Abstract
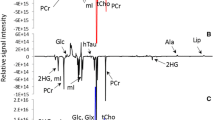
Clinical data have shown that survival rates vary considerably among brain tumor patients, according to the type and grade of the tumor. Metabolite profiles of intact tumor tissues measured with high-resolution magic-angle spinning proton nuclear magnetic resonance spectroscopy (HRMAS 1H NMRS) can provide important information on tumor biology and metabolism. These metabolic fingerprints can then be used for tumor classification and grading, with great potential value for tumor diagnosis. We studied the metabolic characteristics of 30 neuroepithelial tumor biopsies, including two astrocytomas (grade I), 12 astrocytomas (grade II), eight anaplastic astrocytomas (grade III), three glioblastomas (grade IV) and five medulloblastomas (grade IV) from 30 patients using HRMAS 1H NMRS. The results were correlated with pathological features using multivariate data analysis, including principal component analysis (PCA). There were significant differences in the levels of N-acetyl-aspartate (NAA), creatine, myo-inositol, glycine and lactate between tumors of different grades (P<0.05). There were also significant differences in the ratios of NAA/creatine, lactate/creatine, myo-inositol/creatine, glycine/creatine, scyllo-inositol/creatine and alanine/creatine (P<0.05). A soft independent modeling of class analogy model produced a predictive accuracy of 87% for high-grade (grade III–IV) brain tumors with a sensitivity of 87% and a specificity of 93%. HRMAS 1H NMR spectroscopy in conjunction with pattern recognition thus provides a potentially useful tool for the rapid and accurate classification of human brain tumor grades.
Article PDF
Similar content being viewed by others
References
Mertens A C, Yasui Y, Neglia J P, et al. Late mortality experience in five-year survivors of childhood and adolescent cancer: the childhood cancer survivor study. J Clin Oncol, 2001, 19: 3163–3172 11432882, 1:STN:280:DC%2BD3Mzns1Oltg%3D%3D
Mainio A, Hakko H, Timonen M, et al. Depression in relation to survival among neurosurgical patients with a primary brain tumor: a 5-year follow-up study. Neurosurgery, 2005, 56: 1234–1241 15918939, 10.1227/01.NEU.0000159648.44507.7F
Sant M, Van der Sanden G, Capocaccia R. Survival rates for primary malignant brain tumours in Europe. EUROCARE Working Group. Eur J Cancer, 1998, 34: 2241–224 10.1016/S0959-8049(98)00336-0, 1:STN:280:DyaK1M7mvFCntQ%3D%3D
Barnholtz-Sloan J S, Sloan A E, Schwartz A G. Relative survival rates and patterns of diagnosis analyzed by time period for individuals with primary malignant brain tumor, 1973–1997. J Neurosurgery, 2003, 99: 458–466 10.3171/jns.2003.99.3.0458
Iwadate Y, Sakaida T, Hiwasa T, et al. Molecular classification and survival prediction in human gliomas based on proteome analysis. Cancer Res, 2004, 64: 2496–250 15059904, 10.1158/0008-5472.CAN-03-1254, 1:CAS:528:DC%2BD2cXis1yjtbY%3D
Engelhard H H, Stelea A, Cochran E J. Oligodendroglioma: pathology and molecular biology. Surg Neurol, 2002, 58: 111–117 12453646, 10.1016/S0090-3019(02)00751-6
Rickert C H, Paulus W. Prognosis-related histomorphological and immunohistochemical markers in central nervous system tumors of childhood and adolescence. Acta Neuropathol, 2005, 109: 69–92 15647946, 10.1007/s00401-004-0959-3, 1:CAS:528:DC%2BD2MXhsVaksrc%3D
Zarbo R J, Meier F A, Raab S S. Error detection in anatomic pathology. Arch Pathol Lab Med, 2005, 129: 1237–1245 16196511
Salpietro F M, Alafaci C, Lucerna S, et al. Peritumoral edema in meningiomas microsurgical observations of different brain tumor interfaces related to computed tomography. Neurosurgery, 1994, 35: 638–641 7808606, 10.1227/00006123-199410000-00009, 1:STN:280:DyaK2M7gslCrsQ%3D%3D
Simonetti A W, Melssen W J, Szabo de Edelenyi F, et al. Combination of feature-reduced MR spectroscopic and MR imaging data for improved brain tumor classification. NMR Biomed, 2005, 18: 34–43 15657908, 10.1002/nbm.919
Devos A, Simonetti A W, Van der Graaf M, et al. The use of multivariate MR imaging intensities versus metabolic data from MR spectroscopic imaging for brain tumour classification. J Magn Reson, 2005, 173: 218–228 15780914, 10.1016/j.jmr.2004.12.007, 1:CAS:528:DC%2BD2MXisVCmtb0%3D
Henze M, Mohammed A, Schlemmer H P, et al. PET and SPECT for detection of tumor progression in irradiated low-grade astrocytoma: a receiver-operating-characteristic analysis. J Nucl Med, 2004, 45: 579–586 15073253
Roelcke U, Leenders K L. PET in neuro-oncology. J Cancer Res Clin Oncol, 2001, 127: 2–8 11206266, 10.1007/s004320000158, 1:STN:280:DC%2BD3M7is1Clug%3D%3D
Tong Z, Yamaki T, Harada K, et al. In vivo quantification of the metabolites in normal brain and brain tumors by proton MR spectroscopy using water as an internal standard. Magn Reson Imaging, 2004, 22: 1017–1024 15288143, 10.1016/j.mri.2004.02.007, 1:CAS:528:DC%2BD2cXmt1Kls7c%3D
Fayed N, Morales H, Modrego P J, et al. Contrast/noise ratio on conventional MRI and choline/creatine ratio on proton MRI spectroscopy accurately discriminate low-grade from high-grade cerebral gliomas. Acad Radiol, 2006, 13: 728–737 16679275, 10.1016/j.acra.2006.01.047
Majos C, Alonso J, Aguilera C, et al. Proton magnetic resonance spectroscopy (1H MRS) of human brain tumours: assessment of differences between tumour types and its applicability in brain tumour categorization. Eur Radiol, 2003, 13: 582–591 12594562
Majos C, Julia-Sape M, Alonso J, et al. Brain tumor classification by proton MR spectroscopy: comparison of diagnostic accuracy at short and long TE. AJNR Am J Neuroradiol, 2004, 25: 1696–1704 15569733
Maxwell R J, Martinez-Perez I, Cerdan S, et al. Pattern recognition analysis of 1H NMR spectra from perchloric acid extracts of human brain tumor biopsies. Magn Reson Med, 1998, 39: 869–877 9621910, 10.1002/mrm.1910390604, 1:STN:280:DyaK1c3otlahuw%3D%3D
Lehnhardt F G, Bock C, Rohn G, et al. Metabolic differences between primary and recurrent human brain tumors: a 1H NMR spectroscopic investigation. NMR Biomed, 2005, 18: 371–382 15959923, 10.1002/nbm.968, 1:CAS:528:DC%2BD2MXhtF2nsbbN
Tugnoli V T M, Bertoluzza A, Barbarella G, et al. In vitro magnetic resonance spectroscopy of health and neoplastic brain tissues. J Mol Struct, 1999, 482: 365–369 10.1016/S0022-2860(98)00679-6
Peeling J, Sutherland G. High-resolution 1H NMR spectroscopy studies of extracts of human cerebral neoplasms. Magn Reson Med, 1992, 24: 123–136 1556919, 10.1002/mrm.1910240113, 1:STN:280:DyaK383ht1Whtw%3D%3D
Wilson M, Davies N P, Grundy R G, et al. A quantitative comparison of metabolite signals as detected by in vivo MRS with ex vivo1H HR-MAS for childhood brain tumours. NMR Biomed, 2009, 22: 213–219 19067434, 10.1002/nbm.1306, 1:CAS:528:DC%2BD1MXktVygurs%3D
Yang Y, Li C, Nie X, et al. Metabonomic studies of human hepatocellular carcinoma using high-resolution magic-angle spinning 1H NMR spectroscopy in conjunction with multivariate data analysis. J Proteome Res, 2007, 6: 2605–2614 17564425, 10.1021/pr070063h, 1:CAS:528:DC%2BD2sXmtlOnurk%3D
Righi V, Roda J M, Paz J, et al.1H HR-MAS and genomic analysis of human tumor biopsies discriminate between high and low grade astrocytomas. NMR Biomed, 2009, 22: 629–637 19322812, 10.1002/nbm.1377, 1:CAS:528:DC%2BD1MXhtVSnurrE
Chen W X, Lou H Y, Zhang H P, et al. Metabonomic characterization of the low-grade human astrocytomas and meningiomas using magic-angle spinning 1H Nuclear magnetic resonance spectroscopy and principal component analysis. Prog Biochem Biophys, 2008, 35: 1142–1153 1:CAS:528:DC%2BD1cXhsFSmsrvK
Waters N J, Garrod S, Farrant R D, et al. High-resolution magic angle spinning 1H NMR spectroscopy of intact liver and kidney: optimization of sample preparation procedures and biochemical stability of tissue during spectral acquisition. Anal Biochem, 2000, 282: 16–23 10860494, 10.1006/abio.2000.4574, 1:CAS:528:DC%2BD3cXktVGrsLg%3D
Wang Y, Holmes E, Nicholson J K, et al. Metabonomic investigations in mice infected with Schistosoma mansoni: an approach for biomarker identification. Proc Natl Acad Sci USA, 2004, 101: 12676–12681 15314235, 10.1073/pnas.0404878101, 1:CAS:528:DC%2BD2cXnsVent7w%3D
Cheng L L, Burns M A, Taylor J L, et al. Metabolic characterization of human prostate cancer with tissue magnetic resonance spectroscopy. Cancer Res, 2005, 65: 3030–3034 15833828, 1:CAS:528:DC%2BD2MXjt1yksrc%3D
Nicholls A W, Mortishire-Smith R J. Temperature calibration of a high-resolution magic-angle spinning NMR probe for analysis of tissue samples. Magn Reson Chem, 2001, 39: 773–776 10.1002/mrc.924, 1:CAS:528:DC%2BD3MXptVKntL4%3D
Wang Y, Bollard M E, Keun H, et al. Spectral editing and pattern recognition methods applied to high-resolution magic-angle spinning 1H nuclear magnetic resonance spectroscopy of liver tissues. Anal Biochem, 2003, 323: 26–32 14622955, 10.1016/j.ab.2003.07.026, 1:CAS:528:DC%2BD3sXovVWgtbc%3D
Eriksson L J E, Kettanah-Wold N. Introduction to Multi and Megavariate Data Analysis Using Projection Methods (PCA and PLS-DA) 1992–2002. Sweden: Umetrics AB, 2002
Brindle J T, Antti H, Holmes E, et al. Rapid and noninvasive diagnosis of the presence and severity of coronary heart disease using 1H-NMR-based metabonomics. Nat Med, 2002, 8: 1439–1444 12447357, 10.1038/nm1202-802, 1:CAS:528:DC%2BD38XptVSgtrw%3D
Wold S, Antti H, Lindgren F, et al. Orthogonal signal correction of near-infrared spectra. Chemometr Intell Lab, 1998, 44: 175–185 10.1016/S0169-7439(98)00109-9, 1:CAS:528:DyaK1cXnt1SqsbY%3D
Howe F A, Barton S J, Cudlip S A, et al. Metabolic profiles of human brain tumors using quantitative in vivo1H magnetic resonance spectroscopy. Magn Reson Med, 2003, 49: 223–232 12541241, 10.1002/mrm.10367, 1:CAS:528:DC%2BD3sXhsV2it7o%3D
Govindaraju V, Young K, Maudsley A A. Proton NMR chemical shifts and coupling constants for brain metabolites. NMR Biomed, 2000, 13: 129–153 10861994, 10.1002/1099-1492(200005)13:3<129::AID-NBM619>3.0.CO;2-V, 1:CAS:528:DC%2BD3cXksFSgtL8%3D
Kinoshita Y, Yokota A. Absolute concentrations of metabolites in human brain tumors using in vitro proton magnetic resonance spectroscopy. NMR Biomed, 1997, 10: 2–12 9251109, 10.1002/(SICI)1099-1492(199701)10:1<2::AID-NBM442>3.0.CO;2-N, 1:CAS:528:DyaK2sXltl2is74%3D
Cheng L L, Anthony D C, Comite A R, et al. Quantification of microheterogeneity in glioblastoma multiforme with ex vivo high-resolution magic-angle spinning (HRMAS) proton magnetic resonance spectroscopy. Neuro-oncology, 2000, 2: 87–95 11303625, 1:STN:280:DC%2BD3Mzgs1ekug%3D%3D
Meyerand M E, Pipas J M, Mamourian A, et al. Classification of biopsy-confirmed brain tumors using single-voxel MR spectroscopy. AJNR Am J Neuroradiol, 1999, 20: 117–123 9974066, 1:STN:280:DyaK1M7jvFKntA%3D%3D
Koljenovic S, Choo-Smith L P, Bakker Schut T C, et al. Discriminating vital tumor from necrotic tissue in human glioblastoma tissue samples by Raman spectroscopy. Lab Invest, 2002, 82: 1265–1277 12379761, 1:CAS:528:DC%2BD38Xns1Cls78%3D
Fayed N, Davila J, Medrano J, et al. Malignancy assessment of brain tumours with magnetic resonance spectroscopy and dynamic susceptibility contrast MRI. Eur J Radiol, 2008, 67: 427–433 18442889, 10.1016/j.ejrad.2008.02.039
Roda J M, Pascual J M, Carceller F, et al. Nonhistological diagnosis of human cerebral tumors by 1H magnetic resonance spectroscopy and amino acid analysis. Clin Cancer Res, 2000, 6: 3983–3993 11051247, 1:CAS:528:DC%2BD3cXotVykuro%3D
Lehnhardt F G, Rohn G, Ernestus R I, et al.1H- and 31P-MR spectroscopy of primary and recurrent human brain tumors in vitro: malignancy-characteristic profiles of water soluble and lipophilic spectral components. NMR Biomed, 2001, 14: 307–317 11477651, 10.1002/nbm.708, 1:CAS:528:DC%2BD3MXmvVahsbw%3D
Cheng L L, Chang I W, Louis D N, et al. Correlation of high-resolution magic angle spinning proton magnetic resonance spectroscopy with histopathology of intact human brain tumor specimens. Cancer Res, 1998, 58: 1825–1832 9581820, 1:CAS:528:DyaK1cXislensbY%3D
Wilson M, Davies N P, Brundler M A, et al. High resolution magic angle spinning 1H NMR of childhood brain and nervous system tumours. Mol Cancer, 2009, 8: 6–16 19208232, 10.1186/1476-4598-8-6
Lukas L, Devos A, Suykens J A, et al. Brain tumor classification based on long echo proton MRS signals. Artif Intell Med, 2004, 31: 73–89 15182848, 10.1016/j.artmed.2004.01.001, 1:STN:280:DC%2BD2c3ot1Kjtw%3D%3D
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at Springerlink.com
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Chen, W., Lou, H., Zhang, H. et al. Grade classification of neuroepithelial tumors using high-resolution magic-angle spinning proton nuclear magnetic resonance spectroscopy and pattern recognition. Sci. China Life Sci. 54, 606–616 (2011). https://doi.org/10.1007/s11427-011-4193-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-011-4193-7