Abstract
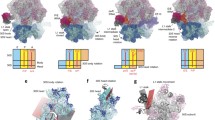
Elucidating protein translational regulation is crucial for understanding cellular function and drug development. A key molecule in protein translation is ribosome, which is a super-molecular complex extensively studied for more than a half century. The structure and dynamics of ribosome complexes were resolved recently thanks to the development of X-ray crystallography, Cryo-EM, and single molecule biophysics. Current studies of the ribosome have shown multiple functional states, each with a unique conformation. In this study, we analyzed the RNA-protein distances of ribosome (2.5 MDa) complexes and compared these changes among different ribosome complexes. We found that the RNA-protein distance is significantly correlated with the ribosomal functional state. Thus, the analysis of RNA-protein binding distances at important functional sites can distinguish ribosomal functional states and help understand ribosome functions. In particular, the mechanism of translational attenuation by nascent peptides and antibiotics was revealed by the conformational changes of local functional sites.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Nakatogawa H, Ito K. The ribosomal exit tunnel functions as a discriminating gate. Cell, 2002, 108: 629–636
Moore PB. The ribosome returned. J Biol, 2009, 8: 8
Ben-Shem A, Jenner L, Yusupova G, Yusupov M. Crystal structure of the eukaryotic ribosome. Science, 2010, 330: 1203–1209
Kurkcuoglu O, Doruker P, Sen TZ, Kloczkowski A, Jernigan RL. The ribosome structure controls and directs mRNA entry, translocation and exit dynamics. Phys Biol, 2008, 5: 046005
Cornish PV, Ermolenko DN, Noller HF, Ha T. Spontaneous intersubunit rotation in single ribosomes. Mol Cell, 2008, 30: 578–588
Julian P, Konevega AL, Scheres SH, Lazaro M, Gil D, Wintermeyer W, Rodnina MV, Valle M. Structure of ratcheted ribosomes with tRNAs in hybrid states. Proc Natl Acad Sci USA, 2008, 105: 16924–16927
Tenson T, Ehrenberg M. Regulatory nascent peptides in the ribosomal tunnel. Cell, 2002, 108: 591–594
Valle M, Zavialov A, Sengupta J, Rawat U, Ehrenberg M, Frank J. Locking and unlocking of ribosomal motions. Cell, 2003, 114: 123–134
Fredrick K, Ibba M. How the sequence of a gene can tune its translation. Cell, 2010, 141: 227–229
Weis F, Bron P, Rolland JP, Thomas D, Felden B, Gillet R. Accommodation of tmRNA-SmpB into stalled ribosomes: a cryo-EM study. RNA, 2010, 16: 299–306
Guda C, Lu S, Scheeff ED, Bourne PE, Shindyalov IN. CE-MC: a multiple protein structure alignment server. Nucleic Acids Res, 2004, 32: W100–103
Gao YG, Selmer M, Dunham CM, Weixlbaumer A, Kelley AC, Ramakrishnan V. The structure of the ribosome with elongation factor G trapped in the posttranslocational state. Science, 2009, 326: 694–699
Dunkle JA, Wang L, Feldman MB, Pulk A, Chen VB, Kapral GJ, Noeske J, Richardson JS, Blanchard SC, Cate JH. Structures of the bacterial ribosome in classical and hybrid states of tRNA binding. Science, 2011, 332: 981–984
Adamski FM, Atkins JF, Gesteland RF. Ribosomal protein L9 interactions with 23 S rRNA: the use of a translational bypass assay to study the effect of amino acid substitutions. J Mol Biol, 1996, 261: 357–371
Ohtani N, Tomita M, Itaya M. An extreme thermophile, Thermus thermophilus, is a polyploid bacterium. J Bacteriol, 2010, 192: 5499–5505
Bulkley D, Innis CA, Blaha G, Steitz TA. Revisiting the structures of several antibiotics bound to the bacterial ribosome. Proc Natl Acad Sci USA, 2010, 107: 17158–17163
Samanta D, Das A, Bhattacharya A, Basu A, Das D, DasGupta C. Mechanism of ribosome assisted protein folding: a new insight into rRNA functions. Biochem Biophys Res Commun, 2009, 384: 137–140
Mitra K, Schaffitzel C, Fabiola F, Chapman MS, Ban N, Frank J. Elongation arrest by SecM via a cascade of ribosomal RNA rearrangements. Mol Cell, 2006, 22: 533–543
Namy O, Moran SJ, Stuart DI, Gilbert RJ, Brierley I. A mechanical explanation of RNA pseudoknot function in programmed ribosomal frameshifting. Nature, 2006, 441: 244–247
Demeshkina N, Jenner L, Yusupova G, Yusupov M. Interactions of the ribosome with mRNA and tRNA. Curr Opin Struct Biol, 2010, 20: 325–332
Ramakrishnan V. The ribosome: some hard facts about its structure and hot air about its evolution. Cold Spring Harb Symp Quant Biol, 2009, 74: 25–33
Rosorius O, Fries B, Stauber RH, Hirschmann N, Bevec D, Hauber J. Human ribosomal protein L5 contains defined nuclear localization and export signals. J Biol Chem, 2000, 275: 12061–12068
Clore GM, Iwahara J Theory, practice, and applications of paramagnetic relaxation enhancement for the characterization of transient low-population states of biological macromolecules and their complexes. Chem Rev, 2009, 109: 4108–4139
Gold ND, Jackson RM. SitesBase: a database for structure-based protein-ligand binding site comparisons. Nucleic Acids Res, 2006, 34: D231–234
Giedroc DP, Cornish PV. Frameshifting RNA pseudoknots: structure and mechanism. Virus Res, 2009, 139: 193–208
Cornish PV, Ermolenko DN, Staple DW, Hoang L, Hickerson RP, Noller HF, Ha T. Following movement of the L1 stalk between three functional states in single ribosomes. Proc Natl Acad Sci USA, 2009, 106: 2571–2576
Takyar S, Hickerson RP, Noller HF. mRNA helicase activity of the ribosome. Cell, 2005, 120: 49–58
Williamson JR. Biophysical studies of bacterial ribosome assembly. Curr Opin Struct Biol, 2008, 18: 299–304
Author information
Authors and Affiliations
Corresponding authors
Additional information
Contributed equally to this work
This article is published with open access at link.springer.com
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0), which permits use, duplication, adaptation, distribution, and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Yu, D., Zhang, C., Qin, P. et al. RNA-protein distance patterns in ribosomes reveal the mechanism of translational attenuation. Sci. China Life Sci. 57, 1131–1139 (2014). https://doi.org/10.1007/s11427-014-4753-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-014-4753-8