Summary
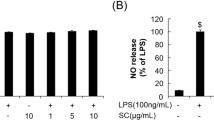
Hydroxyhydroquinone or 1,2,4-benzenetriol (BT) detected in the beverages has a structure that coincides with the water-soluble form of a sesame lignan, sesamol. We previously showed that sesame antioxidants had neuroprotective abilities due to their antioxidant properties and/or inducible nitric oxide synthase (iNOS) inhibition. However, studies show that BT can induce DNA damage through the generation of reactive oxygen species (ROS). Therefore, we were interested to investigate the neuroprotective effect of BT in vitro and in vivo. The results showed that instead of enhancing free radical generation, BT dose-dependently (10–100 μM) attenuated nitrite production, iNOS mRNA and protein expression in lipopolysaccharide (LPS)-stimulated murine BV-2 microglia. BT significantly reduced LPS-induced NF-κB and p38 MAPK activation. It also significantly reduced the generation of ROS in H2O2-induced BV-2 cells and in H2O2-cellfree conditions. The neuroprotective effect of BT was further demonstrated in the focal cerebral ischemia model of Sprague–Dawley rat. Taken together, the inhibition of LPS-induced nitrite production might be due to the suppression of NF-κB, p38 MAPK signal pathway and the ROS scavenging effect. These effects might help to protect neurons from the ischemic injury.
Article PDF
Similar content being viewed by others
References
Hou R.C., Wu C.C., Yang C.H. and Jeng K.C. (2004) Protective effects of sesamin and sesamolin on murine BV-2 microglia cell line under hypoxia. Neurosci. Lett. 367: 10–13
Hou R.C., Huang H.M., Tzen J.T. and Jeng K.C. (2003) Protective effects of sesamin and sesamolin on hypoxic neuronal and PC12 cells. J. Neurosci. Res. 74:123–133
3. Hiramoto K., Li X., Makimoto M., Kato T. and Kikugawa K. (1998) Identification of hydroxyhydroquinone in coffee as a generator of reactive oxygen species that break DNA single strands. Mutat. Res. 419:43–51
4. Greenlee W.F., Chism J.P. and Rickert D.E. (1981) A novel method for the separation and quantitation of benzene metabolites using high-pressure liquid chromatography. Anal. Biochem. 112:367–370
5. Inoue O., Seiji K., Nakatsuka H. et al. (1989) Excretion of 1,2,4-benzenetriol in the urine of workers exposed to benzene. Br. J. Ind. Med. 46:559–565
6. Qu Q., Melikian A.A., Li G. et al. (2000) Validation of biomarkers in humans exposed to benzene: urine metabolites. Am. J. Ind. Med. 37:522–531
7. Hiramoto K., Kida T. and Kikugawa K. (2002) Increased urinary hydrogen peroxide levels caused by coffee drinking. Biol. Pharm. Bull. 25:1467–1471
8. Halliwell B., Long L.H., Yee T.P., Lim S., Kelly R. (2004) Establishing biomarkers of oxidative stress: the measurement of hydrogen peroxide in human urine. Curr. Med. Chem. 11:1085–1092
9. Li A.S., Bandy B., Tsang S. and Davison A.J. (2001) DNA breakage induced by 1,2,4-benzenetriol: relative contributions of oxygen-derived active species and transition metal ions. Free Radic. Biol. Med. 30:943–956
10. Akagawa M., Shigemitsu T. and Suyama K. (2003) Production of hydrogen peroxide by polyphenols and polyphenol-rich beverages under quasi-physiological conditions. Biosci. Biotechnol. Biochem. 67:2632–2640
11. Huang H. and Manton K.G. (2004) The role of oxidative damage in mitochondria during aging:a review. Front Biosci. 9:1100–1117
12. Droge W. (2002) Free radicals in the physiological control of cell function. Physiol. Rev. 82:47–95
13. Rao G.S., Siddiqui S.M., Pandya K.P. and Shanker R. (1988) Relative toxicity of metabolites of benzene in mice. Vet. Hum. Toxicol. 30:517–520
14. Laskin J.D., Rao N.R., Punjabi C.J., Laskin D.L. and Synder R. (1995) Distinct actions of benzene and its metabolites on nitric oxide production by bone marrow leukocytes. J. Leukoc. Biol. 57:422–426
15. Wang M.J., Lin W.W., Chen H.L. et al. (2002) Silymarin protects dopaminergic neurons against lipopolysaccharide-induced neurotoxicity by inhibiting microglia activation. Eur. J. Neurosci. 16:2103–2112
16. Mi H., Hiramoto K., Kujirai K., Ando K., Ikarashi Y. and Kikugawa K. (2001) Effect of food reductones, 2,5-dimethyl-4-hydroxy-3(2H)-furanone (DMHF) and hydroxyhydroquinone (HHQ), on lipid peroxidation and type IV and I allergy responses of mouse. J. Agric. Food Chem. 49:4950–4955
17. Wiemels J. and Smith M.T. (1999) Enhancement of myeloid cell growth by benzene metabolites via the production of active oxygen species. Free Radic. Res. 30:93–103
18. Hedli C.C., Rao N.R., Reuhl K.R., Witmer C.M. and Snyder R. (1996) Effects of benzene metabolite treatment on granulocytic differentiation and DNA adduct formation in HL-60 cells. Arch.Toxicol. 70:135–144
18. Hedli C.C., Rao N.R., Reuhl K.R., Witmer C.M. and Snyder R. (1996) Effects of benzene metabolite treatment on granulocytic differentiation and DNA adduct formation in HL-60 cells. Arch.Toxicol. 70:135–144
19. Hou R.C., Chen H.L., Tzen J.T. and Jeng K.C. (2003) Effect of sesame antioxidants on LPS-induced NO production by BV2 microglial cells. Neuroreport 14:1815–1819
20. Doepker C.L., Dumont K.W., O’Donoghue J. and English J.C. (2000) Lack of induction of micronuclei in human peripheral blood lymphocytes treated with hydroquinone. Mutagenesis 15:479–487
21. Li A.S., Bandy B., Tsang S.S. and Davison A.J. (2000) DNA-breaking versus DNA- protecting activity of four phenolic compounds in vitro. Free Radic. Res. 33:551–566
22. Zhang L., Robertson M.L., Kolachana P. et al. (1993) Benzene metabolite, 1,2,4-benzenetriol, induces micronuclei and oxidative DNA damage in human lymphocytes and HL60 cells. Environ. Mol. Mutagen. 21:339–348
23. Peterson P.K., Hu S., Anderson W.R. and Chao C.C. (1994) Nitric oxide production and neurotoxicity mediated by activated microglia from human versus mouse brain. J. Infect. Dis. 170:457–460
24. Lu F., Selak M., O’Connor J. et al. (2000) Oxidative damage to mitochondrial DNA and activity of mitochondrial enzymes in chronic active lesions of multiple sclerosis. J. Neurol. Sci. 177:95–103
25. Chao C.C., Hu S., Molitor T.W., Shaskan E.G. and Peterson P.K. (1992) Activated microglia mediate neuronal cell injury via a nitric oxide mechanism. J. Immunol. 149:2736–2741
26. Sun A.Y. and Chen Y.M. (1998) Oxidative stress and neurodegenerative disorders. J. Biomed. Sci. 5:401–414
27. Rao N.R. and Snyder R. (1995) Oxidative modifications produced in HL-60 cells on exposure to benzene metabolites. J. Appl. Toxicol. 15:403–409
28. Manning B.W., Adams D.O. and Lewis J.G. (1994) Effects of benzene metabolites on receptor-mediated phagocytosis and cytoskeletal integrity in mouse peritoneal macrophages. Toxicol. Appl. Pharmacol. 126:214–223
29. Tikka T.M. and Koistinaho J.E. (2001) Minocycline provides neuroprotection against N-methyl-D-aspartate neurotoxicity by inhibiting microglia. J. Immunol. 166:7527–7533
30. Maruyama M., Sudo T., Kasuya Y., Shiga T., Hu B. and Osada H. (2000) Immunolocalization of p38 MAP kinase in mouse brain. Brain Res. 887:350–358
31. Woo M.S,, Jang P.G., Park J.S., Kim W.K., Joh T.H. and Kim H.S. (2003) Selective modulation of lipopolysaccharide-stimulated cytokine expression and mitogen-activated protein kinase pathways by dibutyryl-cAMP in BV2 microglial cells. Brain Res. Mol. Brain Res. 113:86–96
32. Bodles A.M. and Barger S.W. (2005) Secreted beta-amyloid precursor protein activates microglia via JNK and p38-MAPK. Neurobiol. Aging 26:9–16
33. Wadsworth T.L and Koop D.R. (2001) Effects of Ginkgo biloba extract (EGb 761) and quercetin on lipopolysaccharide-induced release of nitric oxide. Chem. Biol. Interact. 137:43–58
34. Bastianetto S. and Quirion R. (2004) Natural antioxidants and neurodegenerative diseases. Front. Biosci. 9:3447–3452
35. Rosenstock T.R., Carvalho A.C., Jurkiewicz A., Frussa-Filho R. (2004) and Smaili S.S., Mitochondrial calcium, oxidative stress and apoptosis in a neurodegenerative disease model induced by 3-nitropropionic acid. J. Neurochem. 88:1220–1228
36. Anderson M.F., Nilsson M., Eriksson P.S. and Sims N.R. (2004) Glutathione monoethyl ester provides neuroprotection in a rat model of stroke. Neurosci. Lett. 354:163–165
37. Park J.Y., Kim E.J., Kwon K.J. et al. (2004) Neuroprotection by fructose-1,6- bisphosphate involves ROS alterations via p38 MAPK/ERK. Brain Res. 1026:295–301
38. Katoh S., Mitsui Y., Kitani K. and Suzuki T. (1999) Hyperoxia induces the neuronal differentiated phenotype of PC12 cells via a sustained activity of mitogen-activated protein kinase induced by Bcl-2. Biochem. J. 338:465–470
39. Tanaka S., Takehashi M., Matoh N. et al. (2002) Generation of reactive oxygen species and activation of NF-κB by non-Aβ component of Alzheimer’s disease amyloid. J. Neurochem. 82:305–315
41. Bhat N.R., Zhang P., Lee J.C. and Hogan E.L. (1998) Extracellular signal-regulated kinase and p38 subgroups of mitogen-activated protein kinases regulate inducible nitric oxide synthase and tumor necrosis factor-α gene expression in endotoxin-stimulated primary glial cultures. J. Neurosci. 18:1633–1641
41. Haddad J.J. and Land S.C. (2002) Redox/ROS regulation of lipopolysaccharide- induced mitogen-activated protein kinase (MAPK) activation and MAPK- mediated TNF-α biosynthesis. Br. J. Pharmacol. 135:520–536
42. Li Y., Liu L., Barger S.W., Mrak R.E. and Griffin W.S. (2001) Vitamin E suppression of microglial activation is neuroprotective. J. Neurosci. Res. 66:163–170
43. Egger T., Hammer A., Wintersperger A., Goti D., Malle E. and Sattler W. (2001) Modulation of microglial superoxide production by alpha-tocopherol in vitro: attenuation of p67(phox) translocation by a protein phosphatase-dependent pathway. J. Neurochem. 79:1169–1182
44. Li Y., Liu L., Barger S.W., Mrak R.E. and Griffin W.S. (2001) Vitamin E suppression of microglial activation is neuroprotective. J. Neurosci. Res. 66:163–170
45. Nicholas R.S., Compston A. and Brown D.R. (2001) Inhibition of tumour necrosis factor-α (TNFα)-induced NF-κB p52 converts the metabolic effects of microglial-derived TNFα on mouse cerebellar neurons to neurotoxicity. J. Neurochem. 76:1431–1438
46. Petrova T.V., Akama K.T. and Van Eldik L.J. (1999) Cyclopentenone prostaglandins suppress activation of microglia: down-regulation of inducible nitric-oxide synthase by 15-deoxy-Δ12,14−prostaglandin J2. Proc. Natl. Acad. Sci. USA 96:4668–4673
47. Shen Y., Shen H.M., Shi C.Y. and Ong C.N. (1996) Benzene metabolites enhance reactive oxygen species generation in HL60 human leukemia cells. Hum. Exp. Toxicol. 15:422–427
48. Winn L.M. (2003) Homologous recombination initiated by benzene metabolites:a potential role of oxidative stress. Toxicol. Sci. 72:143–149
Acknowledgements
This study was supported by NSC912320B075A003 grant from the National Science Council of ROC (K.C.J.).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hou, R.CW., Chen, YS., Chen, CH. et al. Protective effect of 1,2,4-benzenetriol on LPS-induced NO production by BV2 microglial cells. J Biomed Sci 13, 89–99 (2006). https://doi.org/10.1007/s11373-005-9039-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11373-005-9039-5