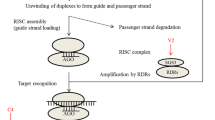
Abstract
Gene silencing (RNA silencing) plays a fundamental role in antiviral defense in plants, fungi and invertebrates. Viruses encode proteins that suppress gene silencing to counter host defense. Viral suppressors of RNA silencing (VSRs) have been identified from almost all plant virus genera and some viruses of insects and mammals. Recent studies have revealed that VSRs counter host defense and interfere with host gene regulation by interacting with RNA or important components of the RNA silencing pathway. Here, we review the current understanding of the complex mechanisms of VSRs that have been revealed by recent studies.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Ding S W. RNA-based antiviral immunity. Nat Rev Immunol, 2010, 10: 632–644
Chapman E J, Carrington J C. Specialization and evolution of endogenous small RNA pathways. Nat Rev Genet, 2007, 8: 884–896
Carthew R W, Sontheimer E J. Origins and mechanisms of miRNAs and siRNAs. Cell, 2009, 136: 642–655
Hutvagner G, Simard M J. Argonaute proteins: key players in RNA silencing. Nat Rev Mol Cell Biol, 2008, 9: 22–32
Voinnet O. Use, tolerance and avoidance of amplified RNA silencing by plants. Trends Plant Sci, 2008, 13: 317–328
Burgyan J, Havelda Z. Viral suppressors of RNA silencing. Trends Plant Sci, 2011, 16: 265–272
Deleris A, Gallego-Bartolome J, Bao J, et al. Hierarchical action and inhibition of plant Dicer-like proteins in antiviral defense. Science, 2006, 313: 68–71
Diaz-Pendon J A, Li F, Li W X, et al. Suppression of antiviral silencing by Cucumber mosaic virus 2b protein in Arabidopsis is associated with drastically reduced accumulation of three classes of viral small interfering RNAs. Plant Cell, 2007, 19: 2053–2063
Qu F, Ye X, Morris T J. Arabidopsis DRB4, AGO1, AGO7, and RDR6 participate in a DCL4-initiated antiviral RNA silencing pathway negatively regulated by DCL1. Proc Natl Acad Sci USA, 2008, 105: 14732–14737
Llave C. Virus-derived small interfering RNAs at the core of plant-virus interactions. Trends Plant Sci, 2010, 15: 701–707
Qu F. Antiviral role of plant-encoded RNA-dependent RNA polymerases revisited with deep sequencing of small interfering RNAs of virus origin. Mol Plant Microbe In, 2010, 23: 1248–1252
Qi X, Bao F S, Xie Z. Small RNA deep sequencing reveals role for Arabidopsis thaliana RNA-dependent RNA polymerases in viral siRNA biogenesis. PLoS ONE, 2009, 4: e4971
Garcia-Ruiz H, Takeda A, Chapman E J, et al. Arabidopsis RNA-dependent RNA polymerases and dicer-like proteins in antiviral defense and small interfering RNA biogenesis during Turnip Mosaic Virus infection. Plant Cell, 2010, 22: 481–496
Wang X B, Jovel J, Udomporn P, et al. The 21-nucleotide, but not 22-nucleotide, viral secondary small interfering RNAs direct potent antiviral defense by two cooperative Argonautes in Arabidopsis thaliana. Plant Cell, 2011, 23: 1625–1638
Wang X B, Wu Q F, Ito T, et al. RNAi-mediated viral immunity requires amplification of virus-derived siRNAs in Arabidopsis thaliana. Proc Natl Acad Sci USA, 2010, 107: 484–489
Harvey J J W, Lewsey M G, Patel K, et al. An antiviral defense role of AGO2 in plants. PLoS ONE, 2011, 6: e14639
Mi S, Cai T, Hu Y, et al. Sorting of small RNAs into Arabidopsis argonaute complexes is directed by the 5′ terminal nucleotide. Cell, 2008, 133: 116–127
Vance V B, Anandalakshmi R, Pruss G J, et al. A viral suppressor of gene silencing in plants. Proc Natl Acad Sci USA, 1998, 95: 13079–13084
Baulcombe D C, Brigneti G, Voinnet O, et al. Viral pathogenicity determinants are suppressors of transgene silencing in Nicotiana benthamiana. EMBO J, 1998, 17: 6739–6746
Carrington J C, Kasschau K D. A counterdefensive strategy of plant viruses: Suppression of posttranscriptional gene silencing. Cell, 1998, 95: 461–470
Ding S W, Wu Q F, Wang X B. Viral suppressors of RNA-Based viral Immunity: host targets. Cell Host Microbe, 2010, 8: 12–15
Williamson J R, Chao J A, Lee J H, et al. Dual modes of RNA-silencing suppression by flock house virus protein B2. Nat Struct Mol Biol, 2005, 12: 952–957
Ding S W, Aliyari R, Wu Q F, et al. Mechanism of induction and suppression of antiviral immunity directed by virus-derived small RNAs in Drosophila. Cell Host Microbe, 2008, 4: 387–397
Silhavy D, Merai Z, Kerenyi Z, et al. Aureusvirus P14 is an efficient RNA silencing suppressor that binds double-stranded RNAs without size specificity. J Virol, 2005, 79: 7217–7226
Masuta C, Goto K, Kobori T, et al. Characterization of silencing suppressor 2b of cucumber mosaic virus based on examination of its small RNA-Binding abilities. Plant Cell Physiol, 2007, 48: 1050–1060
Chen J C H, Rashid U J, Hoffmann J, et al. Multiple targets for suppression of RNA interference by Tomato aspermy virus protein 2B. Biochemistry, 2008, 47: 12655–12657
AdamYuan Y, Chen H Y, Yang J, et al. Structural basis for RNA-silencing suppression by Tomato aspermy virus protein 2b. EMBO Rep, 2008, 9: 754–760
Hall T M T, Vargason J M, Szittya G, et al. Size selective recognition of siRNA by an RNA silencing suppressor. Cell, 2003, 115: 799–811
Patel D J, Ye K Q, Malinina L. Recognition of small interfering RNA by a viral suppressor of RNA silencing. Nature, 2003, 426: 874–878
Silhavy D, Molnar A, Lucioli A, et al. A viral protein suppresses RNA silencing and binds silencing-generated, 21- to 25-nucleotide double-stranded RNAs. EMBO J, 2002, 21: 3070–3080
Burgyan J, Lakatos L, Szittya G, et al. Molecular mechanism of RNA silencing suppression mediated by p19 protein of tombusviruses. EMBO J, 2004, 23: 876–884
Burgyan J, Lakatos L, Csorba T, et al. Small RNA binding is a common strategy to suppress RNA silencing by several viral suppressors. EMBO J, 2006, 25: 2768–2780
Havelda Z, Hornyik C, Crescenzi A, et al. In situ characterization of Cymbidium Ringspot tombusvirus infection-induced posttranscriptional gene silencing in Nicotiana benthamiana. J Virol, 2003, 77: 6082–6086
Dunoyer P, Schott G, Himber C, et al. Small RNA duplexes function as mobile silencing signals between plant cells. Science, 2010, 328: 912–916
Burgyan J, Csorba T, Bovi A, et al. The p122 subunit of Tobacco mosaic virus replicase is a potent silencing suppressor and compromises both small interfering RNA- and MicroRNA-mediated pathways. J Virol, 2007, 81: 11768–11780
Unrau P J, Ebhardt H A, Thi E P, et al. Extensive 3′ modification of plant small RNAs is modulated by helper component-proteinase expression. Proc Natl Acad Sci USA, 2005, 102: 13398–13403
Burgyan J, Lozsa R, Csorba T, et al. Inhibition of 3′ modification of small RNAs in virus-infected plants require spatial and temporal co-expression of small RNAs and viral silencing-suppressor proteins. Nucleic Acids Res, 2008, 36: 4099–4107
Valkonen J P T, Kreuze J F, Savenkov E I, et al. Viral class 1 RNase III involved in suppression of RNA silencing. J Virol, 2005, 79: 7227–7238
Valkonen J P T, Cuellar W J, Kreuze J F, et al. Elimination of antiviral defense by viral RNase III. Proc Natl Acad Sci USA, 2009, 106: 10354–10358
Meister G, Peters L. Argonaute proteins: Mediators of RNA silencing. Mol Cell, 2007, 26: 611–623
Canto T, Gonzalez I, Martinez L, et al. Cucumber Mosaic Virus 2b Protein subcellular targets and interactions: their significance to RNA silencing suppressor activity. Mol Plant Microbe In, 2010, 23: 294–303
Chua N H, Zhang X R, Yuan Y R, et al. Cucumber mosaic virus-encoded 2b suppressor inhibits Arabidopsis Argonaute1 cleavage activity to counter plant defense. Genes Dev, 2006, 20: 3255–3268
Hamera S, Song X G, Su L, et al. Cucumber mosaic virus suppressor 2b binds to AGO4-related small RNAs and impairs AGO4 activities. Plant J, 2011, 69: 104–115
Pikaard C S, Pontes O, Li C F, et al. The Arabidopsis chromatin-modifying nuclear siRNA pathway involves a nucleolar RNA processing center. Cell, 2006, 126: 79–92
Prins M, Bucher E, Hemmes H, et al. The influenza A virus NS1 protein binds small interfering RNAs and suppresses RNA silencing in plants. J Gen Virol, 2004, 85: 983–991
Ding S W, Li W X, Li H W, et al. Interferon antagonist proteins of influenza and vaccinia viruses are suppressors of RNA silencing. Proc Natl Acad Sci USA, 2004, 101: 1350–1355
Ye K Q, Shen M, Xu Y, et al. Size-independent and noncooperative recognition of dsRNA by the Rice stripe virus RNA silencing suppressor NS3. J Mol Biol, 2010, 404: 665–679
Silhavy D, Merai Z, Kerenyi Z, et al. Double-stranded RNA binding may be a general plant RNA viral strategy to suppress RNA silencing. J Virol, 2006, 80: 5747–5756
Valli A, Oliveros J C, Molnar A, et al. The specific binding to 21-nt double-stranded RNAs is crucial for the anti-silencing activity of Cucumber vein yellowing virus P1b and perturbs endogenous small RNA populations. RNA, 2011, 17: 1148–1158
Lakatos L, Hemmes H, Goldbach R, et al. The NS3 protein of Rice hoja blanca tenuivirus suppresses RNA silencing in plant and insect hosts by efficiently binding both siRNAs and miRNAs. RNA, 2007, 13: 1079–1089
Ren B, Guo Y Y, Gao F, et al. Multiple functions of Rice dwarf phytoreovirus Pns10 in suppressing systemic RNA silencing. J Virol, 2010, 84: 12914–12923
Ziegler-Graff V, Pfeffer S, Dunoyer P, et al. P0 of beet western yellows virus is a suppressor of posttranscriptional gene silencing. J Virol, 2002, 76: 6815–6824
Baumberger N, Tsai C H, Lie M, et al. The Polerovirus silencing suppressor P0 targets ARGONAUTE proteins for degradation. Curr Biol, 2007, 17: 1609–1614
Bortolamiol D, Pazhouhandeh M, Marrocco K, et al. The Polerovirus F box protein P0 targets ARGONAUTE1 to suppress RNA silencing. Curr Biol, 2007, 17: 1615–1621
Mirkov T E, Mangwende T, Wang M L, et al. The P0 gene of Sugarcane yellow leaf virus encodes an RNA silencing suppressor with unique activities. Virology, 2009, 384: 38–50
Burgyan J, Csorba T, Lozsa R, et al. Polerovirus protein P0 prevents the assembly of small RNA-containing RISC complexes and leads to degradation of ARGONAUTE1. Plant J, 2010, 62: 463–472
Ziegler-Graff V, Pazhouhandeh M, Dieterle M, et al. F-box-like domain in the Polerovirus protein P0 is required for silencing suppressor function. Proc Natl Acad Sci USA, 2006, 103: 1994–1999
Azevedo J, Garcia D, Pontier D, et al. Argonaute quenching and global changes in Dicer homeostasis caused by a pathogen-encoded GW repeat protein. Genes Dev, 2010, 24: 904–915
Giner A, Lakatos L, Garcia-Chapa M, et al. Viral protein inhibits RISC activity by Argonaute binding through conserved WG/GW motifs. PLoS Pathog, 2010, 6: e1000996
Ladurner A G, Till S. RNA Pol IV plays catch with argonaute 4. Cell, 2007, 131: 643–645
Lagrange T, El-Shami M, Pontier D, et al. Reiterated WG/GW motifs form functionally and evolutionarily conserved ARGONAUTEbinding platforms in RNAi-related components. Genes Dev, 2007, 21: 2539–2544
Karlowski W M, Zielezinski A, Carrere J, et al. Genome-wide computational identification of WG/GW Argonaute-binding proteins in Arabidopsis. Nucleic Acids Res, 2010, 38: 4231–4245
Carrington J C, Xie Z X, Kasschau K D. Negative feedback regulation of Dicer-like1 in Arabidopsis by microRNA-guided mRNA degradation. Curr Biol, 2003, 13: 784–789
Chiu M H, Chen I H, Baulcombe D C, et al. The silencing suppressor P25 of Potato virus X interacts with Argonaute1 and mediates its degradation through the proteasome pathway. Mol Plant Pathol, 2010, 11: 641–649
Voinnet O, Lederer C, Baulcombe D C. A viral movement protein prevents spread of the gene silencing signal in Nicotiana benthamiana. Cell, 2000, 103: 157–167
Moissiard G, Parizotto E A, Himber C, et al. Transitivity in Arabidopsis can be primed, requires the redundant action of the antiviral Dicer-like 4 and Dicer-like 2, and is compromised by viral-encoded suppressor proteins. RNA, 2007, 13: 1268–1278
Bayne E H, Rakitina D V, Morozov S Y, et al. Cell-to-cell movement of Potato Potexvirus X is dependent on suppression of RNA silencing. Plant J, 2005, 44: 471–482
Havelda Z, Varallyay E, Valoczi A, et al. Plant virus-mediated induction of miR168 is associated with repression of ARGONAUTE1 accumulation. EMBO J, 2010, 29: 3507–3519
Du P, Wu J G, Zhang J Y, et al. Viral infection induces expression of novel phased microRNAs from conserved cellular microRNA precursors. PLoS Pathog, 2011, 7: e1002176
Ding S W, Guo H S. A viral protein inhibits the long range signaling activity of the gene silencing signal. EMBO J, 2002, 21: 398–407
Mochizuki T, Sunpapao A, Nakai T, et al. The 2b protein of cucumber mosaic virus is essential for viral infection of the shoot apical meristem and for efficient invasion of leaf primordia in infected tobacco plants. J Gen Virol, 2009, 90: 3015–3021
Schwach F, Vaistij F E, Jones L, et al. An RNA-dependent RNA polymerase prevents meristem invasion by potato virus X and is required for the activity but not the production of a systemic silencing signal. Plant Physiol, 2005, 138: 1842–1852
Gafni Y, Zrachya A, Glick E, et al. Suppressor of RNA silencing encoded by Tomato yellow leaf curl virus-Israel. Virology, 2007, 358: 159–165
Gafni Y, Glick E, Zrachya A, et al. Interaction with host SGS3 is required for suppression of RNA silencing by tomato yellow leaf curl virus V2 protein. Proc Natl Acad Sci USA, 2008, 105: 157–161
Mourrain P, Beclin C, Elmayan T, et al. Arabidopsis SGS2 and SGS3 genes are required for posttranscriptional gene silencing and natural virus resistance. Cell, 2000, 101: 533–542
Peragine A, Yoshikawa M, Wu G, et al. SGS3 and SGS2/SDE1/ RDR6 are required for juvenile development and the production of trans-acting siRNAs in Arabidopsis. Genes Dev, 2004, 18: 2368–2679
Watanabe Y, Kumakura N, Takeda A, et al. SGS3 and RDR6 interact and colocalize in cytoplasmic SGS3/RDR6-bodies. FEBS Lett, 2009, 583: 1261–1266
Doudna J A, Fukunaga R. dsRNA with 5′ overhangs contributes to endogenous and antiviral RNA silencing pathways in plants. EMBO J, 2009, 28: 545–555
Elkashef S, Ding S W. Possible new RNA intermediate in RNA silencing. Nat Chem Biol, 2009, 5: 278–279
Milner J J, Love A J, Laird J, et al. Cauliflower mosaic virus protein P6 is a suppressor of RNA silencing. J Gen Virol, 2007, 88: 3439–3444
Keller M, Haas G, Azevedo J, et al. Nuclear import of CaMV P6 is required for infection and suppression of the RNA silencing factor DRB4. EMBO J, 2008, 27: 2102–2112
Yang X L, Xie Y, Raja P, et al. Suppression of methylation-mediated transcriptional gene silencing by βC1-SAHH protein interaction during Geminivirus-Betasatellite Infection. PLoS Pathog, 2011, 7: e1002329
Brough C L, Gardiner W E, Inamdar N M, et al. DNA methylation inhibits propagation of Tomato golden mosaic-virus DNA in transfected protoplasts. Plant Mol Biol, 1992, 18: 703–712
Zhang Z H, Chen H, Huang X H, et al. BSCTV C2 attenuates the degradation of SAMDC1 to suppress DNA methylation-mediated gene silencing in Arabidopsis. Plant Cell, 2011, 23: 273–288
Bisaro D M. Silencing suppression by geminivirus proteins. Virology, 2006, 344: 158–168
Bisaro D M, Wang H, Buckley K J, et al. Adenosine kinase inhibition and suppression of RNA silencing by geminivirus AL2 and L2 proteins. J Virol, 2005, 79: 7410–7418
Bisaro D M, Wang H, Hao L H, et al. Adenosine kinase is inactivated by geminivirus AL2 and L2 proteins. Plant Cell, 2003, 15: 3020–3032
Moffatt B A, Stevens Y Y, Allen M S, et al. Adenosine kinase deficiency is associated with developmental abnormalities and reduced transmethylation. Plant Physiol, 2002, 128: 812–821
Bisaro D M, Buchmann R C, Asad S, et al. Geminivirus AL2 and L2 proteins suppress transcriptional gene silencing and cause genomewide reductions in cytosine methylation. J Virol, 2009, 83: 5005–5013
Pooggin M A, Trinks D, Rajeswaran R, et al. Suppression of RNA silencing by a geminivirus nuclear protein, AC2, correlates with Transactivation of host genes. J Virol, 2005, 79: 2517–2527
Chapman E J, Prokhnevsky A I, Gopinath K, et al. Viral RNA silencing suppressors inhibit the microRNA pathway at an intermediate step. Genes Dev, 2004, 18: 1179–1186
Voinnet O, Dunoyer P, Lecellier C H, et al. Probing the microRNA and small interfering RNA pathways with virus-encoded suppressors of RNA silencing. Plant Cell, 2004, 16: 1235–1250
Carrington J C, Kasschau K D, Xie Z X, et al. P1/HC-Pro, a viral suppressor of RNA silencing, interferes with Arabidopsis development and miRNA function. Dev Cell, 2003, 4: 205–217
Wang M B, Smith N A, Eamens A L. Viral Small Interfering RNAs target host genes to mediate disease symptoms in plants. PLoS Pathog, 2011, 7: e1002022
Melnyk C W, Molnar A, Baulcombe D C. Intercellular and systemic movement of RNA silencing signals. EMBO J, 2011, 30: 3553–3563
Molnar A, Melnyk C W, Bassett A, et al. Small silencing RNAs in plants are mobile and direct epigenetic modification in recipient cells. Science, 2010, 328: 872–875
Dunoyer P, Brosnan C A, Schott G, et al. An endogenous, systemic RNAi pathway in plants. EMBO J, 2010
Senshu H, Yamaji Y, Minato N, et al. A dual strategy for the suppression of host antiviral silencing: two distinct suppressors for viral replication and viral movement encoded by Potato Virus M. J Virol, 2011, 85: 10269–10278
Cao X S, Zhou P, Zhang X M, et al. Identification of an RNA silencing suppressor from a plant double-stranded RNA virus. J Virol, 2005, 79: 13018–13027
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at Springerlink.com
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 2.0 International License (https://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Jiang, L., Wei, C. & Li, Y. Viral suppression of RNA silencing. Sci. China Life Sci. 55, 109–118 (2012). https://doi.org/10.1007/s11427-012-4279-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-012-4279-x