Abstract
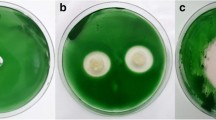
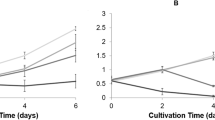
Cyanobacteria massive proliferations are common in freshwater bodies worldwide, causing adverse effects on aquatic ecosystems and public health. Numerous species develop blooms. Most of them correspond to the toxic microcystin-producing cyanobacterium Microcystis aeruginosa. Microorganisms recovered from Antarctic environment can be considered an unexploited source of antimicrobial compounds. Data about their activity against cyanobacteria are scant or inexistent. This study aimed to evaluate the capacity of Antarctic bacteria to inhibit the proliferation of M. aeruginosa BCPUSP232 and to degrade microcystin-LR (MC-LR). Cell-free extracts of seventy-six bacterial strains were initially tested for antimicrobial activity. Unidentified (UN) strains 62 and ES7 and Psychromonas arctica were able to effectively lyse M. aeruginosa. Eight strains showed MIC ranging from 0.55 to 3.00 mg mL−1, with ES7 showing the best antimicrobial activity. Arthrobacter sp. 443 and UN 383 were the most efficient in degrading MC-LR, with 24.87 and 23.85% degradation, respectively. To our knowledge, this is the first report of antimicrobial and MC-LR degradation activities by Antarctic bacteria, opening up perspectives for their future application as an alternative or supporting approach to help mitigate cyanobacterial blooms.



Similar content being viewed by others
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Anan’Ina LN, Yastrebova OV, Demakov VA, Plotnikova EG (2011) Naphthalene-degrading bacteria of the genus Rhodococcus from the Verkhnekamsk salt mining region of Russia Antonie van Leeuwenhoek. Int J Gen Mol Microbiol 100:309–316. https://doi.org/10.1007/s10482-011-9580-3
Bittencourt-Oliveira MC, Oliveira MC, Pinto E (2011) Diversidade de genótipos produtores de microcistinas em linhagens brasileiras de microcystis (Cyanobacteria). Braz J Biol 71:209–216. https://doi.org/10.1590/S1519-69842011000100030
Bourne DG, Jones GJ, Blakeley RL, Jones A, Negri AP, Riddles P (1996) Enzymatic pathway for the bacterial degradation of the cyanobacterial cyclic peptide toxin microcystin LR. Appl Environ Microbiol 62:4086–4094
Bourne DG, Riddles P, Jones GJ, Smith W, Blakeley RL (2001) Characterisation of a gene cluster involved in bacterial degradation of the cyanobacterial toxin microcystin LR. Environ Toxicol 16:523–534. https://doi.org/10.1002/tox.10013
Braun A, Pfeiffer T (2002) Cyanobacterial blooms as the cause of a Pleistocene large mammal assemblage. Paleobiology 28:139–154. https://doi.org/10.1666/0094-8373(2002)028<0139:cbatco>2.0.co;2
Caiola MG, Pellegrini S (1984) Lysis of Microcystis aeruginosa (Kütz) by Bdellovibrio-like bacteria. J Phycol 20:471–475. https://doi.org/10.1111/J.0022-3646.1984.00471.X
Chen WM, Sheu FS, Sheu SY (2011) Novel l-amino acid oxidase with algicidal activity against toxic cyanobacterium Microcystis aeruginosa synthesized by a bacterium Aquimarina sp. Enzym Microb Technol 49:372–379. https://doi.org/10.1016/j.enzmictec.2011.06.016
Codd GA, Bell SG, Brooks WP (1989) Cyanobacterial Toxins in Water. Water Sci Technol 21:1–13. https://doi.org/10.2166/wst.1989.0071
DAFT MJ, McCORD SB, STEWART WDP (1975) Ecological studies on algal-lysing bacteria in fresh waters. Freshw Biol 5:577–596. https://doi.org/10.1111/j.1365-2427.1975.tb00157.x
Demuez M, González-Fernández C, Ballesteros M (2015) Algicidal microorganisms and secreted algicides: new tools to induce microalgal cell disruption. Biotechnol Adv 33:1615–1625. https://doi.org/10.1016/j.biotechadv.2015.08.003
Dias E, Oliveira M, Jones-Dias D, Vasconcelos V, Ferreira E, Manageiro V, Caniça M (2015) Assessing the antibiotic susceptibility of freshwater Cyanobacteria spp. Front Microbiol 6:799. https://doi.org/10.3389/fmicb.2015.00799
Ding Q, Liu K, Xu K, Sun R, Zhang J, Yin L, Pu Y (2018) Further understanding of degradation pathways of microcystin-LR by an indigenous sphingopyxis sp. In environmentally relevant pollution concentrations. Toxins (Basel) 10:536. https://doi.org/10.3390/toxins10120536
Eloff J (1998) A Sensitive and Quick Microplate Method to Determine the Minimal Inhibitory Concentration of Plant Extracts for Bacteria. Planta Med 64:711–713. https://doi.org/10.1055/s-2006-957563
Euzeby JP (1997) List of bacterial names with standing in nomenclature: a folder available on the Internet. Int J Syst Bacteriol:590–592
Fuentes JL, Garbayo I, Cuaresma M, Montero Z, González-del-Valle M, Vílchez C (2016) Impact of microalgae-bacteria interactions on the production of algal biomass and associated compounds. Mar Drugs:14. https://doi.org/10.3390/md14050100
Gantar M, Berry JP, Thomas S, Wang M, Perez R, Rein KS (2008) Allelopathic activity among Cyanobacteria and microalgae isolated from Florida freshwater habitats. FEMS Microbiol Ecol 64:55–64. https://doi.org/10.1111/j.1574-6941.2008.00439.x
Gumbo JR, Cloete TE (2011) The mechanism of Microcystis aeruginosa death upon exposure to Bacillus mycoides. 36:881–886. https://doi.org/10.1016/j.pce.2011.07.050
Hammer Ø, Harper D (1999) PAST: Paleontological statistics software package for education and data analysis. Palaeontol Electron 4:9
Hitzfeld BC, Lampert C, Spaeth N et al (2000) Toxin production in cyanobacterial mats from ponds on the McMurdo Ice Shelf, Antarctica. Toxicon 38:1731–1748. https://doi.org/10.1016/S0041-0101(00)00103-3
Hodgson E (2012) Toxins and venoms. In: Progress in Molecular Biology and Translational Science. Elsevier B.V., In, pp 373–415
Jing W, Sui G, Liu S (2014) Characteristics of a microcystin-LR biodegrading bacterial isolate: Ochrobactrum sp. FDT5. Bull Environ Contam Toxicol 92:119–122. https://doi.org/10.1007/s00128-013-1170-9
Jones GJ, Bourne DG, Blakeley RL, Doelle H (1994) Degradation of the cyanobacterial hepatotoxin microcystin by aquatic bacteria. Nat Toxins 2:228–235. https://doi.org/10.1002/nt.2620020412
Jung SW, Kim B-H, Katano T, Kong DS, Han MS (2008) Pseudomonas fluorescens HYK0210-SK09 offers species-specific biological control of winter algal blooms caused by freshwater diatom Stephanodiscus hantzschii. J Appl Microbiol 105:186–195. https://doi.org/10.1111/j.1365-2672.2008.03733.x
Jungblut AD, Wilbraham J, Banack SA, Metcalf JS, Codd GA (2018) Microcystins, BMAA and BMAA isomers in 100-year-old Antarctic cyanobacterial mats collected during Captain R.F. Scott’s Discovery Expedition. Eur J Phycol 53:115–121. https://doi.org/10.1080/09670262.2018.1442587
Kang YH, Park CS, Han MS (2012) Pseudomonas aeruginosa UCBPP-PA14 a useful bacterium capable of lysing Microcystis aeruginosa cells and degrading microcystins. J Appl Phycol 24:1517–1525. https://doi.org/10.1007/s10811-012-9812-6
Karlson B, Cusack C, Bresnan E (2010) Microscopic and molecular methods for quantitative phytoplankton analysis. Intergovernmental Oceanographic Commission Manuals and Guides. France, UNESCO, pp 110pp
Kennedy J, Marchesi JR, Dobson ADW (2008) Marine metagenomics: strategies for the discovery of novel enzymes with biotechnological applications from marine environments. Microb Cell Factories 7:27. https://doi.org/10.1186/1475-2859-7-27
Kodani S, Imoto A, Mitsutani A, Murakami M (2002) Isolation and identification of the antialgal compound, harmane (1-methyl-β-carboline), produced by the algicidal bacterium, Pseudomonas sp. K44-1. J Appl Phycol 14:109–114. https://doi.org/10.1023/A:1019533414018
Kormas KA, Lymperopoulou DS (2013) Cyanobacterial Toxin Degrading Bacteria: Who Are They? Biomed Res Int 2013:12–12. https://doi.org/10.1155/2013/463894
Kumar P, Hegde K, Brar SK, Cledon M, Kermanshahi-pour A, Roy-Lachapelle A, Galvez-Cloutier R (2018) Biodegradation of microcystin-LR using acclimatized bacteria isolated from different units of the drinking water treatment plant. Environ Pollut 242:407–416. https://doi.org/10.1016/j.envpol.2018.07.008
Lawton LA, Welgamage A, Manage PM, Edwards C (2011) Novel bacterial strains for the removal of microcystins from drinking water. Water Sci Technol 63:1137–1142. https://doi.org/10.2166/wst.2011.352
Lee YK, Ahn CY, Kim HS, Oh HM (2010) Cyanobactericidal effect of Rhodococcus sp. isolated from eutrophic lake on Microcystis sp. Biotechnol Lett 32:1673–1678. https://doi.org/10.1007/s10529-010-0350-5
Lemes G, Kist LW, Bogo MR, Yunes JS (2015) Biodegradation of [D-Leu1] microcystin-LR by a bacterium isolated from sediment of Patos Lagoon estuary, Brazil. J Venom Anim Toxins Incl Trop Dis 21:4. https://doi.org/10.1186/s40409-015-0001-3
Li H, Ai H, Kang L, Sun X, He Q (2016) Simultaneous microcystis algicidal and microcystin degrading capability by a single acinetobacter bacterial strain. Environ Sci Technol 50:11903–11911. https://doi.org/10.1021/acs.est.6b03986
Li J, Li R, Li J (2017) Current research scenario for microcystins biodegradation – a review on fundamental knowledge, application prospects and challenges. Sci Total Environ 595:615–632
Lin S, Geng M, Liu X, Tan J, Yang H (2016) On the control of Microcystis aeruginosa and Synechococccus species using an algicidal bacterium, Stenotrophomonas F6, and its algicidal compounds cyclo-(Gly-Pro) and hydroquinone. J Appl Phycol 28:345–355. https://doi.org/10.1007/s10811-015-0549-x
Liu YM, Chen MJ, Wang MH, Jia RB, Li L (2013) Inhibition of Microcystis aeruginosa by the extracellular substances from an Aeromonas sp. J Microbiol Biotechnol 23:1304–1307. https://doi.org/10.4014/jmb.1304.04025
Liu ZZ, Zhu JP, Li M, Xue QQ, Zeng Y, Wang ZP (2014) Effects of freshwater bacterial siderophore on Microcystis and Anabaena. Biol Control 78:42–48. https://doi.org/10.1016/j.biocontrol.2014.07.010
Liu H, Guo X, Liu L, Yan M, Li J, Hou S, Wan J, Feng L (2020) Simultaneous microcystin degradation and Microcystis aeruginosa inhibition with the single enzyme microcystinase A. Environ Sci Technol 54:8811–8820. https://doi.org/10.1021/acs.est.0c02155
Luo J, Wang Y, Tang S, Liang J, Lin W, Luo L (2013) Isolation and identification of algicidal compound from Streptomyces and algicidal mechanism to Microcystis aeruginosa. PLoS One 8:e76444. https://doi.org/10.1371/journal.pone.0076444
Manage PM, Edwards C, Lawton LA (2009a) Biodegradation of microcystin-LR by natural bacterial populations. In: Obayashi Y, Isobe T, Subramanian A et al (eds) Interdisciplinary Studies on Environmental Chemistry - Environmental Research in Asia. TERRAPUB, Tokyo, pp 277–285
Manage PM, Edwards C, Singh BK, Lawton LA (2009b) Isolation and identification of novel microcystin-degrading bacteria. Appl Environ Microbiol 75:6924–6928. https://doi.org/10.1128/AEM.01928-09
Mankiewicz-Boczek J, Gągała I, Jurczak T, Jaskulska A, Pawełczyk J, Dziadek J (2015) Bacteria homologus to Aeromonas capable of microcystin degradation. Open Life Sci 10. https://doi.org/10.1515/biol-2015-0012
Massey IY, Yang F (2020) A mini review on microcystins and bacterial degradation. Toxins (Basel) 12. https://doi.org/10.3390/toxins12040268
Massey IY, Zhang X, Yang F (2018) Importance of bacterial biodegradation and detoxification processes of microcystins for environmental health. J Toxicol Environ Heal - Part B Crit Rev 21:357–369. https://doi.org/10.1080/10937404.2018.1532701
Mello MM e, Soares MCS, Roland F, Lurling M (2012) Growth inhibition and colony formation in the cyanobacterium Microcystis aeruginosa induced by the cyanobacterium Cylindrospermopsis raciborskii. J Plankton Res 34:987–994. https://doi.org/10.1093/plankt/fbs056
Meriluoto J, Spoof L, Codd GA (Eds.) (2017) Handbook of cyanobacterial monitoring and cyanotoxin analysis. John Wiley & Sons. https://doi.org/10.1002/9781119068761
Nakamura N, Nakano K, Sugiura N, Matsumura M (2003) A novel cyanobacteriolytic bacterium, Bacillus cereus, isolated from a Eutrophic Lake. J Biosci Bioeng 95:179–184
Ndlela LL, Oberholster PJ, Van Wyk JH, Cheng PH (2018) Bacteria as biological control agents of freshwater cyanobacteria: is it feasible beyond the laboratory? Appl Microbiol Biotechnol 102:9911–9923. https://doi.org/10.1007/s00253-018-9391-9
Núñez-Montero K, Barrientos L (2018) Advances in Antarctic research for antimicrobial discovery: a comprehensive narrative review of bacteria from Antarctic environments as potential sources of novel antibiotic compounds against human pathogens and microorganisms of industrial importance. Antibiotics 7:90. https://doi.org/10.3390/antibiotics7040090
Ozaki K, Ohta A, Iwata C, Horikawa A, Tsuji K, Ito E, Ikai Y, Harada KI (2008) Lysis of cyanobacteria with volatile organic compounds. Chemosphere 71:1531–1538. https://doi.org/10.1016/j.chemosphere.2007.11.052
Parte AC, Sardà Carbasse J, Meier-Kolthoff JP, Reimer LC, Göker M (2020) List of prokaryotic names with standing in nomenclature (LPSN) moves to the DSMZ. Int J Syst Evol Microbiol 70:5607–5612. https://doi.org/10.1099/ijsem.0.004332
Reddy GSN, Prakash JSS, Vairamani M et al (2002) Planococcus antarcticus and Planococcus psychrophilus spp. nov. isolated from cyanobacterial mat samples collected from ponds in Antarctica. Extremophiles 6:253–261. https://doi.org/10.1007/s00792-001-0250-7
Rice EL (1985) Allelopathy — an overview. In: Chemically Mediated Interactions between Plants and Other Organisms. Springer, US, pp 81–105
Schlegel I, Doan NT, de Chazal N, Smith GD (1998) Antibiotic activity of new cyanobacterial isolates from Australia and Asia against green algae and cyanobacteria. J Appl Phycol 10:471–479
Sette LD, de Oliveira VM, Manfio GP (2005) Isolation and characterization of alachlor-degrading actinomycetes from soil. Antonie Van Leeuwenhoek 87:81–89. https://doi.org/10.1007/s10482-004-1129-2
Sigee DC, Glenn R, Andrews MJ, Bellinger EG, Butler RD, Epton HAS, Hendry RD (1999) Biological control of cyanobacteria: principles and possibilities. Hydrobiologia 395(396):161–172. https://doi.org/10.1023/A:1017097502124
Silva TR, Duarte AWF, Passarini MRZ, Ruiz ALTG, Franco CH, Moraes CB, de Melo IS, Rodrigues RA, Fantinatti-Garboggini F, Oliveira VM (2018) Bacteria from Antarctic environments: diversity and detection of antimicrobial, antiproliferative, and antiparasitic activities. Polar Biol 41:1521. https://doi.org/10.1007/s00300-018-2322-5
Uchida H, Kouchiwa T, Watanabe K, et al (1998) A Coupled Assay System for the Lysis of Cyanobacteria. Japanese J Water Treat Biol 34:67–75. https://doi.org/10.2521/jswtb.34.67
Van Wichelen J, Vanormelingen P, Codd GA, Vyverman W (2016) The common bloom-forming cyanobacterium Microcystis is prone to a wide array of microbial antagonists. Harmful Algae 55:97–111. https://doi.org/10.1016/J.HAL.2016.02.009
Wang J, Wang C, Li Q, Shen M, Bai P, Li J, Lin Y, Gan N, Li T, Zhao J (2019) Microcystin-LR degradation and gene regulation of microcystin-degrading novosphingobium sp. THN1 at different carbon concentrations. Front Microbiol 10, 10. https://doi.org/10.3389/fmicb.2019.01750
Wang M, Chen S, Zhou W, Yuan W, Wang D (2020) Algal cell lysis by bacteria: a review and comparison to conventional methods. Algal Res 46:101794. https://doi.org/10.1016/j.algal.2020.101794
Wells ML, Trainer VL, Smayda TJ, Karlson BSO, Trick CG, Kudela RM, Ishikawa A, Bernard S, Wulff A, Anderson DM, Cochlan WP (2015) Harmful algal blooms and climate change: learning from the past and present to forecast the future. Harmful Algae 49:68–93. https://doi.org/10.1016/j.hal.2015.07.009
Wu X, Joyce EM, Mason TJ (2011) The effects of ultrasound on cyanobacteria. Harmful Algae 10:738–743. https://doi.org/10.1016/J.HAL.2011.06.005
Yamamoto Y, Kouchiwa T, Hodoki Y, Hotta K, Uchida H, Harada KI (1998) Distribution and identification of actinomycetes lysing cyanobacteria in a eutrophic lake. J Appl Phycol 10:391–397. https://doi.org/10.1023/A:1008077414808
Yang F, Wei HY, Li XQ et al (2013) Isolation and characterization of an algicidal bacterium indigenous to lake taihu with a red pigment able to lyse microcystis aeruginosa. Biomed Environ Sci 26:148–154. https://doi.org/10.3967/0895-3988.2013.02.009
Yang C, Hou X, Wu D, Chang W, Zhang X, Dai X, du H, Zhang X, Igarashi Y, Luo F (2020) The characteristics and algicidal mechanisms of cyanobactericidal bacteria, a review. World J Microbiol Biotechnol 36:188
Yoshikawa K, Adachi K, Nishijima M, et al (2000) Cyanoalanine production by marine bacteria on cyanide-free medium and its specific inhibitory activity toward cyanobacteria
Yu Y, Zeng Y, Li J, Yang C, Zhang X, Luo F, Dai X (2019) An algicidal Streptomyces amritsarensis strain against Microcystis aeruginosa strongly inhibits microcystin synthesis simultaneously. Sci Total Environ 650:34–43. https://doi.org/10.1016/j.scitotenv.2018.08.433
Zaffiro A, Rosenblum L, Wendelken S (2016) Method 546: determination of total microcystins and nodularins in drinking water and ambient water by Adda Enzyme-Linked Immunosorbent Assay. 65
Zakhia F, Jungblut AD, Taton A et al (2008) Cyanobacteria in cold ecosystems. Psychrophiles From Biodivers to Biotechnol:121–135. https://doi.org/10.1007/978-3-540-74335-4_8
Zhang H, Yu Z, Huang Q, Xiao X, Wang X, Zhang F, Wang X, Liu Y, Hu C (2011) Isolation, identification and characterization of phytoplankton-lytic bacterium CH-22 against Microcystis aeruginosa. Limnologica 41:70–77. https://doi.org/10.1016/j.limno.2010.08.001
Zhu FP, Han ZL, Duan JL, Shi XS, Wang TT, Sheng GP, Wang SG, Yuan XZ (2019) A novel pathway for the anaerobic biotransformation of microcystin-LR using enrichment cultures. Environ Pollut 247:1064–1070. https://doi.org/10.1016/j.envpol.2019.02.013
Acknowledgements
We thank Ana Carolina Gossen from the Reservoir Division of Itaipu Binacional – Paraguay, for the support during the laboratory experiments; Maria do Carmo Bittencourt-Oliveira from the University of São Paulo, for kindly providing the BCP USP232 strain; to Luiz Henrique Rosa (UFMG-Brazil) for performing the sampling; São Paulo State Research Support Foundation (FAPESP) - Process: 2016/05640-6 and Research Productivity Grant from CNPq (Valéria Maia de Oliveira) – Process: 308955/2016-1)
Funding
This research was supported by Universidade Federal da Integração Latino-Americana – UNILA.
Author information
Authors and Affiliations
Contributions
GRSB analyzed data, designed research, conducted experiments, and wrote the manuscript. SPFB conducted experiments. VMO analyzed data, designed research, and wrote the manuscript. MRZP analyzed data, designed research, and wrote the manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable in this section.
Consent for publication
Not applicable in this section.
Conflict of interest
The authors declare no competing interests.
Additional information
Responsible Editor: Vitor Vasconcelos
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(DOC 121 kb)
Rights and permissions
About this article
Cite this article
Benegas, G.R.S., Bernal, S.P.F., de Oliveira, V.M. et al. Antimicrobial activity against Microcystis aeruginosa and degradation of microcystin-LR by bacteria isolated from Antarctica. Environ Sci Pollut Res 28, 52381–52391 (2021). https://doi.org/10.1007/s11356-021-14458-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-021-14458-5