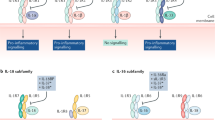
Summary
IL-20 belongs to the IL-10 family and plays a role in skin inflammation and the development of hematopoietic cells. Little is known about its other biological functions and clinical implications, however. Updated information about IL-20, such as its identification, expression, receptors, signaling, biological activities, and potential clinical implications, is illustrated in this review based on our research and on data available in the literature. Our studies of IL-20 show that it is a pleiotropic cytokine with potent inflammatory, angiogenic, and chemoattractive characteristics. Inflammation and angiogenesis are essential for the pathogenesis of rheumatoid arthritis and atherosclerosis. Based on in vitro data and clinical samples, we demonstrated that IL-20 is involved in the diseases of rheumatoid arthritis and atherosclerosis. In addition, we found in our studies that IL-20 signaled through different molecules in several cells. The present review presents the clinical implications of IL-20 in rheumatoid arthritis and atherosclerosis. It may provide new therapeutic options in the future.
Article PDF
Similar content being viewed by others
Abbreviations
- Akt kinase:
-
a serine/threonine kinase
- ApoE:
-
apolipoprotein E
- bFGF:
-
basic fibroblast growth factor
- CIA:
-
collagen-induced arthritis
- COX:
-
cyclooxygenase
- ERK:
-
extracellular signal-regulated protein kinase
- EST:
-
expressed sequence tag
- IFN:
-
interferon
- Jak:
-
Janus kinase
- LDL:
-
low-density lipoprotein
- MCP:
-
monocyte chemoattractant protein
- MIP:
-
macrophage inflammatory protein
- MMP:
-
matrix metalloproteinase
- Ox-LDL:
-
oxidized low-density lipoprotein
- PMA:
-
phorbol myristate acetate
- RA:
-
rheumatoid arthritis
- RANTES:
-
regulated upon activation, normal T-cell expressed and secreted
- RASFs:
-
rheumatoid arthritis synovial fibroblasts
- RT-PCR:
-
reverse transcriptase polymerase chain reaction
- STAT:
-
signal transducers and activators of transcription
- TNF-α:
-
tumor necrosis factor-alpha
- VEGF:
-
vascular endothelial cell growth factor
References
Moore K.W., de Waal Malefyt R., Coffman R.L., O’Garra A. (2001). Interleukin-10 and the interleukin-10 receptor. Annu. Rev. Immunol. 19:683–765
Moore K.W., Vieira P., Fiorentino D.F., Trounstine M.L., Khan T.A., Mosmann T.R. (1990) Homology of cytokine synthesis inhibitory factor (IL-10) to the Epstein-Barr virus gene BCRFI. Science 248(4960):1230–1234
Thompson-Snipes L., Dhar V., Bond M.W., Mosmann T.R., Moore K.W., Rennick D.M. (1991). Interleukin 10: a novel stimulatory factor for mast cells and their progenitors. J. Exp. Med. 173(2):507–510
Rousset F., Garcia E., Defrance T., Peronne C., Vezzio N., Hsu D.H., Kastelein R., Moore K.W., Banchereau J. (1992) Interleukin 10 is a potent growth and differentiation factor for activated human B lymphocytes. Proc. Natl. Acad. Sci. USA. 89(5):1890–1893
Go N.F., Castle B.E., Barrett R., Kastelein R., Dang W., Mosmann T.R., Moore K.W., Howard M. (1990) Interleukin 10, a novel B cell stimulatory factor: unresponsiveness of X chromosome-linked immunodeficiency B cells. J. Exp. Med. 172(6):1625–1631
Gallagher G., Dickensheets H., Eskdale J., Izotova L.S., Mirochnitchenko O.V., Peat J.D., Vazquez N., Pestka S., Donnelly R.P., Kotenko S.V. (2000). Cloning, expression and initial characterization of interleukin-19 (IL-19), a novel homologue of human interleukin-10 (IL-10). Genes. Immun. 1(7):442–450
Blumberg H., Conklin D., Xu W.F., Grossmann A., Brender T., Carollo S., Eagan M., Foster D., Haldeman B.A., Hammond A., Haugen H., Jelinek L., Kelly J.D., Madden K., Maurer M.F., Parrish-Novak J., Prunkard D., Sexson S., Sprecher C., Waggie K., West J., Whitmore T.E., Yao L., Kuechle M.K., Dale B.A., Chandrasekher Y.A. (2001). Interleukin 20: discovery, receptor identification, and role in epidermal function. Cell. 104(1):9–19
Dumoutier L., Louahed J., Renauld J.-C. (2000) Cloning and characterization of IL-10-related T cell-derived inducible factor (IL-TIF), a novel cytokine structurally related to IL-10 and inducible by IL-9. J. Immunol. 164(4):1814–1819
Jiang H., Lin J.J., Su Z.Z., Goldstein N.I., Fisher P.B. (1995) Subtraction hybridization identifies a novel melanoma differentiation associated gene, mda-7, modulated during human melanoma differentiation, growth and progression. Oncogene 11(12):2477–2486
Knappe A., Hor S., Wittmann S., Fickenscher H. (2000) Induction of a novel cellular homolog of interleukin-10, AK155, by transformation of T lymphocytes with herpesvirus saimiri. J. Virol. 74(8):3881–3887
Liao Y.C., Liang W.G., Chen F.W., Hsu J.H., Yang J.J., Chang M.S. (2002). IL-19 induces production of IL-6 and TNF-alpha and results in cell apoptosis through TNF-alpha. J. Immunol. 169(8):4288–4297
Liao S.C., Cheng Y.C., Wang Y.C., Wang C.W., Yang S.M., Yu C.K., Shieh C.C., Cheng K.C., Lee M.F., Chiang S.R., Shieh J.M., Chang M.S. (2004). IL-19 induced Th2 cytokines and was up-regulated in asthma patients. J. Immunol. 173(11):6712–6718
Dumoutier L., Van Roost E., Colau D., Renauld J.C. (2000). Human interleukin-10-related T cell-derived inducible factor: molecular cloning and functional characterization as an hepatocyte-stimulating factor. Proc. Natl. Acad. Sci. USA. 97(18):10144–10149
Radaeva S., Sun R., Pan H.N., Hong F., Gao B. (2004). Interleukin 22 (IL-22) plays a protective role in T cell-mediated murine hepatitis: IL-22 is a survival factor for hepatocytes via STAT3 activation. Hepatology 39(5):1332–1342
Wolk K., Kunz S., Witte E., Friedrich M., Asadullah K., Sabat R. (2004). IL-22 increases the innate immunity of tissues. Immunity 21(2):241–254
Hor S., Pirzer H., Dumoutier L., Bauer F., Wittmann S., Sticht H., Renauld J.C., de Waal Malefyt R., Fickenscher H. (2004). The T-cell lymphokine interleukin-26 targets epithelial cells through the interleukin-20 receptor 1 and interleukin-10 receptor 2 chains. J. Biol. Chem. 279(32):33343–33351
Sheikh F., Baurin V.V., Lewis-Antes A., Shah N.K., Smirnov S.V., Anantha S., Dickensheets H., Dumoutier L., Renauld J.C., Zdanov A., Donnelly R.P., Kotenko S.V. (2004). Cutting edge: IL-26 signals through a novel receptor complex composed of IL-20 receptor 1 and IL-10 receptor 2. J. Immunol. 172(4):2006–2010
Liu L., Ding C., Zeng W., Heuer J.G., Tetreault J.W., Noblitt T.W., Hangoc G., Cooper S., Brune K.A., Sharma G., Fox N., Rowlinson S.W., Rogers D.P., Witcher D.R., Lambooy P.K., Wroblewski V.J., Miller J.R., Broxmeyer H.E. (2003). Selective enhancement of multipotential hematopoietic progenitors in vitro and in vivo by IL-20. Blood 102(9):3206–3209
Zdanov A., Schalk-Hihi C., Gustchina A., Tsang M., Weatherbee J., Wlodawer A. (1995). Crystal structure of interleukin-10 reveals the functional dimer with an unexpected topological similarity to interferon gamma. Structure 3(6):591–601
Hsieh M.Y., Chen W.Y., Jiang M.J., Cheng B.C., Huang T.Y., Chang M.S., IL-20 promotes angiogenesis in a direct and indirect manner. Genes. Immun. 7(3): 234–242, 2006
Dumoutier L., Leemans C., Lejeune D., Kotenko S.V., Renauld J.C. (2001). Cutting edge: STAT activation by IL-19, IL-20 and mda-7 through IL-20 receptor complexes of two types. J. Immunol. 167(7):3545–3549
Parrish-Novak J., Xu W., Brender T., Yao L., Jones C., West J., Brandt C., Jelinek L., Madden K., McKernan P.A., Foster D.C., Jaspers S., Chandrasekher Y.A. (2002). Interleukins 19, 20, and 24 signal through two distinct receptor complexes. Differences in receptor-ligand interactions mediate unique biological functions. J. Biol. Chem. 277(49):47517–47523
Hsu Y.H., Li H.H., Hsieh M.Y., Liu M.F., Huang K.Y., Chin L.S., Chang M.S., Interleukin-20 functions as a proinflammatory molecule in rheumatoid and experimental arthritis. Arthritis. Rheum. (2006) In press
Nagalakshmi M.L., Murphy E., McClanahan T., de Waal Malefyt R. (2004). Expression patterns of IL-10 ligand and receptor gene families provide leads for biological characterization. Int. Immunopharmacol. 4(5):577–592
Wei C.C., Chen W.Y., Wang Y.C., Chen P.J., Lee J.Y., Wong T.W., Chen W.C., Wu J.C., Chen G.Y., Chang M.S., Lin Y.C. (2005). Detection of IL-20 and its receptors on psoriatic skin. Clin. Immunol. 117(1):65–72
Wolk K., Kunz S., Asadullah K. and Sabat R., Cutting edge: immune cells as sources and targets of the IL-10 family members? J. Immunol. 168(11): 5397–5402, 2002
Ding Y., Qin L., Kotenko S.V., Pestka S. and Bromberg J.S., A single amino acid determines the immunostimulatory activity of interleukin 10. J. Exp. Med. 191(2): 213–224, 2000
Aggarwal S., Xie M.H., Maruoka M., Foster J. and Gurney A.L., Acinar cells of the pancreas are a target of interleukin-22. J. Interferon Cytokine Res. 21(12): 1047–1053, 2001
Gurney A.L. IL-22, aTh1 cytokine that targets the pancreas and select other peripheral tissues. Int. Immunopharmacol. 4(5): 669–677, 2004
Gottlieb S.L., Gilleaudeau P., Johnson R., Estes L., Woodworth T.G., Gottlieb A.B., Krueger J.G. (1995). Response of psoriasis to a lymphocyte-selective toxin (DAB389IL-2) suggests a primary immune, but not keratinocyte, pathogenic basis. Nat. Med. 1(5):442–447
Asadullah K., Docke W.D., Volk H.D., Sterry W. (1999). The pathophysiological role of cytokines in psoriasis. Drugs Today (Barc). 35(12):913–924
Romer J., Hasselager E., Norby P.L., Steiniche T., Thorn Clausen J., Kragballe K. (2003). Epidermal overexpression of interleukin-19 and −20 mRNA in psoriatic skin disappears after short-term treatment with cyclosporine a or calcipotriol. J. Invest. Dermatol. 121(6):1306–1311
Hansen A., Cao R., Cao Y., Clausen J.T., Dissing S., Hasselager E., Myren M., Norby P.L., Olsen U.B., Tritsaris K. (2005). IL-20 is an angiogenic factor in psoriasis. J. Invest. Dermatol. 124(4):A4–A4
Otkjaer K., Kragballe K., Funding A.T., Clausen J.T., Noerby P.L., Steiniche T., Iversen L. (2005). The dynamics of gene expression of interleukin-19 and interleukin-20 and their receptors in psoriasis. Br. J. Dermatol. 153(5):911–918
Nickoloff B.J., Wrone-Smith T. (1999). Injection of pre-psoriatic skin with CD4+ T cells induces psoriasis. Am. J. Pathol. 155(1):145–158
Panayi G.S., Lanchbury J.S., Kingsley G.H. (1992). The importance of the T cell in initiating and maintaining the chronic synovitis of rheumatoid arthritis. Arthritis. Rheum. 35(7):729–735
Firestein G.S., Zvaifler N.J. (2002). How important are T cells in chronic rheumatoid synovitis?: II. T cell-independent mechanisms from beginning to end. Arthritis. Rheum. 46(2):298–308
Wipke B.T., Allen P.M. (2001). Essential role of neutrophils in the initiation and progression of a murine model of rheumatoid arthritis. J. Immunol. 167(3):1601–1608
Walmsley M., Katsikis P.D., Abney E., Parry S., Williams R.O., Maini R.N., Feldmann M. (1996). Interleukin-10 inhibition of the progression of established collagen-induced arthritis. Arthritis. Rheum. 39(3):495–503
Finnegan A., Kaplan C.D., Cao Y., Eibel H., Glant T.T., Zhang J. (2003). Collagen-induced arthritis is exacerbated in IL-10-deficient mice. Arthritis. Res. Ther. 5(1):R18–R24
Katsikis P.D., Chu C.Q., Brennan F.M., Maini R.N., Feldmann M. (1994). Immunoregulatory role of interleukin 10 in rheumatoid arthritis. J. Exp. Med. 179(5):1517–1527
Smeets T.J.M., Chandrasekher Y., Haringman J.J., Tak P.P. (2004). IL-20 is expressed in inflamed synovium of patients with psoriatic arthritis and rheumatoid arthritis. Arthritis Res. Therapy 6:S9–S9
Brennan F.M., Zachariae C.O., Chantry D., Larsen C.G., Turner M., Maini R.N., Matsushima K., Feldmann M. (1990). Detection of interleukin 8 biological activity in synovial fluids from patients with rheumatoid arthritis and production of interleukin 8 mRNA by isolated synovial cells. Eur. J. Immunol. 20(9):2141–2144
Koch A.E., Kunkel S.L., Burrows J.C., Evanoff H.L., Haines G.K., Pope R.M., Strieter R.M. (1991). Synovial tissue macrophage as a source of the chemotactic cytokine IL-8. J. Immunol. 147(7):2187–2195
Seitz M., Dewald B., Gerber N., Baggiolini M. (1991). Enhanced production of neutrophil-activating peptide-1/interleukin-8 in rheumatoid arthritis. J. Clin. Invest. 87(2):463–469
Villiger P.M., Terkeltaub R., Lotz M. (1992). Production of monocyte chemoattractant protein-1 by inflamed synovial tissue and cultured synoviocytes. J. Immunol. 149(2):722–727
Koch A.E., Kunkel S.L., Harlow L.A., Johnson B., Evanoff H.L., Haines G.K., Burdick M.D., Pope R.M., Strieter R.M. (1992). Enhanced production of monocyte chemoattractant protein-1 in rheumatoid arthritis. J. Clin. Invest. 90(3):772–779
Rathanaswami P., Hachicha M., Sadick M., Schall T.J., McColl S.R. (1993). Expression of the cytokine RANTES in human rheumatoid synovial fibroblasts. Differential regulation of RANTES and interleukin-8 genes by inflammatory cytokines. J. Biol. Chem. 268(8):5834–5839
Koch A.E., Kunkel S.L., Harlow L.A., Mazarakis D.D., Haines G.K., Burdick M.D., Pope R.M., Strieter R.M. (1994). Macrophage inflammatory protein-1 alpha. A novel chemotactic cytokine for macrophages in rheumatoid arthritis. J. Clin. Invest. 93(3):921–928
Feldmann M. (2002). Development of anti-TNF therapy for rheumatoid arthritis. Nat. Rev. Immunol. 2(5):364–371
Moreland L.W., Baumgartner S.W., Schiff M.H., Tindall E.A., Fleischmann R.M., Weaver A.L., Ettlinger R.E., Cohen S., Koopman W.J., Mohler K., Widmer M.B., Blosch C.M. (1997). Treatment of rheumatoid arthritis with a recombinant human tumor necrosis factor receptor (p75)-Fc fusion protein. N. Engl. J. Med. 337(3):141–147
Elliott M.J., Maini R.N., Feldmann M., Long-Fox A., Charles P., Bijl H., Woody J.N. (1994). Repeated therapy with monoclonal antibody to tumour necrosis factor alpha (cA2) in patients with rheumatoid arthritis. Lancet 344(8930):1125–1127
Ross R. (1999). Atherosclerosis–an inflammatory disease. N. Engl. J. Med. 340(2):115–126
van der Wal A.C., Becker A.E., van der Loos C.M., Das P.K. (1994). Site of intimal rupture or erosion of thrombosed coronary atherosclerotic plaques is characterized by an inflammatory process irrespective of the dominant plaque morphology. Circulation 89(1):36–44
Fuster V., Badimon L., Badimon J.J., Chesebro J.H. (1992). The pathogenesis of coronary artery disease and the acute coronary syndromes (1). N. Engl. J. Med. 326(4):242–250
Lee R.T., Libby P. (1997). The unstable atheroma. Arterioscler. Thromb. Vasc. Biol. 17(10):1859–1867
Mallat Z., Besnard S., Duriez M., Deleuze V., Emmanuel F., Bureau M.F., Soubrier F., Esposito B., Duez H., Fievet C., Staels B., Duverger N., Scherman D., Tedgui A. (1999). Protective role of interleukin-10 in atherosclerosis. Circ. Res. 85(8):e17–24
Paterson J. (1938). Capillary rupture with intimal hemorrhage as a causative factor in coronary thrombosis. Arch. Pathol. 25:474
Tenaglia A.N., Peters K.G., Sketch M.H. Jr., Annex B.H. (1998). Neovascularization in atherectomy specimens from patients with unstable angina: implications for pathogenesis of unstable angina. Am. Heart. J. 135(1):10–14
Chada S., Mhashilkar A.M., Ramesh R., Mumm J.B., Sutton R.B., Bocangel D., Zheng M.Z., Grimm E.A., Ekmekcioglu S. (2004). Bystander activity of Ad-mda7: Human MDA-7 protein kills melanoma cells via an IL-20 receptor-dependent but STAT3-independent mechanism. Mol. Ther. 10(6):1085–1095
O’Brien E.R., Garvin M.R., Dev R., Stewart D.K., Hinohara T., Simpson J.B., Schwartz S.M. (1994). Angiogenesis in human coronary atherosclerotic plaques. Am. J. Pathol. 145(4):883–894
Panutsopulos D., Papalambros E., Sigala F., Zafiropoulos A., Arvanitis D.L., Spandidos D.A. (2005). Protein and mRNA expression levels of VEGF-A and TGF-beta1 in different types of human coronary atherosclerotic lesions. Int. J. Mol. Med. 15(4):603–610
Chen W.Y., Cheng Y.C., Lei H.Y., Chang C.P., Wang C.W., Chang M.S. (2005). IL-24 inhibits the growth of hepatoma cells in vivo. Genes Immun. 6:493–499
Inoue M., Itoh H., Ueda M., Naruko T., Kojima A., Komatsu R., Doi K., Ogawa Y., Tamura N., Takaya K., Igaki T., Yamashita J., Chun T.H., Masatsugu K., Becker A.E., Nakao K. (1998). Vascular endothelial growth factor (VEGF) expression in human coronary atherosclerotic lesions: possible pathophysiological significance of VEGF in progression of atherosclerosis. Circulation 98(20):2108–2116
Zeng B., Prasan A., Fung K.C., Solanki V., Bruce D., Freedman S.B., Brieger D. (2005). Elevated circulating levels of matrix metalloproteinase-9 and −2 in patients with symptomatic coronary artery disease. Intern. Med. J. 35(6):331–335
Heuze-Vourc’h N., Liu M., Dalwadi H., Baratelli F.E., Zhu L., Goodglick L., Pold M., Sharma S., Ramirez R.D., Shay J.W., Minna J.D., Strieter R.M., Dubinett S.M. (2005). IL-20, an anti-angiogenic cytokine that inhibits COX-2 expression. Biochem. Biophys. Res. Commun. 333(2):470–475
Rich B.E., Kupper T.S. (2001). Cytokines: IL-20 – a new effector in skin inflammation. Curr. Biol. 11(13):R531–R534
Rich B.E. (2003). IL-20: a new target for the treatment of inflammatory skin disease. Expert. Opin. Ther. Targets. 7(2):165–174
Gonzalez-Gay M.A., Gonzalez-Juanatey C., Martin J. (2005). Rheumatoid arthritis: a disease associated with accelerated atherogenesis. Semin. Arthritis. Rheum. 35(1):8–17
Wallberg-Jonsson S., Ohman M., Rantapaa-Dahlqvist S. (2004). Which factors are related to the presence of atherosclerosis in rheumatoid arthritis?. Scand. J. Rheumatol. 33(6):373–379
Veale D.J., Ritchlin C., FitzGerald O. (2005). Immunopathology of psoriasis and psoriatic arthritis. Ann. Rheum. Dis. 64 (Suppl 2): ii26–ii29
Veale D.J., FitzGerald O. (2002). Psoriatic arthritis – pathogenesis and epidemiology. Clin. Exp. Rheumatol. 20(6 Suppl 28):S27–33
Costello P., FitzGerald O. (2001). Disease mechanisms in psoriasis and psoriatic arthritis. Curr. Rheumatol. Rep. 3(5):419–427
Kane D., FitzGerald O. (2004). Tumor necrosis factor-alpha in psoriasis and psoriatic arthritis: a clinical, genetic, and histopathologic perspective. Curr. Rheumatol. Rep. 6(4):292–298
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was supported by a grant from Chi-Mei Medical Center, Tainan, Taiwan.
Rights and permissions
About this article
Cite this article
Wei, CC., Hsu, YH., Li, HH. et al. IL-20: biological functions and clinical implications. J Biomed Sci 13, 601–612 (2006). https://doi.org/10.1007/s11373-006-9087-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11373-006-9087-5