Abstract
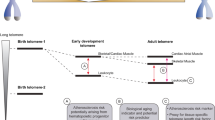
A consistent association has been observed between leukocyte telomere length (LTL) and atherosclerosis, but the mechanisms underlying these associations are still not well understood. Premature biology aging was evident in atherosclerotic plaques, characterized by reduced cell proliferation, irreversible growth arrest and apoptosis, and telomere attrition. As atherosclerosis is a state of chronic low-grade inflammation and increased oxidative stress, shortened LTL in patients with atherosclerosis might stem from the two sources, one is an accelerated rate in hematopoietic stem cells (HSCs) replication to replace leukocytes consumed in the inflammatory process, and another is the increase in the loss of telomere repeats per replication. Thus, diminished HSC reserves at birth and age-dependent telomere attrition afterward are mirrored in shortened LTL during the adulthood. In addition, the inter-individual variation of LTL in the general population can be partly explained by genetic factors regulating telomere maintenance and the rate of HSCs replication. Atherosclerosis is an aging-related disease, and practically all humans develop atherosclerosis if they live long enough. Here we overview the potential roles of LTL dynamics in the imbalance between injurious oxidative stress/inflammation and endothelial repair during the pathogenesis of age-related atherosclerosis, and discuss the possibility that preventing accelerated cellular senescence is a potential target in prevention of atherosclerosis.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Calado RT, Young NS. Telomere diseases. N Engl J Med, 2009, 361: 2353–2365
Blackburn EH. Structure and function of telomeres. Nature, 1991, 350: 569–573
Nilsson PM, Tufvesson H, Leosdottir M, Melander O. Telomeres and cardiovascular disease risk: an update 2013. Transl Res, 2013, 162: 371–380
O’Rourke MF, Hashimoto J. Mechanical factors in arterial aging: a clinical perspective. J Am Coll Cardiol, 2007, 50: 1–13
Matthews C, Gorenne I, Scott S, Figg N, Kirkpatrick P, Ritchie A, Goddard M, Bennett M. Vascular smooth muscle cells undergo telomere-based senescence in human atherosclerosis: effects of telomerase and oxidative stress. Circ Res, 2006, 99: 156–164
Minamino T, Miyauchi H, Yoshida T, Komuro I. Endothelial cell senescence in human atherosclerosis: role of telomere in endothelial dysfunction. Circulation, 2002, 105: 1541–1544
Okuda K, Bardeguez A, Gardner JP, Rodriguez P, Ganesh V, Kimura M, Skurnick J, Awad G, Aviv A. Telomere length in the newborn. Pediatr Res, 2002, 52: 377–381
Hewitt G, Jurk D, Marques FD, Correia-Melo C, Hardy T, Gackowska A, Anderson R, Taschuk M, Mann J, Passos JF. Telomeres are favored targets of a persistent DNA damage response in ageing and stress-induced senescence. Nat Commun, 2012, 3: 708
Ogami M, Ikura Y, Ohsawa M, Matsuo T, Kayo S, Yoshimi N, Hai E, Shirai N, Ehara S, Komatsu R, Naruko T, Ueda M. Telomere shortening in human coronary artery diseases. Arterioscler Thromb Vasc Biol, 2004, 24: 546–550
Chang E, Harley CB. Telomere length and replicative aging in human vascular tissues. Proc Natl Acad Sci USA, 1995, 92: 11190–11194
Brouilette SW, Moore JS, McMahon AD, Thompson JR, Ford I, Shepherd J, Packard CJ, Samani NJ; West of Scotland Coronary Prevention Study Group. Telomere length, risk of coronary heart disease, and statin treatment in the West of Scotland Primary Prevention Study: a nested case-control study. Lancet, 2007, 369: 107–114
O’onnell CJ, Demissie S, Kimura M, Levy D, Gardner JP, White C, D’gostino RB, Wolf PA, Polak J, Cupples LA, Aviv A. Leukocyte telomere length and carotid artery intimal medial thickness: the Framingham Heart Study. Arterioscler Thromb Vasc Biol, 2008, 28: 1165–1171
Willeit P, Willeit J, Brandstätter A, Ehrlenbach S, Mayr A, Gasperi A, Weger S, Oberhollenzer F, Reindl M, Kronenberg F, Kiechl S. Cellular aging reflected by leukocyte telomere length predicts advanced atherosclerosis and cardiovascular disease risk. Arterioscler Thromb Vasc Biol, 2010, 30: 1649–1656
Zhang W, Chen Y, Wang Y, Liu P, Zhang M, Zhang C, Hu FB, Hui R. Short telomere length in blood leukocytes contributes to the presence of atherothrombotic stroke and hemorrhagic stroke and risk of post-stroke death. Clin Sci, 2013, 125: 27–36
Kimura M, Hjelmborg JB, Gardner JP, Bathum L, Brimacombe M, Lu X, Christiansen L, Vaupel JW, Aviv A, Christensen K. Short leukocyte telomeres forecast mortality: a study in elderly Danish twins. Am J Epidemiol, 2008, 167: 799–806
Aviv A, Chen W, Gardner JP, Kimura M, Brimacombe M, Cao X, Srinivasan SR, Berenson GS. Leukocyte telomere dynamics: longitudinal findings among young adults in the Bogalusa Heart Study. Am J Epidemiol, 2009, 169: 323–329
Xiao Q, Kiechl S, Patel S, Oberhollenzer F, Weger S, Mayr A, Metzler B, Reindl M, Hu Y, Willeit J, Xu Q. Endothelial progenitor cells, cardiovascular risk factors, cytokine levels and atherosclerosis—results from a large population-based study. PLoS ONE, 2007, 2: e975
Andrew T, Aviv A, Falchi M, Surdulescu GL, Gardner JP, Lu X, Kimura M, Kato BS, Valdes AM, Spector TD. Mapping genetic loci that determine leukocyte telomere length in a large sample of unselected female sibling pairs. Am J Hum Genet, 2006, 78: 480–486
Armanios M, Blackburn EH. The telomere syndromes. Nat Rev Genet, 2012, 13: 693–704
Levy D, Neuhausen SL, Hunt SC, Kimura M, Hwang SJ, Chen W, Bis JC, Fitzpatrick AL, Smith E, Johnson AD, Gardner JP, Srinivasan SR, Schork N, Rotter JI, Herbig U, Psaty BM, Sastrasinh M, Murray SS, Vasan RS, Province MA, Glazer NL, Lu X, Cao X, Kronmal R, Mangino M, Soranzo N, Spector TD, Berenson GS, Aviv A. Genome-wide association identifies OBFC1 as a locus involved in human leukocyte telomere biology. Proc Natl Acad Sci USA, 2010, 107: 9293–9298
Codd V, Mangino M, van der Harst P, Braund PS, Kaiser M, Beveridge AJ, Rafelt S, Moore J, Nelson C, Soranzo N, Zhai G, Valdes AM, Blackburn H, Mateo Leach I, de Boer RA, Kimura M, Aviv A; Wellcome Trust Case Control Consortium, Goodall AH, Ouwehand W, van Veldhuisen DJ, van Gilst WH, Navis G, Burton PR, Tobin MD, Hall AS, Thompson JR, Spector T, Samani NJ. Common variants near TERC are associated with mean telomere length. Nat Genet, 2010, 42: 197–199
Li S, Makovets S, Matsuguchi T, Blethrow JD, Shokat KM, Blackburn EH. Cdk1-dependent phosphorylation of Cdc13 coordinates telomere elongation during cell-cycle progression. Cell, 2009, 136: 50–61
Eash KJ, Means JM, White DW, Link DC. CXCR4 is a key regulator of neutrophil release from the bone marrow under basal and stress granulopoiesis conditions. Blood, 2009, 113: 4711–4719
Sainz J, Sata M. Open sesame! CXCR4 blockade recruits neutrophils into the plaque. Circ Res, 2008, 102: 154–156
Myocardial Infarction Genetics Consortium. Genome-wide association of early-onset myocardial infarction with single nucleotide polymorphisms and copy number variants. Nat Genet, 2009, 41: 334–341
Boccardi V, Paolisso G. Telomerase activation: a potential key modulator for human healthspan and longevity. Ageing Res Rev, 2014, 15C: 1–5
Concetti F, Carpi FM, Nabissi M, Picciolini M, Santoni G, Napolioni V. The functional polymorphism rs73598374:G>A (p.Asp8Asn) of the ADA gene is associated with telomerase activity and leukocyte telomere length. Eur J Hum Genet, 2014, doi: 10.1038/ejhg.2014.102
Yu TY, Kao YW, Lin JJ. Telomeric transcripts stimulate telomere recombination to suppress senescence in cells lacking telomerase. Proc Natl Acad Sci USA, 2014, 111: 3377–3382
Frenck RW Jr, Blackburn EH, Shannon KM. The rate of telomere sequence loss in human leukocytes varies with age. Proc Natl Acad Sci USA, 1998, 95: 5607–5610
Matthews C, Gorenne I, Scott S, Figg N, Kirkpatrick P, Ritchie A, Goddard M, Bennett M. Vascular smooth muscle cells undergo telomere-based senescence in human atherosclerosis: effects of telomerase and oxidative stress. Circ Res, 2006, 99: 156–164
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at link.springer.com ag
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0), which permits use, duplication, adaptation, distribution, and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Zhang, W., Hui, R. & Yang, S. Telomeres, cardiovascular aging, and potential intervention for cellular senescence. Sci. China Life Sci. 57, 858–862 (2014). https://doi.org/10.1007/s11427-014-4700-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-014-4700-8