Abstract
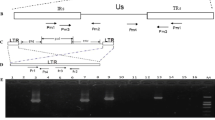
The genomic DNA extracted from chicken embryo fibroblast (CEF) infected with a Chinese field isolate HA9901 of reticuloendotheliosis virus (REV) was used as the template to amplify the REV proviral genomic cDNA by PCR with 6 pairs of primers according to published sequences. Six overlapping fragments were amplified, cloned into the TA vector and sequenced, including a fragment which was amplified from the circular proviral cDNA and covering both 5′-and 3′-ends. The complete sequence of the whole genome was established and analyzed with a DNAstar software. Comparisons of the sequence with two other strains demonstrated that the genomes of REV were relatively conservative, the homogenecity for all genes or LTR fragments of the 3 strains was over 92%, no matter whether they were isolated from different species and regions in different years. But, the homology of Chinese strain HA9901 to a fowl pox virus-associated strain from Chickens was higher than that to strain SNV isolated from ducks.
Similar content being viewed by others
References
Walker, M. H, Rup, B. J., Rubin, A. S. et al., Specificity in the immunosuppression induced by avian reticuloendotheliosis virus, Infect Immun., 1983, 40: 225–235.
Witter, R. L., Reticuloendotheliosis, in Disease of Poultry (ed. Calnek, B. W.), 10th ed., Ames: Iowa University Press, 467–484.
Ridgway, A. A., Swift, R. A., Kung, H. J. et al., In vitro transcription analysis of the viral promoter involved in c-myc activation in chicken lymphomas: Detection and mapping for two RNA initiation sites with the reticuloendotheliosis virus long terminal repeat, J. Virol., 1985, 54: 161–170.
Shimotohno, K., Mizutani, S., Temin, H. M., Sequence of retrovirus provirus resembles that of bacterial transposable elements, Nature, 1980, 285: 550–554.
Gabus-Darlix, C., Darlix, J., Novel internal ribosome entry site and vector containing same, GenBank, GI: 6732807.
Weaver, T. A., Talbot, K. J., Panganiban A. T., Spleen necosis virus gag polyprotein is necessary for partical assembly and release but not for proteolytic processing, J. Virol., 1990, 64: 2642–2652.
Wilhelmsen, L. C., Eggletin, K., Temin, H. M., Nucleic acid sequences of the oncogene v-rel in reticuloendotheliosis virus strain T and its cellular homolog the proto-oncogene c-rel, J. Virol., 1984, 52: 172–182.
Panganiban, A. T., Temin, H. M., Circles with two tandem LTRs are precursors to integrated retrovirus DNA, Cell, 1984, 36(3): 673–679.
Singh, P., Kim, T. J., Tripathy, D. N., Re-emerging fowpox: Evaluation of isolates from vaccinated flocks, Avian Pathol., 2000, 29: 449–455.
Zhang, Z., Cui, Z., Jiang, S. et al., Co-infection with Marek’s disease virus and reticuloendotheliosis virus in tumor samples of chicken, Chin. J. Prevent. Vet. Med. (in Chinese), 2003, 4: 274–278.
Zhang, Z., Cui, Z., Isolation and identification of reticuloendotheliosis vrus from tumors infected with subgroup J avian leukosis virus, Chin. J. Vet. Sci. (in Chinese), 2004, 1: 10–13.
Zhang, Z., Cui, Z., Isolation of recombinant field strains of Marek’s disease virus integrated with reticuloendotheliosis virus genome fragments, Science in China, Ser. C, 2005, 48(1): 81–88.
Cui, Z., Du, Y., Zhao, W., Reticuloendotheliosis virus infection and immunodepression of chicken flocks, Chin. J. Vet. Sci., 2000, 34(4): 1–3.
Cui, Z., Lee, L. F., Silva, R. F. et al., Monoclonal antibodies against avian reticuloendotheliosis virus’ identification of strain-specific and strain-common epitopes, J. Immunol., 1986, 136: 4237–4242.
Ridgway, A. A., Swift, R. A., Kung, H. J. et al., In vitro transcription analysis of the viral promoter involved in c-myc activation in chicken B lymphomas: Detection and mapping of two RNA initiation sites within the reticuloendotheliosis virus long terminal repeat, J. Virol., 1985, 54(1): 161–170.
Chen, I. S., Malk, T. W., O’Pear, J. J. et al., Characterization of reticuloendotheliosis virus strain T DNA and isolation of a novel variant of reticuloendotheliosis virus strain T by molecular cloning, J. Virol., 1981, 40: 800–811.
Trager, W., A new virus of ducks interfering with development of malaria parasite (Plasmodium lophurase), Proc. Soc. Exp. Biol. Med., 1959, 101: 578–582.
Wilhelmsen, K. C., Eggleton, K., Temin, H. M., Nucleic acid sequences of the oncogene v-rel in reticuloendotheliosis virus strain T and its cellular homolog, the proto-oncogene c-rel, J. Virol, 1984, 52(1): 172–182.
Zhao, W., Ding, J., Cui, Z., Analysis of LTR sequence from different REV strains, Journal of Shanghai Agricultural College (in Chinese), 2001, 1(3): 18–21.
Kost, R. V., Jones, D., Isflrt, R. J. et al., Retrovirus insertion into herpesvirus: Characterization of a Marek’s disease virus harboring a solo LTR, Virology, 1992, 192: 161–169.
Heritig, C., Coupar, B. E., Gould, A. R. et al., Field and vaccine strains of fowlpox carry integrated sequences from the avian retrovirus, reticuloendotheliosis virus, Virology, 1997, 235: 367–376.
Isfort, R. J., Jones, D., Kost, R. G. et al., Retrovirus insertion into herpesvirus in vitro and in vivo, Proc. Natl. Acad. Sci., USA, 1992, 89: 991–995.
Garcia, M., Narang, N., Reed, W. M. et al., Molecular characterization of reticuloendotheliosis virus insertions in the genome of field and vaccine strains of fowl poxvirus, Avian Dis., 2003, 47: 343–354.
Singh, P., Schitzlein, W. M., Tripathy, D. N., Reticuloendotheliosis virus sequences within the genomes of field strains of fowlpox virus display variability, J. Virol, 2003, 77(10): 5855–5862.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, Y., Cui, Z. & Jiang, S. Sequence analysis for the complete proviral genome of reticuloendotheliosis virus Chinese strain HA9901. SCI CHINA SER C 49, 149–157 (2006). https://doi.org/10.1007/s11427-006-0149-8
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-006-0149-8