Abstract
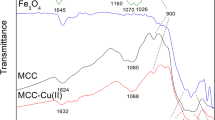
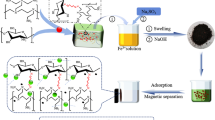
Magnetic phosphorylated chitosan composite (P-MCS), an excellent adsorbent for Co(II), was synthesized in this experiment via a facile fabrication. Its removal efficiency was improved by optimizing pH, contact time, and initial concentration. The adsorption isotherms and kinetic models of Co(II) by P-MCS followed the Langmuir model and the pseudo-second-order model, respectively. However, the rate of adsorption was also affected by intragranular diffusion. The maximum adsorption capacity was 46.1 mg g-1 for Co(II). The results of spectroscopic analysis also indicated that good adsorption performance of Co(II) mainly depends on surface chelation between functional groups and metal ions. The saturation magnetic susceptibilities of P-MCS and P-MCS-Co were 22.29 emu g−1 and 18.18 emu g−1, respectively. The excellent magnetic properties of P-MCS enabled the easy achievement of solid-liquid separation via the use of an external magnetic field. In complex aqueous solutions, K+, Na+, Ca2+, and Mg2+ have less influence on P-MCS adsorption Co(II), but the adsorption capacity on Co(II) is still good. This study shows the feasibility of using P-MCS to treat wastewater containing Co(II).








Similar content being viewed by others
References
Ajmal M, Rao RAK, Ahmad R, Ahmad J (2000) Adsorption studies on Citrus reticulata (fruit peel of orange): removal and recovery of Ni(II) from electroplating wastewater. J Hazard Mater 79(1–2):117–131. https://doi.org/10.1016/S0304-3894(00)00234-X
Cheung WH, Szeto YS, Mckay G (2007) Intraparticle diffusion processes during acid dye adsorption onto chitosan. Bioresour Technol 98(15):2897–2904. https://doi.org/10.1016/j.biortech.2006.09.045
Chiou MS, Li HY (2003) Adsorption behavior of reactive dye in aqueous solution on chemical cross-linked chitosan beads. Chemosphere 50(8):0–1105. https://doi.org/10.1016/s0045-6535(02)00636-7
Erdem E, Karapinar N, Donat R (2004) The removal of heavy metal cations by natural zeolites. J Colloid Interface Sci 280(2):309–314. https://doi.org/10.1016/j.jcis.2004.08.028
Fu F, Wang Q (2011) Removal of heavy metal ions from wastewaters: a review. J Environ Manag 92(3):407–418. https://doi.org/10.1016/j.jenvman.2010.11.011
Guibal E (2004) Interactions of metal ions with chitosan-based sorbents: a review. Sep Purif Technol 38:43–74. https://doi.org/10.1016/j.seppur.2003.10.004
Hendrickson SH (1967) Comparison of the metal-binding properties of nitrilotri(methylenephosphonic) acid and nitrilotriacetic acid: calcium(II), nickel(II,), iron(III), and thorium(IV) complexes. Anal Chem 39(8):998–1000. https://doi.org/10.1021/ac60252a022
Ho YS, Porter JF, Mckay G (2002) Equilibrium isotherm studies for the sorption of divalent metal ions onto peat: copper, nickel and lead single component systems. Water Air Soil Pollut 141(1–4):1–33. https://doi.org/10.1023/a:1021304828010
Huang Z, Li Z, Zheng L, Zhou L, Chai Z, Wang XL, Shi WQ (2017) Interaction mechanism of uranium(VI) with three-dimensional graphene oxide-chitosan composite: insights from batch experiments, IR, XPS, and EXAFS spectroscopy. Chem Eng J 328:1066–1074. https://doi.org/10.1016/S0020-1693(03)00184-1
Jayakumar R, Selvamurugan N, Nair SV, Tokura S, Tamura H (2008) Preparative methods of phosphorylated chitin and chitosan--an overview. Int J Biol Macromol 43(3):221–225. https://doi.org/10.1016/j.ijbiomac.2008.07.004
Kılıç M, Kırbıyık Ç, Çepelioğullar Ö, Pütün AE (2013) Adsorption of heavy metal ions from aqueous solutions by bio-char, a by-product of pyrolysis. Appl Surf Sci 283:856–862. https://doi.org/10.1016/j.apsusc.2013.07.033
Kinniburgh DG (1986) General purpose adsorption isotherms. Environ Sci Technol 20:895–904. https://doi.org/10.1021/es00151a008
Manohar DM, Noeline BF & Anirudhan TS (2006) Adsorption performance of Al-pillared bentonite clay for the removal of cobalt(II) from aqueous phase. Appl Clay Sci 31(3-4):194–206.
McKay G, Otterburn MS, Aga JA (1987) Intraparticle diffusion process occurring during adsorption of dyestuffs. Water Air Soil Pollut 36:381–390. https://doi.org/10.1007/BF00229680
Morsy AM (2015) Adsorptive removal of uranium ions from liquid waste solutions by phosphorylated chitosan. Environ Technol 4:299–310. https://doi.org/10.1016/j.eti.2015.10.002
Najjar AMK, Yunus WMZW, Ahmad MB et al (2000) Preparation and characterization of poly(2-acrylamido-2-methylpropane-sulfonic acid) grafted chitosan using potassium persulfate as redox initiator. J Appl Polym Sci 77(10):5. https://doi.org/10.1002/1097-4628(20000906) <2314::AID-APP25>3.0.CO;2-7
Ravi Kumar, MNV (2000) A review of chitin and chitosan applications. React Funct Polym 46(1):1–27. https://doi.org/10.1016/s1381-5148(00)00038-9
Repo E, Warchol JK, Kurniawan TA et al (2010) Adsorption of Co(II) and Ni(II) by EDTA- and/or DTPA-modified chitosan: kinetic and equilibrium modeling. Chem Eng J 161(1–2):73–82. https://doi.org/10.1016/j.cej.2010.04.030
Schwarzenbach G, Ackermann H, Ruckstuhl P (1949) Komplexone XV. Neue Derivate der Imino-diessigsäure und ihre Erdalkalikomplexe Beziehungen zwischen Acidität und Komplexbildung. Helv Chim Acta 32(4):1175–1186. https://doi.org/10.1002/hlca.19490320403
Sutirman ZA, Sanagi MM, Karim KJA, Ibrahim WAW, Jume BH (2018) Equilibrium, kinetic and mechanism studies of Cu(II) and Cd(II) ions adsorption by modified chitosan beads. Int J Biol Macromol 116:255–263. https://doi.org/10.1016/j.ijbiomac.2018.05.031
Wang J, Chen C (2009) Biosorbents for heavy metals removal and their future. Biotechnol Adv 27:195–226. https://doi.org/10.1016/j.biotechadv.2008.11.002
Wang J, Chen C (2014) Chitosan-based biosorbents: modification and application for biosorption of heavy metals and radionuclides. Bioresour Technol 160:129–141. https://doi.org/10.1016/j.biortech.2013.12.110
Wang ZG, Lv N, Bi WZ, Zhang JL, Ni JZ (2015) Development of the affinity materials for phosphorylated proteins/peptides enrichment in phosphoproteomics analysis. Acs Appl Mater Inter 7(16):8377–8392. https://doi.org/10.1021/acsami.5b01254
Wu D, Wang Y, Li Y, Wei Q, Hu L, Yan T, Feng R, Yan L, Du B (2019) Phosphorylated chitosan/CoFe2O4 composite for the efficient removal of Pb(II) and Cd(II) from aqueous solution: adsorption performance and mechanism studies. J Mol Liq 277:181–188. https://doi.org/10.1016/j.molliq.2018.12.098
Yan G, Viraraghavan T (2001) Heavy metal removal in a biosorption column by immobilized M rouxii biomass. Bioresour Technol 78(3):243–249. https://doi.org/10.1016/S0960-8524(01)00020-7
Zarghami Z, Akbari A, Latifi AM, Amani MA (2016) Design of a new integrated chitosan-PAMAM dendrimer biosorbent for heavy metals removing and study of its adsorption kinetics and thermodynamics. Bioresour Technol 205:230–238. https://doi.org/10.1016/j.biortech.2016.01.052
Zhuang S, Yin Y, Wang JL (2017) Removal of cobalt ions from aqueous solution using chitosan grafted with maleic acid by gamma radiation. Nucl Eng Technol 50(1):211–215. https://doi.org/10.1016/j.jes.2014.06.021
Funding
This work is supported by National Water Pollution Control and Management Technology Major Project (2018ZX07110005), the National Natural Science Foundation of China (51578037), the Guangxi Province Technology Major Project (AA17202032), the Scientific Research Program of Beijing Municipal Education Commission (KM201610016001), and the Fundamental Research Funds for Beijing University of Civil Engineering and Architecture (X18288 and X18289)
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Tito Roberto Cadaval Jr
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 21 kb)
Rights and permissions
About this article
Cite this article
Yuan, D., Zhang, W., Cui, J. et al. Facile fabrication of magnetic phosphorylated chitosan for the removal of Co(II) in water treatment: separation properties and adsorption mechanisms. Environ Sci Pollut Res 27, 2588–2598 (2020). https://doi.org/10.1007/s11356-019-07026-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-019-07026-5