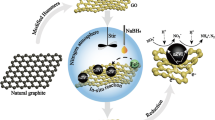
Abstract
An in situ wet chemical deposition method has been applied for the successful surface modification of Ce (IV)-incorporated hydrous Fe(III) oxide (CIHFO) with a hydrophilic graphene precursor, graphene oxide (GO). The surface area of as-prepared composite (GO-CIHFO) has enhanced (189.57 m2 g−1) compared with that of pristine CIHFO (140.711 m2 g−1) and has irregular surface morphology consisting of microcrystals (~ 2–3 nm) and mesoporous (3.5486 nm) structure. The GO-CIHFO composite shows enhanced fluoride scavenging capacity (136.24 mg F g−1) than GO (3 mg F g−1) and pristine CIHFO (32.62 mg F g−1) at pH 7.0. Also, in acidic pH range and at 323 K temperature, the Langmuir capacity of as-prepared composite is 190.61 mg F g−1. It has been observed that fluoride removal by GO–CIHFO occurs from solutions obeying pseudo-second-order kinetics and multilayer adsorption process. The film/boundary layer diffusion process is also the rate-determining step. The nature of the adsorption reaction is reasonably spontaneous and endothermic in thermodynamic sense. It was observed that 1.2 g.L−1 of GO-CIHFO dosage can effectively optimise the fluoride level of natural groundwater samples (9.05 mg L−1) to the desirable permissible limit. Reactivation of used material up to a level of 73.77% with a solution of alkaline pH has proposed reusability of nanocomposites ensuring sustainability of the proposed material as fluoride scavenger in future.











Similar content being viewed by others
References
Adeno F, Mulugeta E, Zewge F, Chebude Y (2014) Adsorptive removal of fluoride from water using nanoscale aluminium oxide hydroxide (AlOOH). Bull Chem Soc Ethiop 28:215–227. https://doi.org/10.4314/bcse.v28i2.6
Alagumuthu G, Rajan M (2008) Monitoring of fluoride concentration in ground water of Kadayam block of Tirunelveli district, India. Rasayan J Chem 1:920–928
Baltrusaitis J, Cwiertny DM, Grassian VH (2007) Adsorption of sulfur dioxide on hematite and goethite particle surfaces. Phys Chem Chem Phys 9:5542–5554. https://doi.org/10.1039/b709167b
Banerjee S, Chattopadhyaya MC (2017) Adsorption characteristics for the removal of a toxic dye, tartrazine from aqueous solutions by a low cost agricultural by-product. Arab J Chem 10:S1629–S1638.-https://doi.org/10.1016/j.arabjc.2013.06.005
Barathi M, Santhana Krishna Kumar A, Rajesh N (2013) Efficacy of novel Al–Zr impregnated cellulose adsorbent prepared using microwave irradiation for the facile defluoridation of water. J Environ Chem Eng 1:1325–1335. https://doi.org/10.1016/j.jece.2013.09.026
Barathi M, Krishna Kumar AS, Kumar CU, Rajesh N (2014) Graphene oxide–aluminium oxyhydroxide interaction and its application for the effective adsorption of fluoride. RSC Adv 4:53711–53721. https://doi.org/10.1039/C4RA10006A
Basu T, Ghosh UC (2013) Nano-structured iron(III)–cerium(IV) mixed oxide: Synthesis, characterization and arsenic sorption kinetics in the presence of co-existing ions aiming to apply for high arsenic groundwater treatment. Appl Surf Sci 283:471–481. https://doi.org/10.1016/j.apsusc.2013.06.132
Bhatnagar A, Kumar E, Sillanpää M (2011) Fluoride removal from water by adsorption—A review. Chem Eng J 171:811–840. https://doi.org/10.1016/j.cej.2011.05.028
Biswas K, Gupta K, Ghosh UC (2009) Adsorption of fluoride by hydrous iron(III)–tin(IV) bimetal mixed oxide from the aqueous solutions. Chem Eng J 149:196–206. https://doi.org/10.1016/j.cej.2008.09.047
Biswas K, Gupta K, Goswami A, Ghosh UC (2010) Fluoride removal efficiency from aqueous solution by synthetic iron(III)–aluminum(III)–chromium(III) ternary mixed oxide. Desalination 255:44–51. https://doi.org/10.1016/j.desal.2010.01.019
Briggs D (ed) (1978) Handbook of x-ray and ultraviolet photoelectron spectroscopy. Heyden, London [usw.], Rheine
Chakrabarti S, Bhattacharya HN (2013) Inferring the hydro-geochemistry of fluoride contamination in Bankura district, West Bengal: A case study. J Geol Soc India 82:379–391. https://doi.org/10.1007/s12594-013-0165-9
Chen L, He B-Y, He S, Wang TJ, Su CL, Jin Y (2012) Fe―Ti oxide nano-adsorbent synthesized by co-precipitation for fluoride removal from drinking water and its adsorption mechanism. Powder Technol 227:3–8. https://doi.org/10.1016/j.powtec.2011.11.030
Chen L, Zhang K, He J, Cai XG, Xu W, Liu JH (2016a) Performance and mechanism of hierarchically porous Ce–Zr oxide nanospheres encapsulated calcium alginate beads for fluoride removal from water. RSC Adv 6:36296–36306. https://doi.org/10.1039/C6RA01337F
Chen P, Zhang W, Li M, Ai P, Tian L, Jiang H (2016b) Facile synthesis of magnetic La–Zr composite as high effective adsorbent for fluoride removal. RSC Adv 6:35859–35867. https://doi.org/10.1039/C5RA27929A
Deng S, Liu H, Zhou W, Huang J, Yu G (2011) Mn–Ce oxide as a high-capacity adsorbent for fluoride removal from water. J Hazard Mater 186:1360–1366. https://doi.org/10.1016/j.jhazmat.2010.12.024
Drewniak S, Muzyka R, Stolarczyk A, Pustelny T, Kotyczka-Morańska M, Setkiewicz M (2016) Studies of reduced graphene oxide and graphite oxide in the aspect of their possible application in gas sensors. Sensors 16:103. https://doi.org/10.3390/s16010103
Dubinin MM, Radushkevich LV (1947) The Equation of the Characteristic Curve of Activated Charcoal. Proc Acad Sci USSR Phys Chem Sect 55:331–337
Freundlich HMF (1906) Over the Adsorption in Solution. Z Phys Chem 57:385–470
Gao C, Yu X-Y, Luo T, Jia Y, Sun B, Liu JH, Huang XJ (2014) Millimeter-sized Mg–Al-LDH nanoflake impregnated magnetic alginate beads (LDH-n-MABs): a novel bio-based sorbent for the removal of fluoride in water. J Mater Chem A 2:2119–2128. https://doi.org/10.1039/C3TA13526H
Ghosh A, Chakrabarti S, Biswas K, Ghosh UC (2014) Agglomerated nanoparticles of hydrous Ce(IV)+Zr(IV) mixed oxide: preparation, characterization and physicochemical aspects on fluoride adsorption. Appl Surf Sci 307:665–676. https://doi.org/10.1016/j.apsusc.2014.04.095
Hamayun M, Mahmood T, Naeem A et al (2014) Equilibrium and kinetics studies of arsenate adsorption by FePO. Chemosphere 99:207–215. https://doi.org/10.1016/j.chemosphere.2013.10.075
Hummers WS, Offeman RE (1958) Preparation of graphitic oxide. J Am Chem Soc 80:1339–1339. https://doi.org/10.1021/ja01539a017
Jagtap S, Yenkie MK, Labhsetwar N, Rayalu S (2012) Fluoride in drinking water and defluoridation of water. Chem Rev 112:2454–2466. https://doi.org/10.1021/cr2002855
Kanrar S, Debnath S, De P et al (2016) Preparation, characterization and evaluation of fluoride adsorption efficiency from water of iron-aluminium oxide-graphene oxide composite material. Chem Eng J 306:269–279. https://doi.org/10.1016/j.cej.2016.07.037
Kuang L, Liu Y, Fu D, Zhao Y (2017) FeOOH-graphene oxide nanocomposites for fluoride removal from water: Acetate mediated nano FeOOH growth and adsorption mechanism. J Colloid Interface Sci 490:259–269. https://doi.org/10.1016/j.jcis.2016.11.071
Langevoort JC, Sutherland I, Hanekamp LJ, Gellings PJ (1987) On the oxide formation on stainless steels AISI 304 and incoloy 800H investigated with XPS. Appl Surf Sci 28:167–179. https://doi.org/10.1016/0169-4332(87)90062-6
Li Y, Zhang P, Du Q et al (2011) Adsorption of fluoride from aqueous solution by graphene. J Colloid Interface Sci 363:348–354. https://doi.org/10.1016/j.jcis.2011.07.032
Li Y, Du Q, Wang J et al (2013) Defluoridation from aqueous solution by manganese oxide coated graphene oxide. J Fluor Chem 148:67–73. https://doi.org/10.1016/j.jfluchem.2013.01.028
Liu H, Deng S, Li Z, Yu G, Huang J (2010) Preparation of Al–Ce hybrid adsorbent and its application for defluoridation of drinking water. J Hazard Mater 179:424–430. https://doi.org/10.1016/j.jhazmat.2010.03.021
McKay G, Otterburn MS, Sweeney AG (1981) Surface mass transfer processes during colour removal from effluent using silica. Water Res 15:327–331. https://doi.org/10.1016/0043-1354(81)90036-1
Meenakshi, Maheshwari RC (2006) Fluoride in drinking water and its removal. J Hazard Mater 137:456–463. https://doi.org/10.1016/j.jhazmat.2006.02.024
Mohan S, Kumar V, Singh DK, Hasan SH (2016a) Synthesis and characterization of rGO/ZrO 2 nanocomposite for enhanced removal of fluoride from water: kinetics, isotherm, and thermodynamic modeling and its adsorption mechanism. RSC Adv 6:87523–87538. https://doi.org/10.1039/C6RA15460C
Mohan S, Kumar V, Singh DK, Hasan SH (2016b) Synthesis and characterization of rGO/ZrO 2 nanocomposite for enhanced removal of fluoride from water: kinetics, isotherm, and thermodynamic modeling and its adsorption mechanism. RSC Adv 6:87523–87538. https://doi.org/10.1039/C6RA15460C
Mohammadian S, Ghanemi K, Nikpour Y (2016) Competitive adsorption of phthalate esters on marine surface sediments: kinetic, thermodynamic, and environmental considerations. Environ Sci Pollut Res 23:24991–25002. https://doi.org/10.1007/s11356-016-7723-9
Mukhopadhyay K, Ghosh A, Das SK, Show B, Sasikumar P, Chand Ghosh U (2017b) Synthesis and characterisation of cerium(IV)-incorporated hydrous iron(III) oxide as an adsorbent for fluoride removal from water. RSC Adv 7:26037–26051. https://doi.org/10.1039/c7ra00265c
Novoselov KS (2004) Electric Field Effect in Atomically Thin Carbon Films. Science 306:666–669. https://doi.org/10.1126/science.1102896
Podder MS, Majumder CB (2016) Application of granular activated carbon/MnFe2O4 composite immobilized on C. glutamicum MTCC 2745 to remove As(III) and As(V): Kinetic, mechanistic and thermodynamic studies. Spectrochim Acta A Mol Biomol Spectrosc 153:298–314. https://doi.org/10.1016/j.saa.2015.08.022
Ranjan D, Talat M, Hasan SH (2009) Biosorption of arsenic from aqueous solution using agricultural residue ‘rice polish’. J Hazard Mater 166:1050–1059. https://doi.org/10.1016/j.jhazmat.2008.12.013
Reichenberg D (1953) Properties of Ion-Exchange Resins in Relation to their Structure. III. Kinetics of Exchange. J Am Chem Soc 75:589–597. https://doi.org/10.1021/ja01099a022
Saha I, Ghosh A, Nandi D et al (2015) ß-Cyclodextrin modified hydrous zirconium oxide: Synthesis, characterization and defluoridation performance from aqueous solution. Chem Eng J 263:220–230. https://doi.org/10.1016/j.cej.2014.11.039
Sharma YC, Weng CH (2007) Removal of chromium(VI) from water and wastewater by using riverbed sand: Kinetic and equilibrium studies. J Hazard Mater 142:449–454. https://doi.org/10.1016/j.jhazmat.2006.08.078
Singh DK, Mohan S, Kumar V, Hasan SH (2016) Kinetic, isotherm and thermodynamic studies of adsorption behaviour of CNT/CuO nanocomposite for the removal of As(iii) and As(v) from water. RSC Adv 6:1218–1230. https://doi.org/10.1039/C5RA20601D
Singh DK, Kumar V, Mohan S, Hasan SH (2017) Polylysine Functionalized Graphene Aerogel for the Enhanced Removal of Cr(VI) through Adsorption: Kinetic, Isotherm, and Thermodynamic Modeling of the Process. J Chem Eng Data 62:1732–1742. https://doi.org/10.1021/acs.jced.7b00188
Tang D, Zhang G (2016) Efficient removal of fluoride by hierarchical Ce–Fe bimetal oxides adsorbent: Thermodynamics, kinetics and mechanism. Chem Eng J 283:721–729. https://doi.org/10.1016/j.cej.2015.08.019
Tomar V, Prasad S, Kumar D (2013) Adsorptive removal of fluoride from water samples using Zr–Mn composite material. Microchem J 111:116–124. https://doi.org/10.1016/j.microc.2013.04.007
Wan Z, Wang J (2016) Ce-Fe-reduced graphene oxide nanocomposite as an efficient catalyst for sulfamethazine degradation in aqueous solution. Environ Sci Pollut Res 23:18542–18551. https://doi.org/10.1007/s11356-016-7051-0
Wen S, Wang Y, Dong S (2015) Performance and characteristics of fluoride adsorption using nanomagnetite graphite-La adsorbent. RSC Adv 5:89594–89602. https://doi.org/10.1039/c5ra15215a
Zhang K, Wu S, Wang X, He J, Sun B, Jia Y, Luo T, Meng F, Jin Z, Lin D, Shen W, Kong L, Liu J (2015) Wide pH range for fluoride removal from water by MHS-MgO/MgCO3 adsorbent: Kinetic, thermodynamic and mechanism studies. J Colloid Interface Sci 446:194–202. https://doi.org/10.1016/j.jcis.2015.01.049
Zhang C, Li Y, Wang T-J, Jiang Y, Fok J (2017) Synthesis and properties of a high-capacity iron oxide adsorbent for fluoride removal from drinking water. Appl Surf Sci 425:272–281. https://doi.org/10.1016/j.apsusc.2017.06.159
Zhao B, Zhang Y, Dou X, Wu X, Yang M (2012a) Granulation of Fe–Al–Ce trimetal hydroxide as a fluoride adsorbent using the extrusion method. Chem Eng J 185–186:211–218. https://doi.org/10.1016/j.cej.2012.01.085
Zhao B, Zhang Y, Dou X, Wu X, Yang M (2012b) Granulation of Fe-Al-Ce trimetal hydroxide as a fluoride adsorbent using the extrusion method. Chem Eng J 185-186:211–218. https://doi.org/10.1016/j.cej.2012.01.085
Zhu J, Lin X, Wu P, Zhou Q, Luo X (2015) Fluoride removal from aqueous solution by Al(III)–Zr(IV) binary oxide adsorbent. Appl Surf Sci 357:91–100. https://doi.org/10.1016/j.apsusc.2015.09.012
Acknowledgements
The authors are grateful to the Department of Geology and also to the Department of Chemistry including the Authority of Presidency University, Kolkata, India, for providing the laboratory facility for carry out this work. The author (K. M.) is grateful to the Department of Science and Technology (DST), Govt. of India, for providing the financial support in the form of a Woman Scientist-B (DST/Disha/SoRF-PM/064/2013/C dated 14/09/2015). P.S. is thankful to Presidency University for the financial support through a FRPDF grant.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Tito Roberto Cadaval Jr
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 21820 kb)
Rights and permissions
About this article
Cite this article
Mukhopadhyay, K., Ghosh, U.C. & Sasikumar, P. Enhanced capacity of fluoride scavenging from contaminated water by nano-architectural reorientation of cerium-incorporated hydrous iron oxide with graphene oxide. Environ Sci Pollut Res 26, 26112–26133 (2019). https://doi.org/10.1007/s11356-019-05756-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-019-05756-0