Abstract
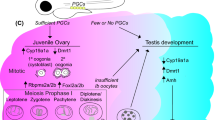
Dmc1 (disrupted meiotic cDNA) is a functionally specific gene, which was firstly discovered in yeast and then found to encode a protein required for homologous chromosome synapsis during the process of meiosis. In this investigation, we cloned the partial cDNAs of Dmc1 of diploid red crucian carp, Japanese crucian carp, common carp, triploid crucian carp and allotetraploid hybrids by using a pair of degenerate primers based on the conservative sequence of amino acids of the DMC1 protein in yeast, mouse and human. The full length cDNAs were then obtained by rapid amplification of cDNA ends (RACE). Our data showed that the full length cDNAs of Dmc1 in the three diploid fishes are all 1375 bp long, while it is 1383 bp long in triploids and 1379 bp long in allotetraploids. And despite of the variation in length, all the cDNAs encode a protein of 342 amino acids. A high homology of 97.3% of the DMC1 protein can be drawn by comparing the amino acid sequences in the three diploids, which is also of 86%, 86% and 95% similarity to human, mouse and zebrafish, respectively. A comparative study of the expression pattern of Dmc1 was carried out by RT-PCR using specific primers against the same sequences of coding regions in different ploidy cyprinid fishes, from which it was showed that Dmc1 was expressed only in gonads of these five kinds of fishes. The expression pattern of Dmc1 in both ovaries and testes from different ploidy fishes within breeding season was also studied by Real-time PCR, and the results showed that the expression of this gene was greatly different among the three different ploidy fishes, which was the highest of triploid and lowest of allotetraploids. The histological sections data showed matured gonads of both diploid red crucian carp and allotetraploids in breeding season, although the latter demonstrated a higher maturation, and no gonadal maturation could be observed in triploids. In conclusion, we suggest that Dmc1 is specifically expressed in the period of meiosis in all the ploidy cyprinid fishes and directly related with the development of gonad in a manner of ploidy-independent way. And further, the high expression of Dmc1 in female triploids might be associated with abnormal meiosis and sterility.
Similar content being viewed by others
References
Sato S, Seki N, Hotta Y, et al. Expression profiles of a human gene identified as a structural homologue of meiosis-specific recA-like genes. DNA Res, 1995, 2(4): 183–186
Matsuda Y, Habu T, Hori T, et al. Chromosome mapping of the mouse homologue of DMC1, the yeast meiosis-specific homologous recombination gene. Chromosome Res, 1996, 4(3): 249–250
Bishop D k, Park D, Xu L, et al. DMC1: a meiosis-specific yeast homolog of E. coli recA required for recombination, synaptonemal complex formation, and cell cycle progression. Cell, 1992, 69(3): 439–456
Shinohara A, Ogawa H, Ogawa T. Rad51 protein involved in repair and recombination in S. cerevisiae is a recA-like protein. Cell, 1992, 69(3): 457–470
Hollingsworth N M, Byers B. The HOP1 gene encodes a meiosis-specific component of yeast chromosomes. Cell, 1990, 61(1): 75–84
Bishop D k. RecA homologs Dmc1 and Rad51 interact to form multiple nuclear complexes prior to meiotic chromosome synapsis. Cell, 1994, 79(6): 1081–1092
Habu T, Taki T, West A, et al. The mouse and human homologs of DMC1, the yeast meiosis-specific homologous recombination gene, have a common unique form of exon-skipped transcript in meiosis. Nucleic Acid Res, 1996, 24(3): 470–477
Huo J. Progress of proteins related with homologous recombination in mammalians. Foreign Med Sci—Genetics (in Chinese), 2001, 24(1): 24–27
Pittman D L, Cobb J, Schimenti K J, et al. Meiotic prophase arrest with failure of chromosome synapses in mice deficient for Dmc1, a germline-specific RecA homolog. Mol Cell, 1998, 1(5): 697–705
Yoshida K, Kondoh G, Matsuda Y, et al. The mouse RecA-like gene Dmc1 is required for homologous chromosome synapsis during meiosis. Mol Cell, 1998, 1(5): 707–718
Kajiura-Kobayashi H, Kobayashi T, Nagahama Y. Cloning of cDNAs and the differential expression of A-type cyclins and Dmc1 during spermatogenesis in the Japanese eel, a teleost fish. Dev Dyn, 2005, 232(2): 1115–1123
Ding Z J, Wang T, Zhong K. Cloning of OsDmc1 as a meiosis-related gene in rice. Chin Sci Bull, 2001, 46(10): 838–843
Liu Y, Zhou G. Cytological study on gonadal development of F1 hybrids produced by crossing Carassius auratus (♀) with Cyprinus carpio (♂). Acta Hydrobiol Sin (in Chinese), 1986, 10(2): 101–108
Liu S J, Liu Y, Zhou G J, et al. The formation of tetraploid stocks of red crucian carp × common carp hybrids as an effect of interspecific hybridization. Aquaculture, 2001, 192: 171–186
Babiak I, Dobosz S, Goryczko K, et al. Androgenesis in rainbow trout using cryopreserved spermatozoa: The effect of processing and biological factors. Theriogenology, 2002, 57(4): 1229–1249
Liu S J, Hu F, Zhou G J, et al. Gonadal structure of triploid crucian carp produced by crossing allotetraploid hybrids of Carassius auratus red var.(♀) × Cyprinus carpio L.(♂) with Japanese crucian carp (Carassius auratus cuvieri T. et S). Acta Hydrobiol Sin (in Chinese), 2002, 24(4): 301–306
Li J Z, Zhang X J, Liu S J, et al. Studies on the gonadal development in allotetraploid hybrids of Carassius auratus red var. (♀) × Cyprinus carpio L.(♂). Acta Hydrobiol Sin (in Chinese), 2002, 26(2): 116–122
Sun Y D, Liu S J, Zhang C, et al. The chromosome number and gonadal structure of F9–F11 allotetraploid crucian carp. Acta Genet Sin (in Chinese), 2003, 30(5): 414–418
Liu S J, Sun Y D, Zhang C, et al. Triploid crucian carp — Allotetraploid hybrids (♀) × goldfish (♂). Acta Genet Sin (in Chinese), 2004, 31(1): 31–38
Feng H, Zeng Z Q, Liu S J, et al. Studies of F1 of transgenic allotetraploid hybrids of Carassius auratus red var.(♀) × Cyprinus carpio L.(♂). Acta Genet Sin (in Chinese), 2002, 29(5): 434–437
Guo X H, Liu S J, Liu Y. Evidence for recombination of mitochondrial DNA in triploid crucian carp. Genetics, 2006, 172: 1475–1479
Long Y, Liu S J, Huang W R, et al. Comparative studies on histological and ultra-structure of the pituitary of different ploidy level fishes. Sci China Ser C-Life Sciences, 2006, 49(5): 446–453
Liu Q, Wang Y Q, Liu S J, et al. Comparative studies on blood and blood cells in different ploidy fishes. Prog Nat Sci (in Chinese), 2004, 14(10): 1111–1117
Zhang C, He X X, Liu S J, et al. The Chromosome pairing in meiosis I in allotetraploid hybrids and allotriploid crucian carp. Acta Zool Sin (in Chinese), 2005, 51(1): 89–84
Livark K J, Schmittgen T D. Analysis of relative gene expression data using real-time quantitative PCR and the 2[-Delta Delta C (T)] method. Methods, 2001, 25(4): 402–408
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by the National Natural Science Funds for Distinguished Young Scholar (Grant No. 30725028), National Natural Science Foundation of China (Grant Nos. 30330480 and 30571444), the State Key Basic Research Project (973 Project) of China (Grant No. 2007CB109206), the Doctoral Station of University of the Education Ministry of China (Grant No. 200405422001) and the Program for Changjiang Scholars and Innovative Research Teams in Universities of China (Grant No. IRT0445)
Rights and permissions
About this article
Cite this article
Tao, M., Liu, S., Long, Y. et al. The cloning of Dmc1 cDNAs and a comparative study of its expression in different ploidy cyprinid fishes. Sci. China Ser. C-Life Sci. 51, 38–46 (2008). https://doi.org/10.1007/s11427-008-0004-1
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11427-008-0004-1