Abstract
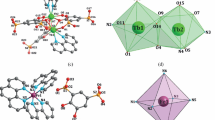
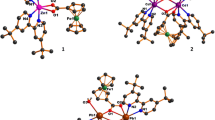
Three new mixed-ligand Fe(II/III) complexes [Fe2(μ2-btec)(μ2-H2btec)(phen)2(H2O)2] n (1), [Fe2(btec) (phen)2(H2O)4] (2), and “[Fe(o-pha)(phen)(H2O)]·H2O” n (3) (phen=1,10-phenanthroline, o-H2pha=o-phthalic acid, H4btec=1,2,4,5-benzenetetracarboxylic acid) have been hydrothermally synthesized and detected by single crystal X-ray diffraction, showing that complexes (1) and (2) are both bridged by the betc4− ligands to form 1D chain and dinuclear structure and complex (3) is bridged by the o-pha groups to form 1D chain structure. The coordinated modes of the carboxyl groups adopt μ2-η1η1ηη1η1 and μ2-η2η1 respectively in complexes (2) and (3). The betc groups in complex (1) show two different coordinated modes: μ2-η1η1η1η1 and μ2-η1η1. In addition, the hydrogen bonds and π...π type interactions make the complex molecule further connect to three-dimensional and two-dimensional networks respectively. These complexes are detected by IR, UV-Vis-NIR and surface photovoltage spectrum (SPS). The SPS of complexes (1)–(3) indicate that there are positive SPV responses in the range of 300–600 nm and show p-type semiconductor characteristic. Because the structure, the valence and the coordinative environment of the Fe ions are all different in the three complexes, the intensity, position and the number of the response bands are different obviously. The results of SPS are corresponding with UV-Vis-NIR spectra.
Similar content being viewed by others
References
Costas M, Mehn M P, Jensen M P, et al. Dioxygen activation at mononuclear nonheme iron active sites: enzymes, models, and in-termediates. Chem Rev, 2004, 104: 939–986
Xu D, Watt G D, Harb J N, et al. Electrical conductivity of ferritin pro-teins by conductive AFM. Nano Lett, 2005, 5: 571–577
Zhang J H, Cui C Y, Zhou X F. Study on elastic modulus of individual ferritin. Chinese Science Bulletin, 2009, 54: 723–726
Vladimir B R, Bruno T, Georg S-F, et al. Synthesis, molecular structure, and catalytic Potential of the tetrairon complex [Fe4(N3O2-L)4 (μ-O)2]4+(L=1-Carboxymethyl-4,7-dimethyl-1,4,7-triazacylononane). Inorg Chem, 2007, 46: 3166–3175
Matsumoto K, Ozawa T, Jitsukawa K, et al. Synthesis, solution behavior, thermal stability, and biological activity of an Fe(III) complex of an artificial siderophore with intramolecular hydrogen bonding networks. Inorg Chem, 2004, 43: 8538–8546
Xu H B, Wang Z M, Liu T, et al. Synthesis, structure, and magnetic properties of (A)[FeIII(oxalate)Cl2] (A= alkyl ammonium cations) with anionic 1D [FeIII(oxalate)Cl2]-chains. Inorg Chem, 2007, 46: 3089–3096
Stefan R I, Bairu S G. Monocrystalline diamond paste-based electrodes and their applications for the determination of Fe(II) in vitamins. Anal Chem, 2003, 75: 5394–5398
Singh U P, Aggarwal V, Sharma A K. Mononuclear cobalt(II) carboxylate complexes: Synthesis, molecular structure and selective oxygenation study. Inorg Chim Acta, 2007, 360: 3226–3232
Song J F, Zhou R S, Xu X Y, et al. Syntheses, crystal structures and properties of two new coordination complexes containing 1,2,4, 5-benzenetetracarboxylate and imidazole: [Ni2(Him)12·(bta)·8H2O] and [Cd2-(Him)4(μ4-bta)]. J Mol Struct, 2008, 874: 34–40
Majumder A, Gramlich V, Rosair G M, et al. Five new cobalt(II) and copper(II)-1,2,4,5-benzenetetracarboxylate supramolecular Architectures: Syntheses, Structures, and magnetic properties. Cryst Growth Des, 2006, 6: 2355–2368
Fabelo O, Pasan J, Canadillas-Delgado L, et al. Crystal structure and magnetic properties of two isomeric three-dimensional pyromellitate-containing cobalt (II) complexes. Inorg Chem, 2008, 47: 8053–8061
Zhou X X, Liu M S, Lin X M, Construction of three low dimensional Zn(II) complexes based on different organic-carboxylic acids. Inorg Chim Acta, 2009, 362: 1441–1447
Shi X, Zhu G, Wang X, et al. From a 1-D chain, 2-D layered network to a 3-D supramolecular framework constructed from a metal-Organic coordination compound. Cryst Growth Des, 2005, 5: 207–213
Xie Y B, Li J R, Zhang C, et al. Syntheses and crystal structures of manganes e(II), cadmium (II), cobalt (II), and zinc (II) complexes with 4-Pyridyl dithioether ligands. Cryst Growth Des, 2005, 5: 1743–1749
Li G, Hou H W, Niu Y Y, et al. Synthesis, crystal structures and mag-netic properties of helical double-stranded three-dimensional network [Co(NO3)2(bpt)2·2CH3OH]n and two-dimensional network [Mn(NCS)2(bpt)3·H2O]n (bpt=N,N′-bis(3-pyridylmethyl) thiourea). Inorg Chim Acta, 2002, 332: 216–222
Lin Y H, Wang D J, Zhao Q D, et al. A study of quantum confinement properties of photogenerated charges in ZnO nanoparticles by surface photovoltage spectroscopy. J Phys Chem B, 2004, 108: 3202–3206
Qi M H, Liu G F. Surface photovoltage, luminescence, and cyclic voltammetry on the first series of lanthanide (III) monobenzoporphyrin compound liquid crystals and relative transition metal benzoporphyrin compound liquid crystals. J Phys Chem B, 2003, 107: 7640–7646
Sun L P, Niu S Y, Jin J, et al. Crystal structure and surface photovoltage properties of Mn(II) coordination supramolecules. Eur J Inorg Chem, 2007, 3845–3852
Sun L P, Niu S Y, Jin J, et al. Crystal structure and surface photovoltage of a series of Ni (II) coordination supramolecular polymer. Inorg Chem Commun, 2006, 9: 679–682
Zhang L, Niu S Y, Jin J, et al. Crystal structure and surface photoelectric property of Mn (II) coordination supramolecules. Chinese Sci Bull, 2007, 52: 2832–2839
Sun L P, Niu S Y, Jin J, et al. Synthesis, structure and surface photovoltage of a series of Ni (II) coordination polymers. Eur J Inorg Chem, 2006, 5130–5137
Zhang L, Niu S Y, Jin J, et al. Synthesis, crystal structure and surface photovoltage of a series of Mn (II) coordination supramolecules. Acta Chim Sini, 2007, 65: 1032–1038
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was supported by the National Natural Science Foundation of China (Grant No. 20571037) and Liaoning Province Foundation (Grant No. 2007T092).
About this article
Cite this article
Shi, Z., Jin, J., Zhao, L. et al. Crystal structure and photoelectricity property of Fe(II/III) coordination supermolecules. Chin. Sci. Bull. 55, 124–130 (2010). https://doi.org/10.1007/s11434-009-0483-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11434-009-0483-x