Abstract
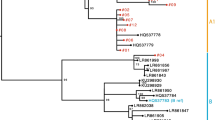
Human papillomavirus 58 (HPV58) is one type of HPV with high risk of causing cervical cancer. Unusually high prevalence of HPV58 has been reported in Asia, Africa and some other areas. However, due to the scattered distribution of global data, in addition to the lack of data of some HPV58 high-incidence nations and regions, like Mainland China, a comprehensive analysis of the global geographical distribution of HPV58 remains blank so far. In this study, HPV58 from the human cervical cancer tissue was detected in Mainland China, and 14 new HPV58-E6/L1 gene sequences were obtained. Moreover, phylogeographic analysis has been conducted combining the HPV58 sequences that have been deposited in GenBank since 1985. The study result shows that the sequences detected from the Shanghai, Jiangsu and Sichuan areas are homologous with those found in the past from Hong Kong and Xi’an, China, as well as Japan and other Southeast Asian areas. Furthermore, Western Africa is considered to be the “root” source of the HPV58 variant, while Mainland China and Southeast Asia are “transit points” and the new sources of HPV58 after receiving the isolates from the “root” source; like HPV16 and HPV18, the HPV58 might also be one of the major HPV types associated with the development and spread of cervical cancer.
Similar content being viewed by others
References
Lombard I, Vincent-Salomon A, Validier P, et al. Human papillomavirus genotype as a major determinant of the course of cervical. J Clin Oncol, 1998, 16: 2613–2619 9704710, 1:STN:280:DyaK1czmvVCjug%3D%3D
Mclachlin C M. Human papillomavirus in cervical neoplasia: Role, risk factors and implications. Clin Lab Med, 2000, 20: 257–270 10863640, 1:STN:280:DC%2BD3cvjsFCqtQ%3D%3D
Franco E L, Duarte-Franco E, Ferenczy A. Cervical cancer: Epidemiology, prevention and the role of human papillomavirus infection. CMAJ, 2001, 164: 1017–1025 11314432, 1:STN:280:DC%2BD3M3itVymtQ%3D%3D
Calleja-Macias I E, Villa L L, Prado J C, et al. Worldwide genomic diversity of the high-risk human papillomavirus types 31, 35, 52, and 58, four close relatives of human papillomavirus type 16. J Virol, 2005, 79: 13630–13640 10.1128/JVI.79.21.13630-13640.2005, 16227283, 1:CAS:528:DC%2BD2MXhtFKjtr%2FE
Clifford G M, Smith J S, Plummer M, et al. Human papillomavirus types in invasive cervical cancer worldwide: A meta-analysis. Br J Cancer, 2003, 88: 63–73 10.1038/sj.bjc.6600688, 12556961, 1:STN:280:DC%2BD3s%2Fks12qtw%3D%3D
Chan P K, Lam C W, Cheung TH, et al. Association of human papillomavirus type 58 variant with the risk of cervical cancer. J Natl Cancer Inst, 2002, 94: 1249–1301 12189229, 1:CAS:528:DC%2BD38XntVyjsLw%3D
Lin Q Q, Yu S Z, Qu W, et al. Human papillomavirus types 52 and 58. Int J Cancer, 1998, 75: 484–485 10.1002/(SICI)1097-0215(19980130)75:3<484::AID-IJC25>3.0.CO;2-7, 9455813, 1:STN:280:DyaK1c7hsFenug%3D%3D
Wu Y, Chen Y, Li L, et al. Associations of high-risk HPV types and viral load with cervical cancer in China. J Clin Virol, 2006, 35: 264–272 10.1016/j.jcv.2005.07.011, 16183329, 1:CAS:528:DC%2BD28XhtFShsL4%3D
Huang S, Afonina I, Miller B A, et al. Human papillomavirus types 52 and 58 are prevalent in cervical cancer from Chinese women. Int J Cancer, 1997, 70: 408–411 10.1002/(SICI)1097-0215(19970207)70:4<408::AID-IJC6>3.0.CO;2-#, 9033647, 1:STN:280:DyaK2s7osF2ltA%3D%3D
Liu J H, Huang X, Liao G W, et al. The comparative study of HPV infection in cervical cancer patients and other risk factors between China and Australia. Chin J Med, 2003, 83: 748–752
Lai H C, Sun C A, Yu M H, et al. Favorable clinical outcome of cervical cancers infected with human papilloma virus type 58 and related types. Int J Cancer, 1999, 84: 553–557 10.1002/(SICI)1097-0215(19991222)84:6<553::AID-IJC2>3.0.CO;2-4, 10567897, 1:STN:280:DC%2BD3c%2Fjs1Crtg%3D%3D
Lai C H, Huang H J, Hsueh S, et al. Human papillomavirus genotype in cervical cancer: A population-based study. Int J Cancer, 2007, 120: 1999–2006 10.1002/ijc.22538, 17266033, 1:CAS:528:DC%2BD2sXjsVCqsbk%3D
Chan P K, Li W H, Chan M Y, et al. High prevalence of human papillomavirus type 58 in Chinese women with cervical cancer and precancerous lesions. J Med Virol, 1999, 59: 232–238 10.1002/(SICI)1096-9071(199910)59:2<232::AID-JMV18>3.0.CO;2-7, 10459162, 1:STN:280:DyaK1MzovFyjtg%3D%3D
Wallace R G, Hodac H, Lathrop R H, et al. Lathrop, et al. A statistical phylogeography of influenza A H5N1. Proc Natl Acad Sci USA, 2007, 104: 4473–4478 10.1073/pnas.0700435104, 17360548, 1:CAS:528:DC%2BD2sXjsFKnsbs%3D
Stewart A C, Eriksson A M, Manos M M, et al. Intratype variation in 12 human papillomavirus types: A worldwide perspective. J Virol, 1996, 70: 3127–3136 8627792, 1:CAS:528:DyaK28Xit1Ohu7g%3D
Cerqueira D M, Camara G N, da Cruz M R, et al. Variants of human papillomavirus types 53, 58 and 66 identified in Central Brazil. Virus Genes, 2003, 26: 83–87 10.1023/A:1022386323921, 12680696, 1:CAS:528:DC%2BD3sXhtFGjtLw%3D
Qu W, Jiang G, Cruz Y, et al. PCR detection of human papillomavirus: Comparison between MY09/MY11 and GP5+/GP6+ primer systems. J Clin Microbiol, 1997, 35: 1304–1310 9163434, 1:CAS:528:DyaK2sXjslOqurs%3D
Cheah P L, Looi L M. Biology and pathological associations of the human papillomaviruses: A review. Malays J Pathol, 1998, 20: 1–10 10879257, 1:STN:280:DC%2BD3czjslChsw%3D%3D
Sichero L, Villa L L. Epidemiological and functional implications of molecular variants of human papillomavirus. Braz J Med Biol Res, 2006, 39: 707–717 10.1590/S0100-879X2006000600002, 16751975, 1:CAS:528:DC%2BD28XntV2gtr0%3D
Yamada T, Wheeler C M, Halpern A L, et al. Human papillomavirus type 16 variant lineages in United States populations characterized by nucleotide sequence analysis of the E6, L2, and L1 coding segments. J Virol, 1995, 69: 7743–7795 7494284, 1:CAS:528:DyaK2MXptlOkur8%3D
Chen Z, Terai M, Fu L, et al. Diversifying selection in human papillomavirus type 16 lineages based on complete genome analyses. J Virol, 2005, 79: 7014–7036 10.1128/JVI.79.11.7014-7023.2005, 15890941, 1:CAS:528:DC%2BD2MXksFKgs7Y%3D
Ho L, Chan S Y, Burk R D, et al. The genetic drift of human papillomavirus type 16 is a means of reconstructing prehistoric viral spread and the movement of ancient human populations. J Virol, 1993, 67: 6413–6435 8411343, 1:CAS:528:DyaK2cXktlGk
Ho L, Chan S Y, Chow V, et al. Sequence variants of human papillomavirus type 16 in clinical samples permit verification and extension of epidemiological studies and construction of a phylogenetic tree. J Clin Microbiol, 1991, 29: 1765–1836 1663516, 1:STN:280:DyaK387ivFGluw%3D%3D
Kirii Y, Iwamoto S, Matsukura T. Human papillomavirus type 58 DNA sequence. Virology, 1991, 185: 424–430 10.1016/0042-6822(91)90791-9, 1656594, 1:CAS:528:DyaK38XmtVKjtrs%3D
Chan P K, Chang A R, Cheung J L, et al. Determinants of cervical human papillomavirus infection: Differences between high- and low-oncogenic risk types. J Infect Dis, 2002, 185: 28–35 10.1086/338010, 11756978
Chan P K, Chang A R, Tam W H, et al. Prevalence and genotype distribution of cervical human papillomavirus infection: Comparison between pregnant women and non-pregnant controls. J Med Virol, 2002, 67: 583–590 10.1002/jmv.10142, 12116008
Xi L F, Touré P, Critchlow C W, et al. Prevalence of specific types of human papillomavirus and cervical squamous intraepithelial lesions in consecutive, previously unscreened, West-African women over 35 years of age. Int J Cancer, 2003, 103: 803–811 10.1002/ijc.10876, 12516102, 1:CAS:528:DC%2BD3sXntVaruw%3D%3D
Xin C Y, Matsumoto K, Yoshikawa H, et al. Analysis of E6 variants of human papillomavirus type 33, 52 and 58 in Japanese women with cervical intraepithelial neoplasia/cervical cancer in relation to their oncogenic potential. Cancer Lett, 2001, 170: 19–24 10.1016/S0304-3835(01)00570-5, 11448530, 1:CAS:528:DC%2BD3MXltVejs78%3D
Cho N H, An H J, Jeong J K, et al. Genotyping of 22 human papillomavirus types by DNA chip in Korean women: comparison with cytologic diagnosis. Am J Obstet Gynecol, 2003, 188: 56–62 10.1067/mob.2003.120, 12548196, 1:CAS:528:DC%2BD3sXhsV2ns74%3D
Bernard H U, Calleja-Macias I E, Dunn S T. Genome variation of human papillomavirus types: Phylogenetic and medical implications. Int J Cancer, 2006, 118: 1071–1076 10.1002/ijc.21655, 16331617, 1:CAS:528:DC%2BD28XhsFeksL0%3D
DeFilippis V R, Ayala F J, Villarreal L P. Evidence of diversifying selection in human papillomavirus type 16 E6 but not E7 oncogenes. J Mol Evol, 2002, 55: 491–499 10.1007/s00239-002-2344-y, 12355268, 1:CAS:528:DC%2BD38XnsVersLw%3D
Villa L L, Costa R L, Petta C A, et al. Prophylactic quadrivalent human papillomavirus (types 6, 11 16 and 18) L1 virus-like particle vaccine in young women a randomized double-blind placebocontrolled multicentre phase II efficacy trial. Lancet Oncol, 2005, 6: 271–278 10.1016/S1470-2045(05)70101-7, 15863374
Harper D M, Franco E L, Wheeler C, et al. Efficacy of a bivalent L1 virus-like particle vaccine in prevention of infection with human papillomavirus types 16 and 18 in young women: A randomized controlled trial. Lancet, 2004, 364: 1757–1765 10.1016/S0140-6736(04)17398-4, 15541448, 1:CAS:528:DC%2BD2cXpslGju78%3D
Author information
Authors and Affiliations
Corresponding authors
Additional information
Supported by the National Science and Technology Pillar Program of China (Grant No. 2007BAI24B01) and the National High Technology Research and Development Program of China (Grant No. 2006AA02Z342) the Shanghai Leading Academic Discipline Project (B111) and Shanghai Science and Technology Committee (Grant No. 07XD14025).
Rights and permissions
About this article
Cite this article
Li, Y., Li, Z., He, Y. et al. Phylogeographic analysis of human papillomavirus 58. SCI CHINA SER C 52, 1164–1172 (2009). https://doi.org/10.1007/s11427-009-0149-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-009-0149-6