Abstract
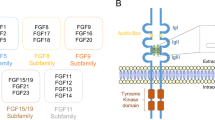
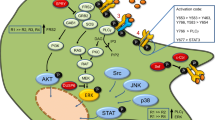
Fibroblast growth factors (FGFs) can be classified as secretory (FGF1-10 and FGF15-23) or intracellular non-secretory forms (FGF11-14). Secretory forms of FGF and their receptors are best known for their regulatory roles in cell growth, differentiation and morphogenesis in the early stages of neural development. However, the functions of intracellular FGFs remain to be explored. FGF12 and FGF14 are found to interact with voltage-gated sodium channels, and regulate the channel activity in neurons. FGF13 is expressed in primary sensory neurons, and is colocalized with sodium channels at the nodes of Ranvier along the myelinated afferent fibers. FGF13 is also expressed in cerebral cortical neurons during the late developmental stage. A recent study showed that FGF13 is a microtubule-stabilizing protein required for regulating the neuronal development in the cerebral cortex. Thus, non-secretory forms of FGF appear to have important roles in the brain, and it would be interesting to further investigate the functions of intracellular FGFs in the nervous system and in neural diseases.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Burgess W H, Maciag T. The heparin-binding (fibroblast) growth factor family of proteins. Annu Rev Biochem, 1989, 58: 575–606
Rifkin D B, Moscatelli D. Recent developments in the cell biology of basic fibroblast growth factor. J Cell Biol, 1989, 109: 1–6
Yamaguchi T P, Rossant J. Fibroblast growth factors in mammalian development. Curr Opin Genet Dev, 1995, 5: 485–91
Guillemot F, Zimmer C. From cradle to grave: the multiple roles of fibroblast growth factors in neural development. Neuron, 2011, 71: 574–588
Goldfarb M. Fibroblast growth factor homologous factors: evolution, structure, and function. Cytokine Growth Factor Rev, 2005, 16: 215–220
Itoh N, Ornitz D M. Functional evolutionary history of the mouse Fgf gene family. Dev Dyn, 2008, 237: 18–27
Smallwood P M, Munoz-Sanjuan I, Tong P, et al. Fibroblast growth factor (FGF) homologous factors: new members of the FGF family implicated in nervous system development. Proc Natl Acad Sci USA, 1996, 93: 9850–9857
Turner C A, Watson S J, Akil H. The fibroblast growth factor family: neuromodulation of affective behavior. Neuron, 2012, 76: 160–174
Wu Q F, Yang L, Li S, et al. Fibroblast growth factor 13 is a microtubule-stabilizing protein regulating neuronal polarization and migration. Cell, 2012, 149: 1549–1564
Wittmack E K, Rush A M, Craner M J, et al. Fibroblast growth factor homologous factor 2B: association with Nav1.6 and selective colocalization at nodes of Ranvier of dorsal root axons. J Neurosci, 2004, 24: 6765–6775
Kole M H, Stuart G J. Signal processing in the axon initial segment. Neuron, 2012, 73: 235–247
Gedeon A K, Glass I A, Connor J M, et al. Genetic localisation of MRX27 to Xq24-26 defines another discrete gene for non-specific X-linked mental retardation. Am J Med Genet, 1996, 64: 121–124
Wozniak D F, Xiao M L, Xu L, et al. Impaired spatial learning and defective theta burst induced LTP in mice lacking fibroblast growth factor 14. Neurobiol Dis, 2007, 26: 14–26
Brusse E, de Koning I, Maat-Kievit A, et al. Spinocerebellar ataxia associated with a mutation in the fibroblast growth factor 14 gene (SCA27): A new phenotype. Mov Disord, 2006, 21: 396–401
Shakkottai VG, Xiao M, Xu L, et al. FGF14 regulates the intrinsic excitability of cerebellar Purkinje neurons. Neurobiol Dis, 2009, 33: 81–88
Xiao M, Xu L, Laezza F, et al. Impaired hippocampal synaptic transmission and plasticity in mice lacking fibroblast growth factor 14. Mol Cell Neurosci, 2007, 34: 366–377
Goetz R, Dover K, Laezza F, et al. Crystal structure of a fibroblast growth factor homologous factor (FHF) defines a conserved surface on FHFs for binding and modulation of voltage-gated sodium channels. J Biol Chem, 2009, 284: 17883–17896
Olsen S K, Garbi M, Zampieri N, et al. Fibroblast growth factor (FGF) homologous factors share structural but not functional homology with FGFs. J Biol Chem, 2003, 278: 34226–34236
Greene J M, Li Y L, Yourey P A, et al. Identification and characterization of a novel member of the fibroblast growth factor family. Eur J Neurosci, 1998, 10: 1911–1925
Dalski A, Atici J, Kreuz F R, et al. Mutation analysis in the fibroblast growth factor 14 gene: frameshift mutation and polymorphisms in patients with inherited ataxias. Eur J Hum Genet, 2005, 13: 118–120
Hartung H, Feldman B, Lovec H, et al. Murine FGF-12 and FGF-13: expression in embryonic nervous system, connective tissue and heart. Mech Dev, 1997, 64: 31–39
Schoorlemmer J, Goldfarb M. Fibroblast growth factor homologous factors are intracellular signaling proteins. Curr Biol, 2001, 11: 793–797
Li G D, Wo Y, Zhong M F, et al. Expression of fibroblast growth factors in rat dorsal root ganglion neurons and regulation after peripheral nerve injury. Neuroreport, 2002, 13: 1903–1907
Xiao H S, Huang Q H, Zhang F X, et al. Identification of gene expression profile of dorsal root ganglion in the rat peripheral axotomy model of neuropathic pain. Proc Natl Acad Sci USA, 2002, 99: 8360–8365
Zhang X, Xiao H S. Gene array analysis to determine the components of neuropathic pain signaling. Curr Opin Mol Ther, 2005, 7: 532–537
Li K C, Zhang F X, Li C L, et al. Follistatin-like 1 suppresses sensory afferent transmission by activating Na+,K+-ATPase. Neuron, 2011, 69: 974–987
Li K C, Wang F, Zhong Y Q, et al. Reduction of follistatin-like 1 in primary afferent neurons contributes to neuropathic pain hypersensitivity. Cell Res, 2011, 21: 697–699
Zhang X. Pain research in China. Sci China Life Sci, 2010, 53: 356–362
Hucho T, Levine J D. Signaling pathways in sensitization: toward a nociceptor cell biology. Neuron, 2007, 55: 365–376
Campbell J N, Meyer R A. Mechanisms of neuropathic pain. Neuron, 2006, 52: 77–92
Lou J Y, Laezza F, Gerber B R, et al. Fibroblast growth factor 14 is an intracellular modulator of voltage-gated sodium channels. J Physiol, 2005, 569: 179–193
Goldfarb M, Schoorlemmer J, Williams A, et al. Fibroblast growth factor homologous factors control neuronal excitability through modulation of voltage-gated sodium channels. Neuron, 2007, 55: 449–463
Thaxton C, Pillai A M, Pribisko A L, et al. Nodes of Ranvier act as barriers to restrict invasion of flanking paranodal domains in myelinated axons. Neuron, 2011, 69: 244–257
Debanne D. The nodal origin of intrinsic bursting. Neuron, 2011, 71: 569–570
Kole M H. First node of Ranvier facilitates high-frequency burst encoding. Neuron, 2011, 71: 671–682
Liu C, Dib-Hajj S D, Waxman S G. Fibroblast growth factor homologous factor 1B binds to the C terminus of the tetrodotoxin-resistant sodium channel rNav1.9a (NaN). J Biol Chem, 2001, 276: 18925–18933
Liu CJ, Dib-Hajj S D, Renganathan M, et al. Modulation of the cardiac sodium channel Nav1.5 by fibroblast growth factor homologous factor 1B. J Biol Chem, 2003, 278: 1029–1036
Laezza F, Lampert A, Kozel M A, et al. FGF14 N-terminal splice variants differentially modulate Nav1.2 and Nav1.6-encoded sodium channels. Mol Cell Neurosci, 2009, 42: 90–101
Rush A M, Wittmack E K, Tyrrell L, et al. Differential modulation of sodium channel Nav1.6 by two members of the fibroblast growth factor homologous factor 2 subfamily. Eur J Neurosci, 2006, 23: 2551–2562
Wang Q, Bardgett M E, Wong M, et al. Ataxia and paroxysmal dyskinesia in mice lacking axonally transported FGF14. Neuron, 2002, 35: 25–38
van Swieten J C, Brusse E, de Graaf B M, et al. A mutation in the fibroblast growth factor 14 gene is associated with autosomal dominant cerebellar ataxia. Am J Hum Genet, 2003, 72: 191–199
Laezza F, Gerber B R, Lou J Y, et al. The FGF14F145S mutation disrupts the interaction of FGF14 with voltage-gated Na+ channels and impairs neuronal excitability. J Neurosci, 2007, 27: 12033–12044
Nishimoto S, Nishida E. Fibroblast growth factor 13 is essential for neural differentiation in Xenopus early embryonic development. J Biol Chem, 2007, 282: 24255–24261
Munoz-Sanjuan I, Simandl B K, Fallon J F, et al. Expression of chicken fibroblast growth factor homologous factor (FHF)-1 and of differentially spliced isoforms of FHF-2 during development and involvement of FHF-2 in chicken limb development. Development, 1999, 126: 409–421
Yao D L, Masonic K, Petullo D, et al. Pretreatment with intravenous FGF-13 reduces infarct volume and ameliorates neurological deficits following focal cerebral ischemia in rats. Brain Res, 1999, 818: 140–146
Ayala R, Shu T, Tsai L H. Trekking across the brain: the journey of neuronal migration. Cell, 2007, 128: 29–43
Marin O, Valiente M, Ge X, et al. Guiding neuronal cell migrations. Cold Spring Harb Perspect Biol, 2010, 2: a001834
Yuan X. Axon guidance and neuronal migration research in China. Sci China Life Sci, 2010, 53: 304–314
Jaglin X H, Chelly J. Tubulin-related cortical dysgeneses: microtubule dysfunction underlying neuronal migration defects. Trends Genet, 2009, 25: 555–566
Witte H, Neukirchen D, Bradke F. Microtubule stabilization specifies initial neuronal polarization. J Cell Biol, 2008, 180: 619–632
Li L, Wei D, Wang Q, et al. MEC-17 deficiency leads to reduced α-tubulin acetylation and impaired migration of cortical neurons. J Neurosci, 2012, 32: 12673–12683
Poulain F E, Sobel A. The microtubule network and neuronal morphogenesis: Dynamic and coordinated orchestration through multiple players. Mol Cell Neurosci, 2010, 43: 15–32
Teng J, Takei Y, Harada A, et al. Synergistic effects of MAP2 and MAP1B knockout in neuronal migration, dendritic outgrowth, and microtubule organization. J Cell Biol, 2001, 155: 65–76
Bai J, Ramos R L, Ackman J B, et al. RNAi reveals doublecortin is required for radial migration in rat neocortex. Nat Neurosci, 2003, 6: 1277–1283
Kappeler C, Saillour Y, Baudoin J P, et al. Branching and nucleokinesis defects in migrating interneurons derived from doublecortin knockout mice. Hum Mol Genet, 2006, 15: 1387–1400
Bouquet C, Soares S, von Boxberg Y, et al. Microtubule-associated protein 1B controls directionality of growth cone migration and axonal branching in regeneration of adult dorsal root ganglia neurons. J Neurosci, 2004, 24: 7204–7213
Takei Y, Teng J, Harada A, et al. Defects in axonal elongation and neuronal migration in mice with disrupted tau and map1b genes. J Cell Biol, 2000, 150: 989–1000
Koizumi H, Higginbotham H, Poon T, et al. Doublecortin maintains bipolar shape and nuclear translocation during migration in the adult forebrain. Nat Neurosci, 2006, 9: 779–786
Lubs H A, Stevenson R E, Schwartz C E. Fragile X and X-linked intellectual disability: four decades of discovery. Am J Hum Genet, 2012, 90: 579–590
Tarpey P S, Smith R, Pleasance E, et al. A systematic, large-scale resequencing screen of X-chromosome coding exons in mental retardation. Nat Genet, 2009, 41: 535–543
Gecz J, Shoubridge C, Corbett M. The genetic landscape of intellectual disability arising from chromosome X. Trends Genet, 2009, 25: 308–316
Malmgren H, Sundvall M, Dahl N, et al. Linkage mapping of a severe X-linked mental retardation syndrome. Am J Hum Genet, 1993, 52: 1046–1052
Gecz J, Baker E, Donnelly A, et al. Fibroblast growth factor homologous factor 2 (FHF2): gene structure, expression and mapping to the Borjeson-Forssman-Lehmann syndrome region in Xq26 delineated by a duplication breakpoint in a BFLS-like patient. Hum Genet, 1999, 104: 56–63
Shiloh Y, Litvak G, Ziv Y, et al. Genetic mapping of X-linked albinism-deafness syndrome (ADFN) to Xq26.3-q27.I. Am J Hum Genet, 1990, 47: 20–27
Solomon N M, Nouri S, Warne G L, et al. Increased gene dosage at Xq26-q27 is associated with X-linked hypopituitarism. Genomics, 2002, 79: 553–559
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at Springerlink.com
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 2.0 International License (https://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Zhang, X., Bao, L., Yang, L. et al. Roles of intracellular fibroblast growth factors in neural development and functions. Sci. China Life Sci. 55, 1038–1044 (2012). https://doi.org/10.1007/s11427-012-4412-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-012-4412-x