Abstract
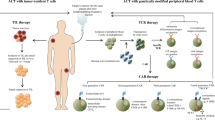
Impaired tumor-specific effector T cells contribute to tumor progression and unfavorable clinical outcomes. As a compensatory T cell-dependent cancer immunoediting strategy, adoptive T cell therapy (ACT) has achieved encouraging therapeutic results, and this strategy is now on the center stage of cancer treatment and research. ACT involves the ex vivo stimulation and expansion of tumor-infiltrating lymphocytes (TILs) with inherent tumor reactivity or T cells that have been genetically modified to express the cognate chimeric antigen receptor or T cell receptor (CAR/TCR), followed by the passive transfer of these cells into a lymphodepleted host. Primed T cells must provide highly efficient and long-lasting immune defense against transformed cells during ACT. Anin-depth understanding of the basic mechanisms of these living drugs can help us improve upon current strategies and design better next-generation T cell-based immunotherapies. From this perspective, we provide an overview of current developments in different ACT strategies, with a focus on frontier clinical trials that offer a proof of principle. Meanwhile, insights into the determinants of ACT are discussed, which will lead to more rational, potent and widespread applications in the future.
Similar content being viewed by others
References
Aaltomaa, S., Lipponen, P., Eskelinen, M., Kosma, V.M., Marin, S., Alhava, E., and Syrjänen, K. (1992). Lymphocyte infiltrates as a prognostic variable in female breast cancer. Eur J Cancer 28, 859–864.
Ali, M., Foldvari, Z., Giannakopoulou, E., Böschen, M.L., Strønen, E., Yang, W., Toebes, M., Schubert, B., Kohlbacher, O., Schumacher, T.N., et al. (2019). Induction of neoantigen-reactive T cells from healthy donors. Nat Protoc 14, 1926–1943.
Arcangeli, S., Rotiroti, M.C., Bardelli, M., Simonelli, L., Magnani, C.F., Biondi, A., Biagi, E., Tettamanti, S., and Varani, L. (2017). Balance of anti-CD123 chimeric antigen receptor binding affinity and density for the targeting of acute myeloid leukemia. Mol Ther 25, 1933–1945.
Baker, P.E., Gillis, S., and Smith, K.A. (1979). Monoclonal cytolytic T-cell lines.. J Exp Med 149, 273–278.
Balakrishnan, A., Rajan, A., Salter, A.I., Kosasih, P.L., Wu, Q., Voutsinas, J., Jensen, M.C., Plückthun, A., and Riddell, S.R. (2019). Multispecific targeting with synthetic ankyrin repeat motif chimeric antigen receptors. Clin Cancer Res 25, 7506–7516.
Belessi, C., Stamatopoulos, K., Stavroyianni, N., Zoi, K., Papadaki, T., and Kosmas, C. (2001). Somatic hypermutation targeting to intrinsic hotspots of immunoglobulin genes in follicular lymphoma and multiple myeloma. Leukemia 15, 1772–1778.
Bielamowicz, K., Fousek, K., Byrd, T.T., Samaha, H., Mukherjee, M., Aware, N., Wu, M.F., Orange, J.S., Sumazin, P., Man, T.K., et al. (2018). Trivalent CAR T cells overcome interpatient antigenic variability in glioblastoma. Neuro-Oncology 20, 506–518.
Chandran, S.S., and Klebanoff, C.A. (2019). T cell receptor-based cancer immunotherapy: Emerging efficacy and pathways of resistance. Immunol Rev 290, 127–147.
Chow, V.A., Shadman, M., and Gopal, A.K. (2018). Translating anti-CD19 CAR T-cell therapy into clinical practice for relapsed/refractory diffuse large B-cell lymphoma. Blood 132, 777–781.
Couzin-Frankel, J. (2013). Cancer immunotherapy. Science 342, 1432–1433.
Csizmar, C.M., Petersburg, J.R., Perry, T.J., Rozumalski, L., Hackel, B.J., and Wagner, C.R. (2019). Multivalent ligand binding to cell membrane antigens: defining the interplay of affinity, valency, and expression density. J Am Chem Soc 141, 251–261.
D’Angelo, S.P., Melchiori, L., Merchant, M.S., Bernstein, D., Glod, J., Kaplan, R., Grupp, S., Tap, W.D., Chagin, K., Binder, G.K., et al. (2018). Antitumor activity associated with prolonged persistence of adoptively transferred NY-ESO-1c259 T cells in synovial sarcoma. Cancer Discov 8, 944–957.
Dafni, U., Michielin, O., Lluesma, S.M., Tsourti, Z., Polydoropoulou, V., Karlis, D., Besser, M.J., Haanen, J., Svane, I.M., Ohashi, P.S., et al. (2019). Efficacy of adoptive therapy with tumor-infiltrating lymphocytes and recombinant interleukin-2 in advanced cutaneous melanoma: a systematic review and meta-analysis. Ann Oncol 30, 1902–1913.
Davenport, A.J., Cross, R.S., Watson, K.A., Liao, Y., Shi, W., Prince, H.M., Beavis, P.A., Trapani, J.A., Kershaw, M.H., Ritchie, D.S., et al. (2018). Chimeric antigen receptor T cells form nonclassical and potent immune synapses driving rapid cytotoxicity. Proc Natl Acad Sci USA 115, E2068–E2076.
Deniger, D.C., Pasetto, A., Robbins, P.F., Gartner, J.J., Prickett, T.D., Paria, B.C., Malekzadeh, P., Jia, L., Yossef, R., Langhan, M.M., et al. (2018). T-cell responses to TP53 “hotspot” mutations and unique neoantigens expressed by human ovarian cancers. Clin Cancer Res 24, 5562–5573.
Dudley, M.E., Wunderlich, J.R., Robbins, P.F., Yang, J.C., Hwu, P., Schwartzentruber, D.J., Topalian, S.L., Sherry, R., Restifo, N.P., Hubicki, A.M., et al. (2002). Cancer regression and autoimmunity in patients after clonal repopulation with antitumor lymphocytes. Science 298, 850–854.
Dunn, G.P., Old, L.J., and Schreiber, R.D. (2004). The immunobiology of cancer immunosurveillance and immunoediting. Immunity 21, 137–148.
Eberlein, T.J., Rosenstein, M., and Rosenberg, S.A. (1982). Regression of a disseminated syngeneic solid tumor by systemic transfer of lymphoid cells expanded in interleukin 2. J Exp Med 156, 385–397.
Fang, L., Ly, D., Wang, S.S., Lee, J.B., Kang, H., Xu, H., Yao, J., Tsao, M. S., Liu, W., and Zhang, L. (2019). Targeting late-stage non-small cell lung cancer with a combination of DNT cellular therapy and PD-1 checkpoint blockade. J Exp Clin Cancer Res 38, 123.
Figueiredo, C.R., Kalirai, H., Sacco, J.J., Azevedo, R.A., Duckworth, A., Slupsky, J.R., Coulson, J.M., and Coupland, S.E. (2020). Loss of BAP1 expression is associated with an immunosuppressive microenvironment in uveal melanoma, with implications for immunotherapy development. J Pathol 250, 420–439.
Finney, H.M., Lawson, A.D., Bebbington, C.R., and Weir, A.N. (1998). Chimeric receptors providing both primary and costimulatory signaling in T cells from a single gene product. J Immunol 161, 2791–2797.
Fraietta, J.A., Lacey, S.F., Orlando, E.J., Pruteanu-Malinici, I., Gohil, M., Lundh, S., Boesteanu, A.C., Wang, Y., O’Connor, R.S., Hwang, W.T., et al. (2018). Determinants of response and resistance to CD19 chimeric antigen receptor (CAR) T cell therapy of chronic lymphocytic leukemia. Nat Med 24, 563–571.
Geukes Foppen, M.H., Donia, M., Svane, I.M., and Haanen, J.B.A.G. (2015). Tumor-infiltrating lymphocytes for the treatment of metastatic cancer. Mol Oncol 9, 1918–1935.
Goff, S.L., Dudley, M.E., Citrin, D.E., Somerville, R.P., Wunderlich, J.R., Danforth, D.N., Zlott, D.A., Yang, J.C., Sherry, R.M., Kammula, U.S., et al. (2016). Randomized, prospective evaluation comparing intensity of lymphodepletion before adoptive transfer of tumor-infiltrating lymphocytes for patients with metastatic melanoma. J Clin Oncol 34, 2389–2397.
Guo, Y., Feng, K., Tong, C., Jia, H., Liu, Y., Wang, Y., Ti, D., Yang, Q., Wu, Z., and Han, W. (2020). Efficiency and side effects of anti-CD38 CART cells in an adult patient with relapsed B-ALL after failure of bi-specific CD19/CD22 CAR T cell treatment. Cell Mol Immunol 17, 430–432.
Hamieh, M., Dobrin, A., Cabriolu, A., van der Stegen, S.J.C., Giavridis, T., Mansilla-Soto, J., Eyquem, J., Zhao, Z., Whitlock, B.M., Miele, M.M., et al. (2019). CAR T cell trogocytosis and cooperative killing regulate tumour antigen escape. Nature 568, 112–116.
Hirayama, A.V., Gauthier, J., Hay, K.A., Voutsinas, J.M., Wu, Q., Pender, B.S., Hawkins, R.M., Vakil, A., Steinmetz, R.N., Riddell, S.R., et al. (2019). High rate of durable complete remission in follicular lymphoma after CD19 CAR-T cell immunotherapy. Blood 134, 636–640.
Hladíková, K., Partlová, S., Koucký, V., Bouček, J., Fonteneau, J.F., Zábrodský, M., Tachezy, R., Grega, M., Špíšek, R., and Fialová, A. (2018). Dysfunction of HPV16-specific CD8+ T cells derived from oropharyngeal tumors is related to the expression of Tim-3 but not PD-1. Oral Oncol 82, 75–82.
Hombrink, P., Raz, Y., Kester, M.G.D., de Boer, R., Weißbrich, B., von dem Borne, P.A., Busch, D.H., Schumacher, T.N.M., Falkenburg, J.H.F., and Heemskerk, M.H.M. (2013). Mixed functional characteristics correlating with TCR-ligand koff-rate of MHC-tetramer reactive T cells within the naive T-cell repertoire. Eur J Immunol 43, 3038–3050.
Jahn, L., Hombrink, P., Hagedoorn, R.S., Kester, M.G.D., van der Steen, D. M., Rodriguez, T., Pentcheva-Hoang, T., de Ru, A.H., Schoonakker, M. P., Meeuwsen, M.H., et al. (2017). TCR-based therapy for multiple myeloma and other B-cell malignancies targeting intracellular transcription factor BOB1. Blood 129, 1284–1295.
Jia, H., Wang, Z., Wang, Y., Liu, Y., Dai, H., Tong, C., Guo, Y., Guo, B., Ti, D., Han, X., et al. (2019). Haploidentical CD19/CD22 bispecific CART cells induced MRD-negative remission in a patient with relapsed and refractory adult B-ALL after haploidentical hematopoietic stem cell transplantation. J Hematol Oncol 12, 57.
Johanns, T.M., Ward, J.P., Miller, C.A., Wilson, C., Kobayashi, D.K., Bender, D., Fu, Y., Alexandrov, A., Mardis, E.R., Artyomov, M.N., et al. (2016). Endogenous neoantigen-specific CD8 T cells identified in two glioblastoma models using a cancer immunogenomics approach. Cancer Immunol Res 4, 1007–1015.
Kacherovsky, N., Cardle, I.I., Cheng, E.L., Yu, J.L., Baldwin, M.L., Salipante, S.J., Jensen, M.C., and Pun, S.H. (2019). Traceless aptamer-mediated isolation of CD8+ T cells for chimeric antigen receptor T-cell therapy. Nat Biomed Eng 3, 783–795.
Kageyama, S., Ikeda, H., Miyahara, Y., Imai, N., Ishihara, M., Saito, K., Sugino, S., Ueda, S., Ishikawa, T., Kokura, S., et al. (2015). Adoptive transfer of MAGE-A4 T-cell receptor gene-transduced lymphocytes in patients with recurrent esophageal cancer. Clin Cancer Res 21, 2268–2277.
Kearl, T.J., Jing, W., Gershan, J.A., and Johnson, B.D. (2013). Programmed death receptor-1/programmed death receptor ligand-1 blockade after transient lymphodepletion to treat myeloma. J Immnol 190, 5620–5628.
Keibel, A., Singh, V., and Sharma, M.C. (2009). Inflammation, microenvironment, and the immune system in cancer progression. Curr Pharm Des 15, 1949–1955.
Krokhotin, A., Du, H., Hirabayashi, K., Popov, K., Kurokawa, T., Wan, X., Ferrone, S., Dotti, G., and Dokholyan, N.V. (2019). Computationally guided design of single-chain variable fragment improves specificity of chimeric antigen receptors. Mol Ther Oncolytics 15, 30–37.
Leko, V., McDuffie, L.A., Zheng, Z., Gartner, J.J., Prickett, T.D., Apolo, A. B., Agarwal, P.K., Rosenberg, S.A., and Lu, Y.C. (2019). Identification of neoantigen-reactive tumor-infiltrating lymphocytes in primary bladder cancer. J Immnol 202, 3458–3467.
Lian, X., Lin, Y.M., Kozono, S., Herbert, M.K., Li, X., Yuan, X., Guo, J., Guo, Y., Tang, M., Lin, J., et al. (2018). Pin1 inhibition exerts potent activity against acute myeloid leukemia through blocking multiple cancer-driving pathways. J Hematol Oncol 11, 73.
Lin, J.K., Muffly, L.S., Spinner, M.A., Barnes, J.I., Owens, D.K., and Goldhaber-Fiebert, J.D. (2019). Cost effectiveness of chimeric antigen receptor T-cell therapy in multiply relapsed or refractory adult large B-cell lymphoma. J Clin Oncol 37, 2105–2119.
Longo, D.L. (2010). New therapies for castration-resistant prostate cancer. N Engl J Med 363, 479–481.
Ma, T., Shi, J., and Liu, H. (2019). Chimeric antigen receptor T cell targeting B cell maturation antigen immunotherapy is promising for multiple myeloma. Ann Hematol 98, 813–822.
Macleod, B.L., Bedoui, S., Hor, J.L., Mueller, S.N., Russell, T.A., Hollett, N.A., Heath, W.R., Tscharke, D.C., Brooks, A.G., and Gebhardt, T. (2014). Distinct APC subtypes drive spatially segregated CD4+ and CD8+ T-cell effector activity during skin infection with HSV-1. PLoS Pathog 10, e1004303.
Mahvi, D.A., Meyers, J.V., Tatar, A.J., Contreras, A., Suresh, M., Leverson, G.E., Sen, S., and Cho, C.S. (2015). Ctla-4 blockade plus adoptive T-cell transfer promotes optimal melanoma immunity in mice. J Immunother 38, 54–61.
Majzner, R.G., and Mackall, C.L. (2018). Tumor antigen escape from CAR T-cell therapy. Cancer Discov 8, 1219–1226.
Marchingo, J.M., Kan, A., Sutherland, R.M., Duffy, K.R., Wellard, C.J., Belz, G.T., Lew, A.M., Dowling, M.R., Heinzel, S., and Hodgkin, P.D. (2014). Antigen affinity, costimulation, and cytokine inputs sum linearly to amplify T cell expansion. Science 346, 1123–1127.
Mason, S., and Warner, N.L. (1970). The immunoglobulin nature of the antigen recognition site on cells mediating transplantation immunity and delayed hypersentivity. J Immunol 104, 762–765.
Maude, S.L., Laetsch, T.W., Buechner, J., Rives, S., Boyer, M., Bittencourt, H., Bader, P., Verneris, M.R., Stefanski, H.E., Myers, G.D., et al. (2018). Tisagenlecleucel in children and young adults with B-cell lymphoblastic leukemia. N Engl J Med 378, 439–448.
Moritz, D., Wels, W., Mattern, J., and Groner, B. (1994). Cytotoxic T lymphocytes with a grafted recognition specificity for ERBB2-expressing tumor cells. Proc Natl Acad Sci USA 91, 4318–4322.
Nerreter, T., Letschert, S., Götz, R., Doose, S., Danhof, S., Einsele, H., Sauer, M., and Hudecek, M. (2019). Super-resolution microscopy reveals ultra-low CD19 expression on myeloma cells that triggers elimination by CD19 CAR-T. Nat Commun 10, 3137.
Nguyen, L.T., Saibil, S.D., Sotov, V., Le, M.X., Khoja, L., Ghazarian, D., Bonilla, L., Majeed, H., Hogg, D., Joshua, A.M., et al. (2019). Phase II clinical trial of adoptive cell therapy for patients with metastatic melanoma with autologous tumor-infiltrating lymphocytes and low-dose interleukin-2. Cancer Immunol Immunother 68, 773–785.
Paluskievicz, C.M., Cao, X., Abdi, R., Zheng, P., Liu, Y., and Bromberg, J. S. (2019). T regulatory cells and priming the suppressive tumor microenvironment. Front Immunol 10, 2453.
Piñeiro Fernández, J., Luddy, K.A., Harmon, C., and O’Farrelly, C. (2019). Hepatic tumor microenvironments and effects on NK cell phenotype and function. Int J Mol Sci 20, 4131.
Pont, M.J., Hill, T., Cole, G.O., Abbott, J.J., Kelliher, J., Salter, A.I., Hudecek, M., Comstock, M.L., Rajan, A., Patel, B.K.R., et al. (2019). γ-Secretase inhibition increases efficacy of BCMA-specific chimeric antigen receptor T cells in multiple myeloma. Blood 134, 1585–1597.
Ramakrishna, S., Highfill, S.L., Walsh, Z., Nguyen, S.M., Lei, H., Shern, J. F., Qin, H., Kraft, I.L., Stetler-Stevenson, M., Yuan, C.M., et al. (2019). Modulation of target antigen density improves CAR T-cell functionality and persistence. Clin Cancer Res 25, 5329–5341.
Raman, M.C.C., Rizkallah, P.J., Simmons, R., Donnellan, Z., Dukes, J., Bossi, G., Le Provost, G.S., Todorov, P., Baston, E., Hickman, E., et al. (2016). Direct molecular mimicry enables off-target cardiovascular toxicity by an enhanced affinity TCR designed for cancer immunotherapy. Sci Rep 6, 18851.
Riley, T.P., Keller, G.L.J., Smith, A.R., Davancaze, L.M., Arbuiso, A.G., Devlin, J.R., and Baker, B.M. (2019). Structure based prediction of neoantigen immunogenicity. Front Immunol 10, 2047.
Rosenberg, S.A., Packard, B.S., Aebersold, P.M., Solomon, D., Topalian, S. L., Toy, S.T., Simon, P., Lotze, M.T., Yang, J.C., Seipp, C.A., et al. (1988). Use of tumor-infiltrating lymphocytes and interleukin-2 in the immunotherapy of patients with metastatic melanoma. N Engl J Med 319, 1676–1680.
Schmidts, A., Ormhøj, M., Choi, B.D., Taylor, A.O., Bouffard, A.A., Scarfò, I., Larson, R.C., Frigault, M.J., Gallagher, K., Castano, A.P., et al. (2019). Rational design of a trimeric APRIL-based CAR-binding domain enables efficient targeting of multiple myeloma. Blood Adv 3, 3248–3260.
Schneider, D., Xiong, Y., Wu, D., Nölle, V., Schmitz, S., Haso, W., Kaiser, A., Dropulic, B., and Orentas, R.J. (2017). A tandem CD19/CD20 CAR lentiviral vector drives on-target and off-target antigen modulation in leukemia cell lines. J Immunother Cancer 5, 42.
Shah, W., Yan, X., Jing, L., Zhou, Y., Chen, H., and Wang, Y. (2011). A reversed CD4/CD8 ratio of tumor-infiltrating lymphocytes and a high percentage of CD4+FOXP3+ regulatory T cells are significantly associated with clinical outcome in squamous cell carcinoma of the cervix. Cell Mol Immunol 8, 59–66.
Sibener, L.V., Fernandes, R.A., Kolawole, E.M., Carbone, C.B., Liu, F., McAffee, D., Birnbaum, M.E., Yang, X., Su, L.F., Yu, W., et al. (2018). Isolation of a structural mechanism for uncoupling t cell receptor signaling from peptide-MHC binding. Cell 174, 672–687.e27.
Stadtmauer, E.A., Fraietta, J.A., Davis, M.M., Cohen, A.D., Weber, K.L., Lancaster, E., Mangan, P.A., Kulikovskaya, I., Gupta, M., Chen, F., et al. (2020). CRISPR-engineered T cells in patients with refractory cancer. Science 367, eaba7365.
Stinchcombe, J.C., Bossi, G., Booth, S., and Griffiths, G.M. (2001). The immunological synapse of CTL contains a secretory domain and membrane bridges. Immunity 15, 751–761.
Stock, S., Übelhart, R., Schubert, M.L., Fan, F., He, B., Hoffmann, J.M., Wang, L., Wang, S., Gong, W., Neuber, B., et al. (2019). Idelalisib for optimized CD19-specific chimeric antigen receptor T cells in chronic lymphocytic leukemia patients. Int J Cancer 145, 1312–1324.
Sukari, A., Abdallah, N., and Nagasaka, M. (2019). Unleash the power of the mighty T cells-basis of adoptive cellular therapy. Crit Rev Oncol/Hematol 136, 1–12.
Thanindratarn, P., Dean, D.C., Nelson, S.D., Hornicek, F.J., and Duan, Z. (2019). Chimeric antigen receptor T (CAR-T) cell immunotherapy for sarcomas: From mechanisms to potential clinical applications. Cancer Treat Rev 82, 101934.
Ti, D., Niu, Y., Wu, Z., Fu, X., and Han, W. (2018). Genetic engineering of T cells with chimeric antigen receptors for hematological malignancy immunotherapy. Sci China Life Sci 61, 1320–1332.
van Bruggen, J.A.C., Martens, A.W.J., Fraietta, J.A., Hofland, T., Tonino, S.H., Eldering, E., Levin, M.D., Siska, P.J., Endstra, S., Rathmell, J.C., et al. (2019). Chronic lymphocytic leukemia cells impair mitochondrial fitness in CD8+ T cells and impede CAR T-cell efficacy. Blood 134, 44–58.
van der Bruggen, P., Traversari, C., Chomez, P., Lurquin, C., De Plaen, E., Van den Eynde, B., Knuth, A., and Boon, T. (1991). A gene encoding an antigen recognized by cytolytic T lymphocytes on a human melanoma. Science 254, 1643–1647.
Walseng, E., Köksal, H., Sektioglu, I.M., Fåne, A., Skorstad, G., Kvalheim, G., Gaudernack, G., Inderberg, E.M., and Wälchli, S. (2017). A TCR-based chimeric antigen receptor. Sci Rep 7, 10713.
Wang, L., Yao, R., Zhang, L., Fan, C., Ma, L., and Liu, J. (2019a). Chimeric antigen receptor T cell therapy and other therapeutics for malignancies: Combination and opportunity. Int Immunopharmacol 70, 498–503.
Wang, N., Hu, X., Cao, W., Li, C., Xiao, Y., Cao, Y., Gu, C., Zhang, S., Chen, L., Cheng, J., et al. (2020). Efficacy and safety of CAR19/22 T-cell cocktail therapy in patients with refractory/relapsed B-cell malignancies. Blood 135, 17–27.
Wang, S.E., Yu, Y., Criswell, T.L., Debusk, L.M., Lin, P.C., Zent, R., Johnson, D.H., Ren, X., and Arteaga, C.L. (2010). Oncogenic mutations regulate tumor microenvironment through induction of growth factors and angiogenic mediators. Oncogene 29, 3335–3348.
Wang, Y., Xu, L., Yan, C., and Huang, X. (2019b). Modification of donor lymphocyte infusion: how to improve the outcome? Sci China Life Sci 62, 1253–1256.
Wedagedera, J.R., and Burroughs, N.J. (2006). T-cell activation: A queuing theory analysis at low agonist density. Biophys J 91, 1604–1618.
Wrzesinski, C., Paulos, C.M., Kaiser, A., Muranski, P., Palmer, D.C., Gattinoni, L., Yu, Z., Rosenberg, S.A., and Restifo, N.P. (2010). Increased intensity lymphodepletion enhances tumor treatment efficacy of adoptively transferred tumor-specific T cells. J Immunother 33, 1–7.
Yewdell, J.W., Dersh, D., and Fåhraeus, R. (2019). Peptide channeling: the key to MHC class I immunosurveillance? Trends Cell Biol 29, 929–939.
Zhang, L., Conejo-Garcia, J.R., Katsaros, D., Gimotty, P.A., Massobrio, M., Regnani, G., Makrigiannakis, A., Gray, H., Schlienger, K., Liebman, M. N., et al. (2003). Intratumoral T cells, recurrence, and survival in epithelial ovarian cancer. N Engl J Med 348, 203–213.
Zhao, Q., Ahmed, M., Tassev, D.V., Hasan, A., Kuo, T.Y., Guo, H.F., O’Reilly, R.J., and Cheung, N.K.V. (2015). Affinity maturation of T-cell receptor-like antibodies for Wilms tumor 1 peptide greatly enhances therapeutic potential. Leukemia 29, 2238–2247.
Zhao, Y., Zheng, Z., Khong, H.T., Rosenberg, S.A., and Morgan, R.A. (2006). Transduction of an HLA-DP4-restricted NY-ESO-1-specific TCR into primary human CD4+ lymphocytes. J Immunother 29, 398–406.
Zhou, G., Noordam, L., Sprengers, D., Doukas, M., Boor, P.P.C., van Beek, A.A., Erkens, R., Mancham, S., Grünhagen, D., Menon, A.G., et al. (2018). Blockade of LAG3 enhances responses of tumor-infiltrating T cells in mismatch repair-proficient liver metastases of colorectal cancer. Oncoimmunology 7, e1448332.
Zikich, D., Schachter, J., and Besser, M.J. (2016). Predictors of tumor-infiltrating lymphocyte efficacy in melanoma. Immunotherapy 8, 35–43.
Zinkernagel, R.M., and Doherty, P.C. (1976). Virus-immune cytotoxic T cells are sentized to by virus specifically altered structures coded for in H-2K or H-2D: a biological role for major histocompatibility antigens. Adv Exp Med Biol 66, 387–389.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (81830002, 31870873, and 31991171 to WeidongHan; 31971378 to Dongdong Ti; 81672797 to Xiaolei Li).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Compliance and ethics
The author(s) declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Ti, D., Bai, M., Li, X. et al. Adaptive T cell immunotherapy in cancer. Sci. China Life Sci. 64, 363–371 (2021). https://doi.org/10.1007/s11427-020-1713-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-020-1713-9