Abstract
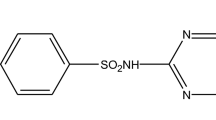
The present study proposes the synthesis and characterization of graphene oxide (GO) and its application in the adsorption of the antibiotic cephalexin (CFX) in aqueous solution. The characterization of graphene oxide was obtained by scanning electron microscopy (SEM), transmission electron microscopy (TEM), and zeta potential. The influence of pH on the batch adsorption process was investigated by analysing adsorption equilibrium isotherms and adsorption kinetics. The images obtained by SEM and TEM presented the typical morphology attributed to GO sheets. The kinetic adsorption tests showed that equilibrium was reached in 420 min, and an adsorption capacity of 164 mg g−1 was obtained. The models that best fit the experimental data were pseudo-second as well as the Langmuir isotherm. Therefore, GO was effective for removing the CFX antibiotic from aqueous solution by using a batch adsorption process.





Similar content being viewed by others
References
Al-Khalisy RS, Al-Haidary AMA, Al-Dujaili AH (2010) Aqueous phase adsorption of cephalexin onto bentonite and activated carbon. Sep Sci Technol 45(9):1286–1294. https://doi.org/10.1080/01496391003689017
Alekseev VG, Nikiforova AA, Markelova SV (2006) Reaction of cefalexine with manganese(II), cobalt(II), nickel(II), zinc(II), and cadmium(II) ions. Russ J Gen Chem 76(9):1468–1470. https://doi.org/10.1134/s1070363206090192
Alharbi TMD, Harvey D, Alsulami IK, Dehbari N, Duan X, Lamb RN, Lawrance WD, Raston CL (2018) Shear stress mediated scrolling of graphene oxide. Carbon 137:419–424. https://doi.org/10.1016/j.carbon.2018.05.040
Ali G, Yusoff M, Chong KF (2016) Graphene : electrochemical production and its energy storage properties. J Eng Appl Sci 11:9712–9717
Ali I, Basheer AA, Mbianda XY, Burakov A, Galunin E, Burakova I, Mkrtchyan E, Tkachev A, Grachev V (2019) Graphene based adsorbents for remediation of noxious pollutants from wastewater. Environ Int 127:160–180. https://doi.org/10.1016/j.envint.2019.03.029
Apul OG, Wang Q, Zhou Y, Karanfil T (2013) Adsorption of aromatic organic contaminants by graphene nanosheets: comparison with carbon nanotubes and activated carbon. Water Res 47(4):1648–1654. https://doi.org/10.1016/j.watres.2012.12.031
Azari A, Salari M, Dehghani MH, Alimohammadi M, Ghaffari H, Sharafi K, Shariatifar N, Baziar M (2017) Efficiency of magnitized graphene oxide nanoparticles in removal of 2,4-dichlorophenol from aqueous solution. Journal of Mazandaran University of Medical Sciences 26:265–281
Bergeron S, Raj B, Nathaniel R, Corbin A, LaFleur G (2017) Presence of antibiotic resistance genes in raw source water of a drinking water treatment plant in a rural community of USA. Int Biodeterior Biodegradation. https://doi.org/10.1016/j.ibiod.2017.05.024
Dehghan A, Zarei A, Jaafari J, Shams M, Mousavi Khaneghah A (2019) Tetracycline removal from aqueous solutions using zeolitic imidazolate frameworks with different morphologies: a mathematical modeling. Chemosphere 217:250–260. https://doi.org/10.1016/j.chemosphere.2018.10.166
Dehghani MH, Faraji M, Mohammadi A, Kamani H (2017a) Optimization of fluoride adsorption onto natural and modified pumice using response surface methodology: isotherm, kinetic and thermodynamic studies. Korean J Chem Eng 34:454–462. https://doi.org/10.1007/s11814-016-0274-4
Dehghani MH, Tajik S, Panahi A, Khezri M, Zarei A, Heidarinejad Z, Yousefi M (2018a) Adsorptive removal of noxious cadmium from aqueous solutions using poly urea-formaldehyde: a novel polymer adsorbent. MethodsX 5:1148–1155. https://doi.org/10.1016/j.mex.2018.09.010
Dehghani MH, Zarei A, Mesdaghinia A, Nabizadeh R, Alimohammadi M, Afsharnia M (2017b) Response surface modeling, isotherm, thermodynamic and optimization study of arsenic (V) removal from aqueous solutions using modified bentonite-chitosan (MBC). Korean J Chem Eng 34:757–767. https://doi.org/10.1007/s11814-016-0330-0
Dehghani MH, Zarei A, Mesdaghinia A, Nabizadeh R, Alimohammadi M, Afsharnia M, McKay G (2018b) Production and application of a treated bentonite–chitosan composite for the efficient removal of humic acid from aqueous solution. Chem Eng Res Des 140:102–115. https://doi.org/10.1016/j.cherd.2018.10.011
Ding S, Sun S, Xu H, Yang B, Liu Y, Wang H, Chen D, Zhang R (2019) Preparation and adsorption property of graphene oxide by using waste graphite from diamond synthesis industry. Mater Chem Phys 221:47–57. https://doi.org/10.1016/j.matchemphys.2018.09.036
Freundlich H (1907) Über die adsorption in lösungen. Z Phys Chem 57(1):385–470. https://doi.org/10.1515/zpch-1907-5723
Freundlich HMF (1906) Over the adsorption in solution. J Phys Chem 57:385–471. https://doi.org/10.3390/ijerph9030970
Ghaedi M, Ansari A, Habibi MH, Asghari AR (2014) Removal of malachite green from aqueous solution by zinc oxide nanoparticle loaded on activated carbon: kinetics and isotherm study. J Ind Eng Chem 20(1):17–28. https://doi.org/10.1016/j.jiec.2013.04.031
Hancu G, Sasebeşi A, Rusu A, Kelemen H, Ciurba A (2015) Study of the Electrophoretic Behavior of Cephalosporins by Capillary Zone Electrophoresis. Adv Pharm Bull 5(2):223–229. https://doi.org/10.15171/apb.2015.031
Ho Y, McKay G, Wase D, Forster C (2000) Study of the sorption of divalent metal ions on to peat. Adsorpt Sci Technol 18(7):639–650. https://doi.org/10.1260/0263617001493693
Ho YS, McKay G (1998) Kinetic models for the sorption of dye from aqueous solution by wood. Process Saf Environ Prot 76(2):183–191. https://doi.org/10.1205/095758298529326
Hu C, Wang S, Sun J, Liu H, Qu J (2016) An effective method for improving electrocoagulation process: optimization of Al13 polymer formation. Colloids Surf A Physicochem Eng Asp 489:234–240. https://doi.org/10.1016/j.colsurfa.2015.10.063
Hu X, Yu Y, Hou W, Zhou J, Song L (2013) Effects of particle size and pH value on the hydrophilicity of graphene oxide. Appl Surf Sci 273:118–121. https://doi.org/10.1016/j.apsusc.2013.01.201
Hummers WS, Offeman RE (1958) Preparation of graphitic oxide. J Am Chem Soc 80(6):1339–1339. https://doi.org/10.1021/ja01539a017
Jafari M, Aghamiri S, Khaghanic G (2011) Batch adsorption of cephalosporins antibiotics from aqueous solution by means of multi-walled carbon nanotubes. World Appl Sci J 14(11):1642–1650
Johnson DW, Dobson BP, Coleman KS (2015) A manufacturing perspective on graphene dispersions. Curr Opin Colloid Interface Sci 20(5):367–382. https://doi.org/10.1016/j.cocis.2015.11.004
Khalili D (2016) Graphene oxide: a promising carbocatalyst for the regioselective thiocyanation of aromatic amines, phenols, anisols and enolizable ketones by hydrogen peroxide/kscn in water. New J Chem 40. https://doi.org/10.1039/c5nj02314a
Khosravi R, Zarei A, Heidari M, Ahmadfazeli A, Vosughi M, Fazlzadeh M (2018) Application of ZnO and TiO2 nanoparticles coated onto montmorillonite in the presence of H2O2 for efficient removal of cephalexin from aqueous solutions. Korean J Chem Eng 35:1000–1008 10.1007/s11814-018-0005-0
Kong D, Liang B, Yun H, Cheng H, Ma J, Cui M, Wang A, Ren N (2015) Cathodic degradation of antibiotics: characterization and pathway analysis. Water Res 72:281–292. https://doi.org/10.1016/j.watres.2015.01.025
Kovtyukhova NI, Ollivier PJ, Martin BR, Mallouk TE, Chizhik SA, Buzaneva EV, Gorchinskiy AD (1999) Layer-by-layer assembly of ultrathin composite films from micron-sized graphite oxide sheets and polycations. Chem Mater 11(3):771–778. https://doi.org/10.1021/cm981085u
Lagergren, S.Y. Zur Theorie der sogenannten Adsorption gelöster Stoffe. (1898)
Langmuir I (1918) The adsorption of gases on plane surfaces of glass, mica and platinum. J Am Chem Soc 40(9):1361–1403. https://doi.org/10.1021/ja02242a004
Legnoverde MS, Basaldella EI (2016) Influence of particle size on the adsorption and release of cephalexin encapsulated in mesoporous silica SBA-15. Mater Lett 181:331–334. https://doi.org/10.1016/j.matlet.2016.06.053
Legnoverde MS, Simonetti S, Basaldella EI (2014) Influence of pH on cephalexin adsorption onto SBA-15 mesoporous silica: theoretical and experimental study. Appl Surf Sci 300:37–42. https://doi.org/10.1016/j.apsusc.2014.01.198
Leili M, Fazlzadeh M, Bhatnagar A (2018) Green synthesis of nano-zero-valent iron from nettle and thyme leaf extracts and their application for the removal of cephalexin antibiotic from aqueous solutions. Environ Technol 39(9):1158–1172. https://doi.org/10.1080/09593330.2017.1323956
Liu H, Liu W, Zhang J, Zhang C, Ren L, Li Y (2011) Removal of cephalexin from aqueous solutions by original and cu(II)/Fe(III) impregnated activated carbons developed from lotus stalks kinetics and equilibrium studies. J Hazard Mater 185(2):1528–1535. https://doi.org/10.1016/j.jhazmat.2010.10.081
Liu W, Xie H, Zhang J, Zhang C (2012) Sorption removal of cephalexin by HNO3 and H2O2 oxidized activated carbons. Sci China Chem 55(9):1959–1967. https://doi.org/10.1007/s11426-011-4488-3
Memon JR, Memon SQ, Bhanger MI, Memon GZ, El-Turki A, Allen GC (2008) Characterization of banana peel by scanning electron microscopy and FT-IR spectroscopy and its use for cadmium removal. Colloids Surf B Biointerfaces 66(2):260–265. https://doi.org/10.1016/j.colsurfb.2008.07.001
Miao M-S, Liu Q, Shu L, Wang Z, Liu Y-Z, Kong Q (2016) Removal of cephalexin from effluent by activated carbon prepared from alligator weed: kinetics, isotherms, and thermodynamic analyses. Process Saf Environ Prot 104:481–489. https://doi.org/10.1016/j.psep.2016.03.017
Mirzaei R, Yunesian M, Nasseri S, Gholami M, Jalilzadeh E, Shoeibi S, Mesdaghinia A (2018) Occurrence and fate of most prescribed antibiotics in different water environments of Tehran. Iran Sci Total Environ 619-620:446–459. https://doi.org/10.1016/j.scitotenv.2017.07.272
Mohseni-Bandpi A, Al-Musawi TJ, Ghahramani E, Zarrabi M, Mohebi S, Vahed SA (2016) Improvement of zeolite adsorption capacity for cephalexin by coating with magnetic Fe3O4 nanoparticles. J Mol Liq 218:615–624. https://doi.org/10.1016/j.molliq.2016.02.092
Mrestani Y, Neubert R, Munk A, Wiese M (1998) Determination of dissociation constants of cephalosporins by capillary zone electrophoresis. J Chromatogr A 803(1):273–278. https://doi.org/10.1016/S0021-9673(97)01213-2
Nasrollahzadeh M, Ganji F, Taghizadeh SM, Daraei B, Loboa EG, Vasheghani-Farahani E (2016) Development and optimization of cephalexin-loaded solid lipid nanoparticles using D-optimal design. Adv Sci Eng Med 8:695–704. https://doi.org/10.1166/asem.2016.1917
Nassar MY, Ahmed IS, Hendy HS (2018) A facile one-pot hydrothermal synthesis of hematite (α-Fe2O3) nanostructures and cephalexin antibiotic sorptive removal from polluted aqueous media. J Mol Liq 271:844–856. https://doi.org/10.1016/j.molliq.2018.09.057
Nazari G, Abolghasemi H, Esmaieli M (2016) Batch adsorption of cephalexin antibiotic from aqueous solution by walnut shell-based activated carbon. J Taiwan Inst Chem Eng 58:357–365. https://doi.org/10.1016/j.jtice.2015.06.006
Özçakır, E., Eskizeybek, V. A facile and effective method for size sorting of large flake Graphene oxide. (2016)
Ramesha GK, Vijaya Kumara A, Muralidhara HB, Sampath S (2011) Graphene and graphene oxide as effective adsorbents toward anionic and cationic dyes. J Colloid Interface Sci 361(1):270–277. https://doi.org/10.1016/j.jcis.2011.05.050
Rashtbari Y, Hazrati S, Afshin S, Fazlzadeh M, Vosoughi M (2018) Data on cephalexin removal using powdered activated carbon (PPAC) derived from pomegranate peel. Data Brief 20:1434–1439. https://doi.org/10.1016/j.dib.2018.08.204
Samarghandi MR, Al-Musawi TJ, Mohseni-Bandpi A, Zarrabi M (2015) Adsorption of cephalexin from aqueous solution using natural zeolite and zeolite coated with manganese oxide nanoparticles. J Mol Liq 211:431–441. https://doi.org/10.1016/j.molliq.2015.06.067
Sharma A, Varshney M, Nanda SS, Shin HJ, Kim N, Yi DK, Chae K-H, Ok Won S (2018) Structural, electronic structure and antibacterial properties of graphene-oxide nano-sheets. Chem Phys Lett 698:85–92. https://doi.org/10.1016/j.cplett.2018.03.010
Toda K, Furue R, Hayami S (2015) Recent progress in applications of graphene oxide for gas sensing: a review. Anal Chim Acta 878:43–53. https://doi.org/10.1016/j.aca.2015.02.002
Verlicchi P, Al Aukidy M, Zambello E (2012) Occurrence of pharmaceutical compounds in urban wastewater: removal, mass load and environmental risk after a secondary treatment—a review. Sci Total Environ 429:123–155. https://doi.org/10.1016/j.scitotenv.2012.04.028
Vilt ME, Ho WSW (2009) Supported liquid membranes with strip dispersion for the recovery of cephalexin. J Membr Sci 342(1):80–87. https://doi.org/10.1016/j.memsci.2009.06.026
Wang X, Bai H, Shi G (2011) Size fractionation of graphene oxide sheets by pH-assisted selective sedimentation. J Am Chem Soc 133(16):6338–6342. https://doi.org/10.1021/ja200218y
Watanabe S, Tsuda M, Terada T, Katsura T, Inui K-I (2010) Reduced renal clearance of a zwitterionic substrate cephalexin in MATE1-deficient mice. J Pharmacol Exp Ther 334(2):651–656. https://doi.org/10.1124/jpet.110.169433
Watkinson AJ, Murby EJ, Costanzo SD (2007) Removal of antibiotics in conventional and advanced wastewater treatment: implications for environmental discharge and wastewater recycling. Water Res 41(18):4164–4176. https://doi.org/10.1016/j.watres.2007.04.005
Wu H, Feng Q, Yang H, Alam E, Gao B, Gu D (2017) Modified biochar supported Ag/Fe nanoparticles used for removal of cephalexin in solution: characterization, kinetics and mechanisms. Colloids Surf A Physicochem Eng Asp 517:63–71. https://doi.org/10.1016/j.colsurfa.2017.01.005
Xu J, Lv H, Yang S-T, Luo J (2013) Preparation of graphene adsorbents and their applications in water purification. In: Reviews in Inorganic Chemistry, vol. 33, vol 2–3, p 139
Xu X (2019) Facile assembly of three-dimensional cylindrical egg white embedded graphene oxide composite with good reusability for aqueous adsorption of rare earth elements. Colloids Surf 570:127–140. https://doi.org/10.1016/j.colsurfa.2019.03.022
Yang A, Zhu Y, Huang CP (2018) Facile preparation and adsorption performance of graphene oxide-manganese oxide composite for uranium. Sci Rep 8(1):9058. https://doi.org/10.1038/s41598-018-27111-y
Yang J-Y, Yue B-Y, Teng J, Xu X, Zhao X-R, Jiang X-Y, Yu J-G, Zhou F-L (2019) Aqueous metal ions adsorption by poly(ethylene glycol)-modified graphene oxide: surface area and surface chemistry effects. Desalin Water Treat 138:147–158. https://doi.org/10.5004/dwt.2019.23315
Yang SJ, Kang JH, Jung H, Kim T, Park C (2013a) Preparation of a freestanding, macroporous reduced graphene oxide film as an efficient and recyclable sorbent for oils and organic solvents. J Mater Chem A 1:9427–9432. https://doi.org/10.1039/c3ta10663b
Yang SJ, Kim T, Jung H, Park CR (2013b) The effect of heating rate on porosity production during the low temperature reduction of graphite oxide. Carbon 53:73–80. https://doi.org/10.1016/j.carbon.2012.10.032
Zhang P, Gong J-L, Zeng G-M, Deng C-H, Yang H-C, Liu H-Y, Huan S-Y (2017) Cross-linking to prepare composite graphene oxide-framework membranes with high-flux for dyes and heavy metal ions removal. Chem Eng J 322:657–666. https://doi.org/10.1016/j.cej.2017.04.068
Zhao X-R, Xu X, Teng J, Zhou N, Zhou Z, Jiang X-Y, Jiao F-P, Yu J-G (2019) Three-dimensional porous graphene oxide-maize amylopectin composites with controllable pore-sizes and good adsorption-desorption properties: facile fabrication and reutilization, and the adsorption mechanism. Ecotoxicol Environ Saf 176:11–19. https://doi.org/10.1016/j.ecoenv.2019.03.069
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Tito Roberto Cadaval Jr
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wernke, G., Shimabuku-Biadola, Q.L., dos Santos, T.R.T. et al. Adsorption of cephalexin in aqueous media by graphene oxide: kinetics, isotherm, and thermodynamics. Environ Sci Pollut Res 27, 4725–4736 (2020). https://doi.org/10.1007/s11356-019-07146-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-019-07146-y