhou G, Chen C, et al. Acute toxicity and biodistribution of different sized titanium dioxide particles in mice after oral administration, Toxicol Lett, 2007, 168: 176–185
Chen L, Yokel R A, Hennig B, et al. Manufactured aluminum oxide nanoparticles decrease expression of tight junction proteins in brain vasculature. J Neuroimmune Pharmacol, 2008, 3: 286–295
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by the German Academic Exchange Service (DAAD) and Chinese Scholarship Council (CSC)
About this article
Cite this article
Li, X., Xu, S., Zhang, Z. et al. Apoptosis induced by titanium dioxide nanoparticles in cultured murine microglia N9 cells. Chin. Sci. Bull. 54, 3830–3836 (2009). https://doi.org/10.1007/s11434-009-0548-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11434-009-0548-x