Abstract
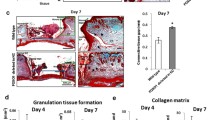
Myofibroblasts, recognized classically by α-smooth muscle actin (α-SMA) expression, play a key role in the wound-healing process, promoting wound closure and matrix deposition. Although a body of evidence shows that keratinocytes explanted onto a wound bed promote closure of a skin injury, the underlying mechanisms are not well understood. The basal layer of epidermis is rich in undifferentiated keratinocytes (UKs). We showed that UKs injected into granulation tissue could switch into α-SMA positive cells, and accelerate the rate of skin wound healing. In addition, when the epidermis sheets isolated from foreskin cover up the wound bed or are induced in vitro, keratinocytes located at the basal layers or adjacent sites were observed to convert into myofibroblast-like cells. Thus, UKs have a potential for myofibroblastic transition, which provides a novel mechanism by which keratinocyte explants accelerate skin wound healing.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Modarressi A, Pietramaggiori G, Godbout C, Vigato E, Pittet B, Hinz B. Hypoxia impairs skin myofibroblast differentiation and function. J Invest Dermatol, 2010, 130: 2818–2827
Hartmann A, Quist J, Hamm H, Bröcker EB, Friedl P. Transplantation of autologous keratinocyte suspension in fibrin matrix to chronic venous leg ulcers: improved long-term healing after removal of the fibrin carrier. Dermatol Surg, 2008, 34: 922–929
Xiao SC, Zhu SH, Li HY, Yang J, Xia ZF. A new strategy of using keratinocytes for skin graft: animal experimental study of keratinocyte suspension combined with the dermal substitutes. Eur Surg Res, 2009, 43: 1–7
James SE, Booth S, Dheansa B, Mann DJ, Reid MJ, Shevchenko RV, Gilbert PM. Sprayed cultured autologous keratinocytes used alone or in combination with meshed autografts to accelerate wound closure in difficult-to-heal burns patients. Burns, 2010, 36: e10–20
Rasmussen CA, Gibson AL, Schlosser SJ, Schurr MJ, Allen-Hoffmann BL. Chimeric composite skin substitutes for delivery of autologous keratinocytes to promote tissue regeneration. Ann Surg, 2010, 251: 368–376
Wiwanitkit V. Novel approach for chronic wound treatment by autologous transplantation. Can J Surg, 2010, 53: E7
Mannik J, Alzayady K, Ghazizadeh S. Regeneration of multilineage skin epithelia by differentiated keratinocytes. J Invest Dermatol, 2010, 130: 388–397
Lugo LM, Lei P, Andreadis ST. Vascularization of the dermal support enhances wound re-epithelialization by in situ delivery of epidermal keratinocytes. Tissue Eng Part A, 2011, 17: 665–675
Huckfeldt R, Flick AB, Mikkelson D, Lowe C, Finley PJ. Wound closure after split-thickness skin grafting is accelerated with the use of continuous direct anodal microcurrent applied to silver nylon wound contact dressings. J Burn Care Res, 2007, 28: 703–707
Jewell L, Guerrero R, Quesada AR, Chan LS, Garner WL. Rate of healing in skin-grafted burn wounds. Plast Reconstr Surg, 2007, 120: 451–456
Mahmoud SM, Mohamed AA, Mahdi SE, Ahmed ME. Split-skin graft in the management of diabetic foot ulcers. J Wound Care, 2008, 17: 303–306
Bukovcan P, Koller J. Treatment of partial-thickness scalds by skin xenografts—a retrospective study of 109 cases in a three-year period. Acta Chir Plast, 2010, 52: 7–12
Fragiadaki M, Mason RM. Epithelial-mesenchymal transition in renal fibrosis—evidence for and against. Int J Exp Pathol, 2011, 92: 143–150
Parola M, Pinzani M. Hepatic wound repair. Fibrogenesis Tissue Repair, 2009, 2: 4
Chapman HA. Epithelial-mesenchymal interactions in pulmonary fibrosis. Annu Rev Physiol, 2011, 73: 413–435
Nakamura M, Tokura Y. Epithelial-mesenchymal transition in the skin. J Dermatol Sci, 2011, 61: 7–13
Kalluri R, Neilson EG. Epithelial-mesenchymal transition and its implications for fibrosis. J Clin Invest, 2003, 112: 1776–1784
Han W, Chen M, Li M, Wu Z, Zhao Y, Wang Y, Wang L, Yu L, Fu X. Acclimatized induction reveals the multipotency of adult human undifferentiated keratinocytes. Cell Reprogram, 2010, 12: 283–294
Terada N, Hamazaki T, Oka M, Hoki M, Mastalerz DM, Nakano Y, Meyer EM, Morel L, Petersen BE, Scott EW. Bone marrow cells adopt the phenotype of other cells by spontaneous cell fusion. Nature, 2002, 416: 542–545
Weimann JM, Johansson CB, Trejo A, Blau HM. Stable reprogrammed heterokaryons form spontaneously in Purkinje neurons after bone marrow transplantation. Nat Cell Biol, 2003, 5: 959–966
Sarig R, Fuchs O, Tencer L, Panski A, Nudel U, Yaffe D. Cloned myogenic cells can transdifferentiate in vivo into neuron-like cells. PLoS ONE, 2010, 5: e8814
Wood FM, Kolybaba ML, Allen P. The use of cultured epithelial autograft in the treatment of major burn injuries: a critical review of the literature. Burns, 2006, 32: 395–401
Reid MJ, Currie LJ, James SE, Sharpe JR. Effect of artificial dermal substitute, cultured keratinocytes and split thickness skin graft on wound contraction. Wound Repair Regen, 2007, 15: 889–896
Shukla VK, Tiwary SK, Barnwal S, Gulati AK, Pandey SS. Effect of autologous epidermal cell suspension transplantation in chronic non-healing wounds: a pilot study. Can J Surg, 2010, 53: 6–10
Kiwanuka E, Hackl F, Philip J, Caterson EJ, Junker JP, Eriksson E. Comparison of healing parameters in porcine full-thickness wounds transplanted with skin micrografts, split-thickness skin grafts, and cultured keratinocytes. J Am Coll Surg, 2011, 213: 728–735
Dedja A, Zaglia T, Dall’Olmo L, Chioato T, Thiene G, Fabris L, Ancona E, Schiaffino S, Ausoni S, Cozzi E. Hybrid cardiomyocytes derived by cell fusion in heterotopic cardiac xenografts. FASEB J, 2006, 20: 2534–2536
Rappersberger K, Binder M, Zonzits E, Hornick U, Wolff K. Immunogold staining of intermediate-sized filaments of the vimentin type in human skin: a postembedding immunoelectron microscopic study. J Invest Dermatol, 1990, 94: 700–705
Dean NR, Brennan J, Haynes J, Goddard C, Cooter RD. Immunohistochemical labeling of normal melanocytes. Appl Immunohistochem Mol Morphol, 2002, 10: 199–204
Author information
Authors and Affiliations
Corresponding authors
Additional information
This article is published with open access at link.springer.com
Electronic supplementary material
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 2.0 International License (https://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Li, M., Ti, D., Han, W. et al. Microenvironment-induced myofibroblast-like conversion of engrafted keratinocytes. Sci. China Life Sci. 57, 209–220 (2014). https://doi.org/10.1007/s11427-014-4613-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-014-4613-6