Abstract
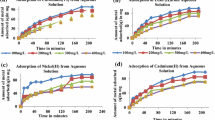
The study examines the adsorption capabilities of an environmentally friendly activated carbon derived from a novel activating agent, i.e., an edible alkali prepared from black gram plant ash, for the removal of Cr(III) and Cr(VI) ions from an aqueous environment. The results of the systematic research show impressive removal efficiencies of 95.12% for Cr(III) ions and 99.6% for Cr(VI) ions. The kinetics and equilibrium data of the adsorption process confirm to the pseudo-second-order kinetics and Freundlich isotherm model. The thermodynamic analysis reveals the adsorption process as feasible and spontaneous across the temperature range of 298–313 K. The mechanism entails electrostatic attraction and adsorption of Cr(III) and Cr(VI) ions on oppositely charged surfaces and the participation of oxygen-containing functional groups on WHAC-BGA surface in the reduction of Cr(VI) to Cr(III). This study provides valuable insights for optimizing strategies to combat chromium contamination in water sources, offering a sustainable solution with the potential for real-world application.







Similar content being viewed by others
Data availability
Date will be made available on request.
Abbreviations
- WH :
-
Water hyacinth
- WHAC-BGA :
-
Activated carbon of Water Hyacinth by using Black Gram Plant Ash as the activating agent
- C 0 :
-
Initial concentration
- C t :
-
Concentration at pre-specified time
- q e :
-
Equilibrium adsorption capacity
- V :
-
Volume of solution
- M :
-
Mass of adsorbent
- q t :
-
Adsorption capacity at any time
- C e :
-
Equilibrium concentration
- q 0 :
-
Maximum monolayer adsorption capacity
- K L :
-
Langmuir constant
- R L :
-
Dimensionless separation parameter
- K f :
-
Freundlich constant
- n f :
-
Freundlich constant
- A T :
-
Equilibrium binding constant
- b T :
-
Temkin constant
- K DR :
-
Energy of biosorption
- qm :
-
Maximum monolayer adsorption capacity
- E :
-
Mean free energy
- k 1 :
-
Pseudo-first order rate constant
- k 2 :
-
Pseudo-second order rate constant
- k p :
-
Intraparticle diffusion rate constant
- k id :
-
Adsorption rate constant
- K 0 :
-
Equilibrium constant
- ∆H 0 :
-
Standard change in enthalpy
- ∆S 0 :
-
Standard change in entropy
- ∆G 0 :
-
Standard change in Gibb’s free energy
References
Adebayo GB, Adegoke HI, Fauzeeyat S (2020) Adsorption of Cr(VI) ions onto goethite, activated carbon and their composite: kinetic and thermodynamic studies. Appl Water Sci 10:213
Al-Musawi TJ, McKay G, Kadhim A, Joybari MM, Balarak D (2022) Activated carbon prepared from hazelnut shell waste and magnetized by Fe3O4 nanoparticles for highly efficient adsorption of fluoride. Biomass Conversion and Biorefinery, pp 1–16
Anjum A, Mazari SA, Hashmi Z, Jatoi AS, Abro R, Bhutto AW, Mubarak NM, Dehghani MH, Karri RR, Mahvi AH, Nasseri S (2023) A review of novel green adsorbents as a sustainable alternative for the remediation of chromium (VI) from water environments. Heliyon 9:e15575
Asgari G, Roshani B, Ghanizadeh G (2012) The investigation of kinetic and isotherm of fluoride adsorption onto functionalize pumice stone. J Hazard Mater 217–218:123–132
Assadi A, Dehghani MH, Rastkari N, Nasseri S, Mahvi AHJEPE (2012) Photocatalytic reduction of hexavalent chromium in aqueous solutions with zinc oxide nanoparticles and hydrogen peroxide. Environ Prot Eng 38:5–16
Babapour M, HadiDehghani M, Alimohammadi M, MoghadamArjmand M, Salari M, Rasuli L, Mubarak NM, Ahmad Khan N (2022) Adsorption of Cr(VI) from aqueous solution using mesoporous metal-organic framework-5 functionalized with the amino acids: Characterization, optimization, linear and nonlinear kinetic models. J Mol Liq 345:117835
Babel S, Kurniawan TA (2004) Cr(VI) removal from synthetic wastewater using coconut shell charcoal and commercial activated carbon modified with oxidizing agents and/or chitosan. Chemosphere 54:951–967
Barrera H, Ureña-Núñez F, Bilyeu B, Barrera-Díaz C (2006) Removal of chromium and toxic ions present in mine drainage by Ectodermis of Opuntia. J Hazard Mater 136:846–853
Biswas K, Saha SK, Ghosh UC (2007) Adsorption of Fluoride from Aqueous Solution by a Synthetic Iron(III)−Aluminum(III) Mixed Oxide. Ind Eng Chem Res 46:5346–5356
Cavaco SA, Fernandes S, Augusto CM, Quina MJ, Gando-Ferreira LM (2009) Evaluation of chelating ion-exchange resins for separating Cr(III) from industrial effluents. J Hazard Mater 169:516–523
Chen S, Yue Q, Gao B, Xu X (2010) Equilibrium and kinetic adsorption study of the adsorptive removal of Cr(VI) using modified wheat residue. J Colloid Interface Sci 349:256–264
Chen Y, An D, Sun S, Gao J, Qian L (2018) Reduction and removal of chromium VI in water by powdered activated carbon. Materials 11(2):269
Cimino G, Passerini A, Toscano G (2000) Removal of toxic cations and Cr(VI) from aqueous solution by hazelnut shell. Water Res 34:2955–2962
Debnath S, Ghosh UC (2008) Kinetics, isotherm and thermodynamics for Cr(III) and Cr(VI) adsorption from aqueous solutions by crystalline hydrous titanium oxide. J Chem Thermodyn 40:67–77
Dehghani MH, Taher MM, Bajpai AK, Heibati B, Tyagi I, Asif M, Agarwal S, Gupta VK (2015) Removal of noxious Cr (VI) ions using single-walled carbon nanotubes and multi-walled carbon nanotubes. Chem Eng J 279:344–352
Dehghani MH, Heibati B, Asadi A, Tyagi I, Agarwal S, Gupta VK (2016) Reduction of noxious Cr(VI) ion to Cr(III) ion in aqueous solutions using H2O2 and UV/H2O2 systems. J Ind Eng Chem 33:197–200
Dehghani MH, Zarei A, Mesdaghinia A, Nabizadeh R, Alimohammadi M, Afsharnia MJD, Treatment W (2017) Adsorption of Cr (VI) ions from aqueous systems using thermally sodium organo-bentonite biopolymer composite (TSOBC): response surface methodology, isotherm, kinetic and thermodynamic studies. Desalin Water Treat 85:298–312
Demirbaş A (2005) Adsorption of Cr(III) and Cr(VI) Ions from Aqueous Solutions on to Modified Lignin. Energy Sources 27:1449–1455
Deveci H, Kar Y (2013) Adsorption of hexavalent chromium from aqueous solutions by bio-chars obtained during biomass pyrolysis. J Ind Eng Chem 19:190–196
Devi B, Baruah NP, Bharadwaj A, Devi A (2023a) Adsorptive removal of fluoride ions from aqueous solution using activated carbon supported tetrametallic oxide system. Chem Eng Res Des 197:380–391
Devi B, Goswami M, Rabha S, Kalita S, Sarma HP, Devi A (2023b) Efficacious Sorption Capacities for Pb(II) from Contaminated Water: A Comparative Study Using Biowaste and Its Activated Carbon as Potential Adsorbents. ACS Omega 8:15141–15151
Duranoğlu D, Trochimczuk AW, Beker U (2012) Kinetics and thermodynamics of hexavalent chromium adsorption onto activated carbon derived from acrylonitrile-divinylbenzene copolymer. Chem Eng J 187:193–202
El Haddad M (2018) Removal of Basic Fuchsin dye from water using mussel shell biomass waste as an adsorbent: Equilibrium, kinetics, and thermodynamics. J Taibah Univ Sci 10:664–674
Elangovan R, Philip L, Chandraraj K (2008a) Biosorption of chromium species by aquatic weeds: Kinetics and mechanism studies. J Hazard Mater 152:100–112
Elangovan R, Philip L, Chandraraj K (2008b) Biosorption of hexavalent and trivalent chromium by palm flower (Borassus aethiopum). Chem Eng J 141:99–111
Ghiaci M, Kia R, Abbaspur A, Seyedeyn-Azad F (2004) Adsorption of chromate by surfactant-modified zeolites and MCM-41 molecular sieve. Sep Purif Technol 40:285–295
Guo J, Xu WS, Chen YL, Lua AC (2005) Adsorption of NH3 onto activated carbon prepared from palm shells impregnated with H2SO4. J Colloid Interface Sci 281:285–290
Hua C, Zhang R, Bai F, Lu P, Liang X (2017) Removal of chromium (VI) from aqueous solutions using quaternized chitosan microspheres. Chin J Chem Eng 25:153–158
Kalita S, Pathak M, Devi G, Sarma HP, Bhattacharyya KG, Sarma A, Devi A (2017) Utilization of Euryale ferox Salisbury seed shell for removal of basic fuchsin dye from water: equilibrium and kinetics investigation. RSC Adv 7:27248–27259
Khandare D, Mukherjee S (2019) A Review of Metal oxide Nanomaterials for Fluoride decontamination from Water Environment. Mater Today: Proceedings 18:1146–1155
Krishnani KK, Ayyappan S (2006) Heavy Metals Remediation of Water Using Plants and Lignocellulosic Agrowastes. In: Ware GW et al (eds) Reviews of Environmental Contamination and Toxicology: Continuation of Residue Reviews. Springer New York, New York, NY, pp 59–84
Kumar S, Shahnaz T, Selvaraju N, Rajaraman PV (2020) Kinetic and thermodynamic studies on biosorption of Cr(VI) on raw and chemically modified Datura stramonium fruit. Environ Monit Assess 192:248
Kyzas GZ, Tolkou AK, Al Musawi TJ, Mengelizadeh N, Mohebi S, Balarak D (2022) Fluoride removal from water by using green magnetic activated carbon derived from canola stalks. Water Air Soil Pollut 233(10):424
Lalvani SB, Hubner A, Wil TS (2000) Chromium Adsorption by Lignin. Energy Sources 22:45–56
Li J, Lin Q, Zhang X, Yan Y (2009a) Kinetic parameters and mechanisms of the batch biosorption of Cr(VI) and Cr(III) onto Leersia hexandra Swartz biomass. J Colloid Interface Sci 333:71–77
Li Y, Gao B, Wu T, Sun D, Li X, Wang B, Lu F (2009b) Hexavalent chromium removal from aqueous solution by adsorption on aluminum magnesium mixed hydroxide. Water Res 43:3067–3075
Liu Y-X, Yuan D-X, Yan J-M, Li Q-L, Ouyang T (2011) Electrochemical removal of chromium from aqueous solutions using electrodes of stainless steel nets coated with single wall carbon nanotubes. J Hazard Mater 186:473–480
Liu H, Liang S, Gao J, Ngo HH, Guo W, Guo Z, Wang J, Li Y (2014) Enhancement of Cr(VI) removal by modifying activated carbon developed from Zizania caduciflora with tartaric acid during phosphoric acid activation. Chem Eng J 246:168–174
Mallampati R, Xuanjun L, Adin A, Valiyaveettil S (2015) Fruit Peels as Efficient Renewable Adsorbents for Removal of Dissolved Heavy Metals and Dyes from Water. ACS Sustain Chem Eng 3:1117–1124
Miretzky P, Cirelli AF (2010) Cr(VI) and Cr(III) removal from aqueous solution by raw and modified lignocellulosic materials: A review. J Hazard Mater 180:1–19
Mohan D, Pittman CU (2006) Activated carbons and low cost adsorbents for remediation of tri- and hexavalent chromium from water. J Hazard Mater 137:762–811
Qi W, Zhao Y, Zheng X, Ji M, Zhang Z (2016) Adsorption behavior and mechanism of Cr(VI) using Sakura waste from aqueous solution. Appl Surf Sci 360:470–476
Qiu Y, Zhang Q, Gao B, Li M, Fan Z, Sang W, Hao H, Wei X (2020) Removal mechanisms of Cr(VI) and Cr(III) by biochar supported nanosized zero-valent iron: Synergy of adsorption, reduction and transformation. Environ Pollut 265:115018
Rahman N, Haseen U (2014) Equilibrium Modeling, Kinetic, and Thermodynamic Studies on Adsorption of Pb(II) by a Hybrid Inorganic-Organic Material: Polyacrylamide Zirconium(IV) Iodate. Ind Eng Chem Res 53:8198–8207
Rajapaksha AU, Alam MS, Chen N, Alessi DS, Igalavithana AD, Tsang DCW, Ok YS (2018) Removal of hexavalent chromium in aqueous solutions using biochar: Chemical and spectroscopic investigations. Sci Total Environ 625:1567–1573
Ren B, Wang K, Zhang B, Li H, Niu Y, Chen H, Zhang H (2019) Adsorption behavior of PAMAM dendrimers functionalized silica for Cd (II) from aqueous solution: Experimental and theoretical calculation. J Taiwan Inst Chem Eng 101:80–91
Sapna RS, Nair M, Kumar D (2018) Trimetallic oxide entrapped in alginate polymeric matrix employed for adsorption studies of fluoride. Surf Interfaces 13:112–132
Sarin V, Singh TS, Pant KK (2006) Thermodynamic and breakthrough column studies for the selective sorption of chromium from industrial effluent on activated eucalyptus bark. Biores Technol 97:1986–1993
Selvaraj K, Manonmani S, Pattabhi S (2003) Removal of hexavalent chromium using distillery sludge. Biores Technol 89:207–211
Shepherd CM, Jones RL (1971). Hexavalent chromium: Toxicological effects and means for removal from aqueous solution. Naval Research Laboratory, Washington DC, USA, p 0021
Taiwo AF, Chinyere NJ (2016) Sorption Characteristics for Multiple Adsorption of Heavy Metal Ions Using Activated Carbon from Nigerian Bamboo. J Mater Sci Chem Eng 04:39–48
Valentin-Reyes J, Garcia-Reyes RB, Garcia-Gonzalez A, Soto-Regalado E, Cerino-Cordova F (2019) Adsorption mechanisms of hexavalent chromium from aqueous solutions on modified activated carbons. J Environ Manag 236:815–822
Wan Ngah WS, Hanafiah MAKM (2008) Adsorption of copper on rubber (Hevea brasiliensis) leaf powder: Kinetic, equilibrium and thermodynamic studies. Biochem Eng J 39:521–530
Wang XS, Chen LF, Li FY, Chen KL, Wan WY, Tang YJ (2010) Removal of Cr (VI) with wheat-residue derived black carbon: Reaction mechanism and adsorption performance. J Hazard Mater 175:816–822
Wang H, Wang W, Zhou S, Gao X (2023) Adsorption mechanism of Cr(VI) on woody-activated carbons. Heliyon 9:e13267
Zeng H, Zeng H, Zhang H, Shahab A, Zhang K, Lu Y, ..., Ullah H (2021) Efficient adsorption of Cr (VI) from aqueous environments by phosphoric acid activated eucalyptus biochar. J Clean Prod 286:124964
Zhang H, Tang Y, Cai D, Liu X, Wang X, Huang Q, Yu Z (2010) Hexavalent chromium removal from aqueous solution by algal bloom residue derived activated carbon: equilibrium and kinetic studies. J Hazard Mater 181:801–808
Zhang Y-J, Ou J-L, Duan Z-K, Xing Z-J, Wang Y (2015) Adsorption of Cr(VI) on bamboo bark-based activated carbon in the absence and presence of humic acid. Colloids Surf, A 481:108–116
Zhou X, Korenaga T, Takahashi T, Moriwake T, Shinoda S (1993) A process monitoring/controlling system for the treatment of wastewater containing chromium(VI). Water Res 27:1049–1054
Acknowledgements
The authors extend their sincere appreciation to the Director of the Institute of Advanced Study in Science and Technology (IASST), Guwahati under the Department of Science & Technology (DST), Government of India for the successful completion of the work. The authors would also like to express their gratitude to the Sophisticated Analytical Instrumentation Centre (SAIC), IASST for generously providing access to essential instrumental resources. Furthermore, we acknowledge the valuable assistance of CSIR-NEIST in conducting the X-ray Photoelectron Spectroscopy (XPS) analysis, which greatly contributed to our research efforts.
Author information
Authors and Affiliations
Contributions
Bhaswati Devi: Conceptualization, investigation, visualization, writing, review and editing. Manisha Goswami: Investigation. Arundhuti Devi: Conceptualization, writing, review, editing and supervision.
Corresponding author
Ethics declarations
Ethics approval
“All authors have read, understood, and have complied as applicable with the statement on “Ethical responsibilities of Authors” as found in the Instructions for Authors”.
Consent for publication
All authors and the institute where the analysis was done have the consent for the publication and have no objections.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Tito Roberto Cadaval Jr
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Devi, B., Goswami, M. & Devi, A. Entrapment behaviours of trivalent and hexavalent chromium from aqueous medium using edible alkali-derived activated carbon of Eichhornia crassipes (water hyacinth). Environ Sci Pollut Res 31, 6025–6039 (2024). https://doi.org/10.1007/s11356-023-31545-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-023-31545-x