Abstract
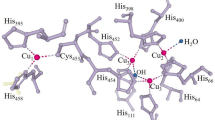
2,6-Dimethoxyphenol (DMP) as a substrate was widely used in determination of laccase activity. It is surprising, however, that its catalyzed oxidation products have not been completely determined until now. Studies were thus conducted on Rhus laccase (RL) and immobilized Rhus laccase (IRL)-catalyzed oxidation reactions of 2,6-dimethoxyphenol in water-organic solvent systems. These reactions proceeded well in water-(im)miscible organic solvent systems pre-saturated with water. Only one product, 3,3′,5,5′-tetramethoxy-1,1′biphenyl-4,4′-diol (TMBP), was produced by RL catalysis, and it was thoroughly characterized by FT-IR, NMR, GC-MS, etc. A simple enzymatic mechanism of this reaction is propos
Similar content being viewed by others
References
Wan Y Y, Du Y M, Miyakoshi T. Rhus laccase catalysis and product characterization of 1,2-Dimethoxyphenol in organic solutions. Chinese Chem Lett, 2008, 19(3): 333–336
Baldrian P. Fungal laccases-occurrence and properties. FEMS Microbiol Rev, 2005, 30: 215–242
Wan Y Y, Du Y M. Structure and catalytic mechanism of laccases. Chemistry, 2007, 70: 662–670
Okazaki S Y, Goto M, Wariishi H, Tanaka H, Furusaki S. Characterization and catalytic property of surfactant-laccase complex in organic media. Biotechnol Progr, 2000, 16: 583–588
Machonkin T E, Quintanar L, Palmer A E, Hassett R, Severance S, Kosman D J, Solomon E I. Spectroscopy and reactivity of the type 1 copper site in Fet3p from Saccharomyces cerevisiae: Correlation of structure with reactivity in the multicopper oxidases. J Am Chem Soc, 2001, 123: 5507–5517
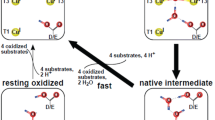
Lee S K, George S D, Antholine W E, Hedman B, Hodgson K O, Solomon E I. Nature of the intermediate formed in the reduction of O2 to H2O at the trinuclear copper cluster active site in native laccase. J Am Chem Soc, 2002, 124: 6180–6193
Chen C L, Gratzl J S, Kirkman A G, Miyakoshi T. Proceedings of the 5th brazilian symposium on the chemistry of lignins and other wood components. Curitiba, PR, Brazil. 8-13, 1997, 547–556
Shiba T, Xiao L, Miyakoshi T, Chen C L. Oxidation of isoeugenol and coniferyl alcohol catalyzed by laccases isolated from Rhus vernicifera stokes and Pycnoporus coccineus. J Mol Catal B: Enzym, 2000, 10: 605–615
Rodakiewicz-Nowak J, Kasture S M, Dudek B, Haber J. Effect of various water-miscible solvents on enzymatic activity of fungal laccases. J Mol Catal B: Enzym, 2000, 11: 1–11
Du Y M. New advance on study Chinese lacquer. Chemistry, 1986, 1–9
Mayer A M, Staples R C. Laccase: new functions for an old enzyme. Phytochemistry, 2002, 60: 551–565
Mustafa R, Muniglia R, Rovel G, Girardin M. Phenolic colorants obtained by enzymatic synthesis using a fungal laccase in a hydro-organic biphasic system. Food Res Int, 2005, 38: 995–1000
Nicotra S, Intra A, Ottolina G, Riva S, Danieli B. Laccase-mediated oxidation of the steroid hormone 17β-estradiol in organic solvents. Tetrahedron-Asymmetr, 2004, 15: 2927–2931
Wan Y Y, Du Y M, Yang F X, Xu Y, Chen R Z, Kennedy J F. Purification and characterization of hydrosoluble components from the sap of Chinese lacquer tree Rhus vernicifera. Int J Biol Macromol, 2006, 38: 232–240
Wan Y Y, Lu R, Du Y M, Honda T, Miyakoshi T. Does Donglan lacquer tree belong to Rhus vernicifera species? Int J Biol Macromol, 2007, 41: 497–503
Wan Y Y, Du Y M, Shi X W, Li J, Ru L, Miyakoshi T. Immobilization and characterization of Rhus laccase from Chinese Rhus vernicifera on modified chitosan. Process Biochem, 2006, 41: 1378–1382
Du Y M, Zhang J M. Studies on oxidation performance and reaction product of Rhus laccase in reverse micelle. Chem J Chin Univ, 1995, 16: 715–718
Wan Y Y, Lu R, Akiyama K, Miyakoshi T, Du Y M. Enzymatic synthesis of bioactive compounds by Rhus laccase from Chinese Rhus vernicifera. Sci China Ser B: Chem, 2007, 50: 179–182
Author information
Authors and Affiliations
Corresponding author
Additional information
This paper is dedicated to professor Du YuMin on the occasion of his 70th birthday
Rights and permissions
About this article
Cite this article
Wan, Y., Du, Y. & Miyakoshi, T. Enzymatic catalysis of 2,6-dimethoxyphenol by laccases and products characterization in organic solutions. Sci. China Ser. B-Chem. 51, 669–676 (2008). https://doi.org/10.1007/s11426-008-0071-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-008-0071-y