Summary
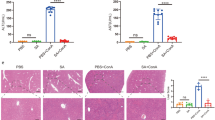
Tumor necrosis factor-alpha (TNF-α) plays a central role in cellular necrosis, apoptosis, organ failure, tissue damage, inflammation and fibrosis. These processes, occurring in liver injury, may lead to cirrhosis. Thalidomide, α-N-phthalidoglutarimide, (C13H10N2)4, has been shown to have immunomodulatory and anti-inflammatory properties, possibly mediated through its anti-TNF-α effect. In this study, we investigated the in vitro and in vivo effects of thalidomide on hepatic fibrosis. A cell line of rat hepatic stellate cells (HSC-T6) was stimulated with transforming growth factor-β1 (TGF-β1) or TNF-α. The inhibitory effects of thalidomide on the NFκB signaling cascade and fibrosis markers including α-smooth muscle actin (α-SMA) and collagen, were assessed. An in vivo therapeutic study was conducted in dimethylnitrosamine (DMN)-treated rats, which were randomly assigned to 1 of 4 groups: vehicle (0.7% carboxyl methyl cellulose, CMC), thalidomide (40 mg/kg), thalidomide (200 mg/kg), or silymarin (50 mg/kg), each given by gavage twice daily for 3 weeks starting after 1 week of DMN administration. Thalidomide (100–800 nM) concentration-dependently inhibited NFκB transcriptional activity induced by TNF-α, including IKKα expression and IκBα phosphorylation in HSC-T6 cells. In addition, thalidomide also suppressed TGF-β1-induced α-SMA expression and collagen deposition in HSC-T6 cells. Fibrosis scores of livers from DMN-treated rats receiving high dose of thalidomide (0.89±0.20) were significantly reduced in comparison with those of DMN-treated rats receiving vehicle (1.56±0.18). Hepatic collagen contents of DMN rats were also significantly reduced by either thalidomide or silymarin treatment. Immunohistochemical double staining results showed that α-SMA- and NFκB-positive cells were decreased in the livers from DMN rats receiving either thalidomide or silymarin treatment. In addition, real-time PCR analysis indicated that hepatic mRNA expressions of TGF-β1, α-SMA, collagen 1α2, TNF-α and iNOS genes were attenuated by thalidomide treatment. In conclusion, our results showed that thalidomide inhibited activation of HSC-T6 cells by TNF-α and ameliorated liver fibrosis in DMN-intoxicated rats.
Article PDF
Similar content being viewed by others
Abbreviations
- α-SMA:
-
α-smooth muscle actin
- Col 1α2:
-
collagen 1α2
- DMN:
-
dimethylnitrosamine
- G3PDH:
-
glyceraldehyde-3-phosphate dehydrogenase
- HSC:
-
hepatic stellate cell
- iNOS:
-
inducible nitric oxide synthase
- TGF-β1:
-
transforming growth factor-β1
- TNF-α:
-
tumor necrosis factor-α
References
Friedman S.L. (2003). Liver fibrosis- from bench to bedside. J. Hepatol. 38: S38-S53
Bataller R., Brenner D.A., (2005). Liver fibrosis. J. Clin. Invest. 115: 209–218
Pinzani M., Marra F. (2001). Cytokine receptors and signaling in hepatic stellate cells. Semin. Liver. Dis. 21: 397–416
Lotersztajn S., Julien B., Teixeira-Clerc F., Grenard P., Mallat A. (2005). Hepatic fibrosis: molecular mechanisms and drug targets. Ann. Rev. Pharmacol. Toxicol. 45: 605–628
Barnes P.J., Karin M. (1997). Mechanisms of disease: Nuclear factor кB – a pivotal transcription factor in chronic inflammatory diseases. N. Engl. J. Med. 336: 1066–1071
Kumar A., Takada Y., Boriek A.M., Aggarwal B.B. (2004). Nuclear factor кB: its role in health and disease. J. Mol. Med. 82: 434–448
Viatour P., Merville M.P., Bours V., Chariot A. (2005). Phosphorylation of NFкB and I кB proteins: implication in cancer and inflammation. Trends Biochem. Sci. 30: 43–52
Mercurio F., Zhu H., Murray B.W. (1997). IKK-1 and IKK-2: cytokine-activated IkappaB kinases essential for NF-kappaB activation. Science 278: 860–866
Zandi E., Rothwarf D.M., Delhase M., Hayakawa M., Karin M. (1997). The IkappaB kinase complex (IKK) contains two kinase subunits, IKK alpha and IKK beta, necessary for IkappaB phosphorylation and NF-kappa B activation. Cell 91: 243–252
Perkins N.D. (2000). The Rel/NFkappa B family: friend and foe. Trends Biochem. Sci. 25: 434–440
Freedman A.R., Sharma R.J., Nabel G.J., Emarson S.G., Griffin G.E. (1992). Cellular distribution of nuclear factor kappa B binding activity in rat liver. Biochem. J. 18: 6353–6364
Hellerbrand C., Jobin C., Limuro Y., Licato L., Sartor R.B., Brenner D.A. (1998). Inhibition of NF kappa B in activated rat hepatic stellate cells by proteasome inhibitors and an IkappaB superrepressor. Hepatology 27: 1285–1295
Lang A., Schoonhoven R., Tuvia S., Brenner D.A., Rippe R.A. (2000). Nuclear factor kappa B in proliferation, activation, and apoptosis in rat hepatic stellate cells. J. Hepatol. 33: 49–58
Saile B., Matthes N., El Armouche H., Neubauer K., Ramadori G. (2001). The bcl, NF кB and p53/p21WAF1 systems are involved in spontaneous apoptosis and in the anti-apoptotic effect of TGF-\(\beta\) or TNF-\(\alpha\) on activated hepatic stellate cells. Eur. J. Cell Biol. 80: 554–561
Bradham C.A., Plumpe J., Manns M.P., Brenner D.A., Trautwein C. (1998). Mechanisms of hepatic toxicity I. TNF-induced liver injury. Am. J. Physiol. 275: G387-G392
Iimuro Y., Gallucci R.M., Luster M.I., Kono H., Thurman R.G. (1997). Antibodies to tumor necrosis factor \(\alpha\) attenuate hepatic necrosis and inflammation caused by chronic exposure to ethanol in the rat. Hepatology 26:1530–1537
Moreira A.L., Sampaio E.P., Zmuidzinas A., Frindt P., Smith K.A., Kaplan G. (1993). Thalidomide exerts its inhibitory actions on tumor necrosis factor alpha by enhancing mRNA degradation. J. Exp. Med. 177: 1675–1680
Sampaio E.P., Sarno E.N., Galilly R., Cohn Z.A., Kaplan G. (1991). Thalidomide selectively inhibits tumor necrosis alpha production by stimulated human monocytes. J. Exp. Med. 173: 699–703
Deng L., Ding W.H., Granstein R.D. (2003). Thalidomide inhibits tumor necrosis factor-\(\alpha\) production and antigen presentation by Langerhans cells. J. Invest. Dermatol. 121: 1060–1065
Ehrenpreis E.D., Kane S.V., Cohen L.B., Cohen R.D., Hanauer S.B. (1999). Thalidomide therapy for patients with refractory Crohn’s disease: an open-label trial. Gastroenterology 117: 1271–1277
Reyes-Teran G., Sirrra-Madero J.G., Martinez del Cerro V., Arroyo-Figueroa H., Pasquetti A., Calva J.J. (1996). Effects of thalidomide on HIV-associated wasting syndrome: a randomized, double-blind, placebo-controlled clinical trail. AIDS 10: 1501–1507
Stevens R.J., Andujar C., Edwards C.J., Ames P.R., Barwick A.R., Khamashta M.A. (1997). Thalidomide in the treatment of the cutaneuous manifestation of lupus erythematous: experience in sixteen consecutive patients. Br. J. Rheumatol. 36: 353–359
Volgelsang G.B., Farmar E.R., Hess A.D., Altamonte V., Beschorner W.E., Jabs D.A. (1992). Thalidomide for the treatment of chronic graft versus host disease. N. Eng. J. Med. 326: 1055–1058
Enomoto N., Takei Y., Hirose M., Ikejima K., Miwa H., Kitamura T. (2002). Thalidomide prevent alcohol injury in rats through suppression of Kupffer cell sensitization and TNF-\(\alpha\) production. Gastroenterology 123: 291–300
Muriel P., Frenandez-Martinez E., Perez-Alvarez V., Lara-Ochoa F., Ponce S., Garcia J. (2003). Thalidomide ameliorates carbon tetrachloride induced cirrhosis in rats. Eur. J. Gastroenterol. Hepatol. 15: 951–957
Yeh T.S., Ho Y.P., Huang S.F., Yeh J.N., Jan Y.Y. and Chen M.F., Thalidomide salvages lethal hepatic necroinflammation and accelerates recovery from cirrhosis in rats. J. Hepatol. 41: 606–612, 2004
Fernandez-Martinez E., Morales-Rios M.S., Perez-Alvarez V., Muriel P. (2001). Effect of thalidomide and 3-phthalimido-3-(3,4-dimethoxyphenyl)-propanamide on bile duct obstruction –induced cirrhosis in rat. Drug Dev. Res. 54: 209–218
Vogel S., Piantedosi R., Frnak J., Lalazar A., Rockey D.C., Friedman S.L., Blaner WS. (2000). An immortalized rat liver stellate cell (HSC-T6); a new cell model for the study of retinoid metabolism in vitro. J. Lipid. Res. 41: 882–893
Hsu Y.-C., Lin Y.-L., Chiu Y.-T., Shiao M.-S., Lee C.-Y., Huang Y.-T. (2005). Anti-fibrotic effects of Salvia miltiorrhiza on dimethylnitrosamine-intoxicated rats. J. Biomed. Sci. 12: 185–195
Park S.G., Ryu H.M., Lim S.-O., Kim Y.-I., Soon B., Hwang S.B., Jung G. (2005). Interferon-\(\gamma\) inhibits hepatitis B virus-induced NF-кB activation through nuclear localization of NF-кB-inducing kinase. Gastroenterology 128: 2042–2053
Alpert D. and Vilček J. (2000). Inhibition of I кB kinase activity by sodium salicylate in vitro does not reflect its inhibitory mechanism in intact cells. J Biol. Chem. 275: 10925–10929
Yang F., Oz H.S., Barve S., De Villiers W.J.S., Mcclain C.J., Varilek G. (2001). The green tea polyphenol (-)-epigallocatechin-3-gallate blocks nuclear factor-кB activation by inhibiting I кB kinase activity in the intestinal epithelial cell line IEC-6. Mol. Pharmacol. 60: 528–533
Williams E.J., Benyon R.C., Trim N., Hadwin R., Grove B.H., Arthur M.J.P., Unemori E.N., Iredale J.P. (2001) Relaxin inhibits effective collagen deposition by cultured hepatic stellate cells and decreases rat liver fibrosis in vivo. Gut 49: 577–583
Shiba M., Shimizu I., Yasuda M., Ii K., Ito S. (1998). Expression of type I and type III collagens during the course of dimethylnitrosamine-induced hepatic fibrosis in rats. Liver 18: 196–204
Hansen M.B., Nielsen S.E., Berg K. (1989). Re-examination and further development of a precise and rapid dye method for measuring cell growth/cell kill. J. Immunol. Methods 119: 203–210
Hsu Y.-C., Chiu Y.-T., Lee C.-Y., Lin Y.-L., Huang Y.-T. (2004). Increases in fibrosis-related gene transcripts in livers of dimethylnitrosamine-intoxicated rats. J. Biomed. Sci. 11: 408–417
Jezequel A.M., Mancini R., Rinaldesi M.L., Macarri G., Venturini C., Orlandi F. (1987). A morphological study of the early stages of hepatic fibrosis induced by low doses of dimethylnitrosamine in the rat. J. Hepatol. 5: 174–181
Lopez-De Leon A., Rojkind M. (1985). A simple micromethod for collagen and total protein determination informalin-fixed paraffin-emdeded sections. J. Histochem. Cytochem. 33: 737–743
Sheuer P.J. (1991). Classification of chronic viral hepatitis: a need for reassessment. J. Hepatol. 13: 372–374
Kitamura Y., Ninomiya H. (2003). Smad expression of hepatic stellate cells in liver cirrhosis in vivo and hepatic stellate cell line in vitro. Pathol. Int. 53: 18–26
Chomczynski P., Sacchi N. (1987). Single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction. Anal. Biochem. 162: 156–159
Helm F.C., Frankus E., Friderichs E., Graudums I., Flohe L. (1981). Comparative teratological investigation of compounds structurally and pharmacologically related to thalidomide. Arzneimittel-Forschung 31: 941–949
Larrea E., Garcia N., Qian C., Civiera M.P., Prieto J. (1996). Tumor necrosis factor alpha gene expression and the response to interferon in chronic hepatitis C. Hepatology 23:210–217
Rodriguez R.E., Gonzalez R.E., Santolaria F.F., Milena A.A., Rodriguez M.F., Oramas R.J. (1995). Cytokines levels in acute alcoholic hepatitis: a sequential study. Drug Alcohol Depend. 39:23–27
Chamulitrat W., Blanzk M.E., Jordan S.J., Luster M.I., Mason R.P. (1995). Tumor necrosis factor alpha and nitric oxide production in endotoxin-primed rats administered carbon tetrachloride. Life Sci. 57: 2273–2280
Ghosh S., May M.J., Kopp E.B. (1998). NF кB and rel proteins: evolutionary conserved mediators of immune responses. Annu. Rev. Immunol. 16: 225–260
Wright M.C., Issa R., Smart D.E., Trim N., Murray G.I., Prismrose J.N. (2001). Gliotoxin stimulates the apoptosis of human and rat hepatic stellate cells and enhances the resolution of liver fibrosis in rats. Gastroenterology 121: 685–698
Keifer J.A., Guttridge D.C., Ashburner B.P., Baldwin A.S. Jr. (2001). Inhibition of NF кB activity by thalidomide through suppression of I кB kinase activity. J. Biol. Chem. 276: 22382–22387
Eriksson T., Bjorkmann S., Hoglund P. (2001). Clinical pharmacology of thalidomide. Eur. J. Clin. Pharmacol. 57: 365–376
Chen A., Zhang L., Xu J., Tang J. (2002). The anti-oxidant (-)-epigallocatechin-3-gallate inhibits activated hepatic stellate cell growth and suppresses acetaldehyde-induced gene expression. Biochem. J. 368: 695–704
Canby A., Friedman S., Gores G.J. (2004). Apoptosis: nexus of liver injury and fibrosis. Hepatology 39: 273–278
Parman T. (1999). Free radical-mediated oxidative DNA damage in the mechanism of thalidomide teratogenicity. Nat. Med. 5: 582–585
Acknowledgements
We gratefully acknowledge the kind provision of HSC-T6 cells by Dr. Scott L. Friedman, Division of Liver Diseases, The Mount Sinai School of Medicine, New York, NY, USA. We also cordially appreciate the supply of thalidomide by the TTY Biopharm Company Ltd. (Taipei, Taiwan). This work was supported in part by grants of the National Science Council, Taiwan (NSC 92-2320-077-006, NSC 93-2320-010-072 and NSC93-2320-010-001).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chong, LW., Hsu, YC., Chiu, YT. et al. Anti-Fibrotic Effects of Thalidomide on Hepatic Stellate Cells and Dimethylnitrosamine-Intoxicated Rats. J Biomed Sci 13, 403–418 (2006). https://doi.org/10.1007/s11373-006-9079-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11373-006-9079-5