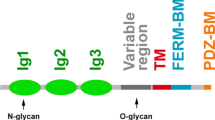
Abstract
Synapse is a highly specialized inter-cellular structure between neurons or between a neuron and its target cell that mediates cell-cell communications. Ample results indicate that synaptic adhesion molecules are critically important in modulating the complexity and specificity of the synapse. And disruption of adhesive properties of synapses may lead to neurodevelopmental or neurodegenerative diseases. In this review, we will use the Drosophila NMJ as a model system for glutamatergic synapses to discuss the structure and function of homophilic and heterophilic synaptic adhesion molecules with special focus on recent findings in neurexins and neuroligins in Drosophila.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Sudhof T C. Neuroligins and neurexins link synaptic function to cognitive disease. Nature, 2008, 455: 903–911
Adams M D, Celniker S E, Holt R A, et al. The genome sequence of Drosophila melanogaster. Science, 2000, 287: 2185–2195
Jan L Y, Jan Y N. Properties of the larval neuromuscular junction in Drosophila melanogaster. J Physiol, 1976, 262: 189–214
Keshishian H, Broadie K, Chiba A, et al. The Drosophila neuromuscular junction: a model system for studying synaptic development and function. Annu Rev Neurosci, 1996, 19: 545–575
Koh Y H, Gramates L S, Budnik V. Drosophila larval neuromuscular junction: molecular components and mechanisms underlying synaptic plasticity. Microsc Res Tech, 2000, 49: 14–25
Prokop A. Integrating bits and pieces: synapse structure and formation in Drosophila embryos. Cell Tissue Res, 1999, 297: 169–186
Richmond J E, Broadie K S. The synaptic vesicle cycle: exocytosis and endocytosis in Drosophila and C. elegans. Curr Opin Neurobiol, 2002, 12: 4997–507
Marrus S B, Portman S L, Allen M J, et al. Differential localization of glutamate receptor subunits at the Drosophila neuromuscular junction. J Neurosci, 2004, 24: 14067–1415
Schuster C M, Ultsch A, Schloss P, et al. Molecular cloning of an invertebrate glutamate receptor subunit expressed in Drosophila muscle. Science, 1991, 254: 112–114
Shishido E, Takeichi M, Nose A. Drosophila synapse formation: regulation by transmembrane protein with Leu-rich repeats, CAPRICIOUS. Science, 1998, 280: 2118–2121
Abrell S, Jackle H. Axon guidance of Drosophila SNb motoneurons depends on the cooperative action of muscular Kruppel and neuronal capricious activities. Mech Dev, 2001, 109: 3–12
Taniguchi H, Shishido E, Takeichi M, et al. Functional dissection of Drosophila capricious: its novel roles in neuronal pathfinding and selective synapse formation. J Neurobiol, 2000, 42: 104–116
Kohsaka H, Nose A. Target recognition at the tips of postsynaptic filopodia: accumulation and function of Capricious. Development, 2009, 136: 1127–1135
Nose A, Umeda T, Takeichi M. Neuromuscular target recognition by a homophilic interaction of connectin cell adhesion molecules in Drosophila. Development, 1997, 124: 1433–1441
Raghavan S, White R A. Connectin mediates adhesion in Drosophila. Neuron, 1997, 18: 873–880
Wright J W, Snyder M A, Schwinof K M, et al. A role for fasciclin II in the guidance of neuronal migration. Development, 1999, 126: 3217–3228
Grenningloh G, Rehm E J, Goodman C S. Genetic analysis of growth cone guidance in Drosophila: fasciclin II functions as a neuronal recognition molecule. Cell, 1991, 67: 45–57
Urbach R, Technau G M. Molecular markers for identified neuroblasts in the developing brain of Drosophila. Development, 2003, 130: 3621–3637
Schmucker D, Jackle H, Gaul U. Genetic analysis of the larval optic nerve projection in Drosophila. Development, 1997, 124: 937–948
Garcia-Alonso L, VanBerkum M F, Grenningloh G, et al. Fasciclin II controls proneural gene expression in Drosophila. Proc Natl Acad Sci USA, 1995, 92: 10501–10505
Vactor D V, Sink H, Fambrough D, et al. Genes that control neuromuscular specificity in Drosophila. Cell, 1993, 73: 1137–1153
Davis G W, Schuster C M, Goodman C S. Genetic analysis of the mechanisms controlling target selection: target-derived Fasciclin II regulates the pattern of synapse formation. Neuron, 1997, 19: 561–573
Lin D M, Fetter R D, Kopczynski C, et al. Genetic analysis of Fasciclin II in Drosophila: defasciculation, refasciculation, and altered fasciculation. Neuron, 1994, 13: 1055–1069
Lin D M, Goodman C S. Ectopic and increased expression of Fasciclin II alters motoneuron growth cone guidance. Neuron, 1994, 13: 507–523
Sanchez-Soriano N, Prokop A. The influence of pioneer neurons on a growing motor nerve in Drosophila requires the neural cell adhesion molecule homolog FasciclinII. J Neurosci, 2005, 25: 78–87
Kohsaka H, Takasu E, Nose A. In vivo induction of postsynaptic molecular assembly by the cell adhesion molecule Fasciclin2. J Cell Biol, 2007, 179: 1289–1300
Morimoto T, Nobechi M, Komatsu A, et al. Subunit-specific and homeostatic regulation of glutamate receptor localization by CaMKII in Drosophila neuromuscular junctions. Neuroscience, 2010, 165: 1284–1292
Beumer K, Matthies H J, Bradshaw A, et al. Integrins regulate DLG/FAS2 via a CaM kinase II-dependent pathway to mediate synapse elaboration and stabilization during postembryonic development. Development, 2002, 129: 3381–3391
Silies M, Klambt C. APC/C(Fzr/Cdh1)-dependent regulation of cell adhesion controls glial migration in the Drosophila PNS. Nat Neurosci, 2010, 13: 1357–1364
Woods D F, Hough C, Peel D, et al. Dlg protein is required for junction structure, cell polarity, and proliferation control in Drosophila epithelia. J Cell Biol, 1996, 134: 1469–1482
Chiba A, Snow P, Keshishian H, et al. Fasciclin III as a synaptic target recognition molecule in Drosophila. Nature, 1995, 374: 166–68
Alenius M, Bohm S. Identification of a novel neural cell adhesion molecule-related gene with a potential role in selective axonal projection. J Biol Chem, 1997, 272: 26083–26086
Hortsch M. Structural and functional evolution of the L1 family: are four adhesion molecules better than one? Mol Cell Neurosci, 2000, 15: 1–10
Yamamoto M, Ueda R, Takahashi K, et al. Control of axonal sprouting and dendrite branching by the Nrg-Ank complex at the neuron-glia interface. Curr Biol, 2006, 16: 1678–1683
Hall S G, Bieber A J. Mutations in the Drosophila neuroglian cell adhesion molecule affect motor neuron pathfinding and peripheral nervous system patterning. J Neurobiol, 1997, 32: 325–340
Martin V, Mrkusich E, Steinel M C, et al. The L1-type cell adhesion molecule Neuroglian is necessary for maintenance of sensory axon advance in the Drosophila embryo. Neural Dev, 2008, 3: 10
Salinas P C, Price S R. Cadherins and catenins in synapse development. Curr Opin Neurobiol, 2005, 15: 73–80
Suzuki S C, Takeichi M. Cadherins in neuronal morphogenesis and function. Dev Growth Differ, 2008, 50Suppl 1: S119–130
Yonekura S, Ting C Y, Neves G, et al. The variable transmembrane domain of Drosophila N-cadherin regulates adhesive activity. Mol Cell Biol, 2006, 26: 6598–6608
Iwai Y, Usui T, Hirano S, et al. Axon patterning requires DN-cadherin, a novel neuronal adhesion receptor, in the Drosophila embryonic CNS. Neuron, 1997, 19: 77–89
Schmucker D, Clemens J C, Shu H, et al. Drosophila Dscam is an axon guidance receptor exhibiting extraordinary molecular diversity. Cell, 2000, 101: 671–684
Soba P, Zhu S, Emoto K, et al. Drosophila sensory neurons require Dscam for dendritic self-avoidance and proper dendritic field organization. Neuron, 2007, 54: 403–416
Hattori D, Chen Y, Matthews B J, et al. Robust discrimination between self and non-self neurites requires thousands of Dscam1 isoforms. Nature, 2009, 461: 644–648
Bhat M A, Rios J C, Lu Y, et al. Axon-glia interactions and the domain organization of myelinated axons requires neurexin IV/Caspr/Paranodin. Neuron, 2001, 30: 369–383
Baumgartner S, Littleton J T, Broadie K, et al. A Drosophila neurexin is required for septate junction and blood-nerve barrier formation and function. Cell, 1996, 87: 1059–1068
Bhat M A, Izaddoost S, Lu Y, et al. Discs Lost, a novel multi-PDZ domain protein, establishes and maintains epithelial polarity. Cell, 1999, 96: 833–845
Banerjee S, Blauth K, Peters K, et al. Drosophila neurexin IV interacts with roundabout and is required for repulsive midline axon guidance. J Neurosci, 2010, 30: 5653–5667
Wheeler S R, Banerjee S, Blauth K, et al. Neurexin IV and Wrapper interactions mediate Drosophila midline glial migration and axonal ensheathment. Development, 2009, 136: 1147–1157
Zweier C, de Jong E K, Zweier M, et al. CNTNAP2 and NRXN1 are mutated in autosomal-recessive Pitt-Hopkins-like mental retardation and determine the level of a common synaptic protein in Drosophila. Am J Hum Genet, 2009, 85: 655–666
Chen K, Gracheva E O, Yu S C, et al. Neurexin in embryonic Drosophila neuromuscular junctions. PLoS One, 2010, 5: e11115
Li J, Ashley J, Budnik V, et al. Crucial role of Drosophila neurexin in proper active zone apposition to postsynaptic densities, synaptic growth, and synaptic transmission. Neuron, 2007, 55: 741–755
Zeng X, Sun M, Liu L, et al. Neurexin-1 is required for synapse formation and larvae associative learning in Drosophila. FEBS Lett, 2007, 581: 2509–2516
Sun M, Xing G, Yuan L, et al. Neuroligin 2 is required for synapse development and function at the Drosophila neuromuscular junction. J Neurosci, 2011, 31: 687–699
Banovic D, Khorramshahi O, Owald D, et al. Drosophila neuroligin 1 promotes growth and postsynaptic differentiation at glutamatergic neuromuscular junctions. Neuron, 2010, 66: 724–738
Dean C. Neuroligins and neurexins: linking cell adhesion, synapse formation and cognitive function. Trends Neurosci, 2006, 29: 21–29. Epub 2005 Dec 2007
Sun M, Liu L, Zeng X, et al. Genetic interaction between Neurexin and CAKI/CMG is important for synaptic function in Drosophila neuromuscular junction. Neurosci Res, 2009, 64: 362–371
Kittel R J, Wichmann C, Rasse T M, et al. Bruchpilot promotes active zone assembly, Ca2+ channel clustering, and vesicle release. Science, 2006, 312: 1051–1054
Wagh D A, Rasse T M, Asan E, et al. Bruchpilot, a protein with homology to ELKS/CAST, is required for structural integrity and function of synaptic active zones in Drosophila. Neuron, 2006, 49: 833–844
Owald D, Sigrist S J. Assembling the presynaptic active zone. Curr Opin Neurobiol, 2009, 19: 311–318
Owald D, Fouquet W, Schmidt M, et al. A Syd-1 homologue regulates pre- and postsynaptic maturation in Drosophila. J Cell Biol, 2010, 188: 565–579
de Wit J, Sylwestrak E, O’sullivan M L, et al. LRRTM2 interacts with Neurexin1 and regulates excitatory synapse formation. Neuron, 2009, 64: 799–806
Ko J, Fuccillo M V, Malenka R C, et al. LRRTM2 functions as a neurexin ligand in promoting excitatory synapse formation. Neuron, 2009, 64: 791–798
Author information
Authors and Affiliations
Corresponding authors
Additional information
This article is published with open access at Springerlink.com
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 2.0 International License (https://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Sun, M., Xie, W. Cell adhesion molecules in Drosophila synapse development and function. Sci. China Life Sci. 55, 20–26 (2012). https://doi.org/10.1007/s11427-012-4273-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-012-4273-3