Abstract
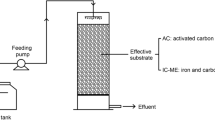
A process combining catalyzed Fe(0)-carbon microelectrolysis (IC-ME) with activated carbon (AC) adsorption was developed for advanced reclaimed water treatment. Simultaneous nitrate reduction and chemical oxygen demand (COD) removal were achieved, and the effects of composite catalyst (CC) addition, AC addition, and initial pH were investigated. The reaction kinetics and reaction mechanisms were calculated and analyzed. The results showed that CC addition could enhance the reduction rate of nitrate and effectively inhibit the production of ammonia. Moreover, AC addition increased the adsorption capacity of biorefractory organic compounds (BROs) and enhanced the degradation of BRO. The reduction of NO3−–N at different pH values was consistently greater than 96.9%, and NH4+–N was suppressed by high pH. The presence of CC ensured the reaction rate of IC-ME at high pH. The reaction kinetics orders and constants were calculated. Catalyzed iron scrap (IS)-AC showed much better nitrate reduction and BRO degradation performances than IS-AC and AC. The IC-ME showed great potential for application to nitrate and BRO reduction in reclaimed water.







Similar content being viewed by others
References
Ahn SC, Oh SY, Cha DK (2008) Enhanced reduction of nitrate by zero-valent iron at elevated temperatures. J Hazard Mater 156:17–22. https://doi.org/10.1016/j.jhazmat.2007.11.104
Alessio S (2015) Use of nanoscale zero-valent iron (NZVI) particles for chemical denitrification under different operating conditions. Metals 5:1507–1519
Chen J, Liu S, Yan J, Wen J, Hu Y, Zhang W (2017) Intensive removal efficiency and mechanisms of carbon and ammonium in municipal wastewater treatment plant tail water by ozone oyster shells fix-bed bioreactor − membrane bioreactor combined system. Ecol Eng 101:75–83. https://doi.org/10.1016/j.ecoleng.2016.11.029
Cho D-W, Chon C-M, Jeon B-H, Kim Y, Khan MA, Song H (2010) The role of clay minerals in the reduction of nitrate in groundwater by zero-valent iron. Chemosphere 81:611–616. https://doi.org/10.1016/j.chemosphere.2010.08.005
Deng S, Li D, Yang X, Xing W, Li J, Zhang Q (2016a) Biological denitrification process based on the Fe(0)–carbon micro-electrolysis for simultaneous ammonia and nitrate removal from low organic carbon water under a microaerobic condition. Bioresour Technol 219:677–686. https://doi.org/10.1016/j.biortech.2016.08.014
Deng S, Li D, Yang X, Zhu S, Xing W (2016b) Advanced low carbon-to-nitrogen ratio wastewater treatment by electrochemical and biological coupling process. Environ Sci PollutR Int 23:5361–5373. https://doi.org/10.1007/s11356-015-5711-0
Deng S, Li D, Yang X, Xing W, Li J, Zhang Q (2017) Iron [Fe(0)]-rich substrate based on iron–carbon micro–electrolysis for phosphorus adsorption in aqueous solutions. Chemosphere 168:1486–1493. https://doi.org/10.1016/j.chemosphere.2016.11.043
Desheng L, Taixing F, Yanbing S, Weizhong W (2013) Electrochemical technology for denitrification of tail water from wastewater treatment plant. CIESC J (China) 64:1084–1090
Desheng L, Qianyi H, Yuwei C, Shihai D (2015) Chemical catalytic performance on nitrate removal of simulated groundwater. CIESC J (China) 66:2288–2294
Devi P, Saroha AK (2015) Simultaneous adsorption and dechlorination of pentachlorophenol from effluent by Ni–ZVI magnetic biochar composites synthesized from paper mill sludge. Chem Eng J 271:195–203. https://doi.org/10.1016/j.cej.2015.02.087
Feng C, Huang L, Yu H, Yi X, Wei C (2015) Simultaneous phenol removal, nitrification and denitrification using microbial fuel cell technology. Water Res 76:160–170. https://doi.org/10.1016/j.watres.2015.03.001
Gao L, Zhou W, Huang J, He S, Yan Y, Zhu W, Wu S, Zhang X (2017) Nitrogen removal by the enhanced floating treatment wetlands from the secondary effluent. Bioresour Technol 234:243–252. https://doi.org/10.1016/j.biortech.2017.03.036
He S, Wang Y, Li C, Li Y, Zhou J (2018) The nitrogen removal performance and microbial communities in a two-stage deep sequencing constructed wetland for advanced treatment of secondary effluent. Bioresour Technol 248:82–88. https://doi.org/10.1016/j.biortech.2017.06.150
Hongbiao X, Pengpeng X, Hong X, SHaokun Z, Zhongkai R (2016) Finite element simulation and experiment of composite rolling of stainless steel/iron scrap. J Plasticity Eng (China) 23:69–72
Hwang Y, Mines PD, Jakobsen MH, Andersen HR (2015) Simple colorimetric assay for dehalogenation reactivity of nanoscale zero-valent iron using 4-chlorophenol. Appl Catal B Environ 166-167:18–24. https://doi.org/10.1016/j.apcatb.2014.10.059
Kiskira K, Papirio S, van Hullebusch ED, Esposito G (2017) Fe(II)-mediated autotrophic denitrification: a new bioprocess for iron bioprecipitation/biorecovery and simultaneous treatment of nitrate-containing wastewaters. Int Biodeterior Biodegrad 119:631–648. https://doi.org/10.1016/j.ibiod.2016.09.020
Lee KP, Arnot TC, Mattia D (2011) A review of reverse osmosis membrane materials for desalination—development to date and future potential. J Membr Sci 370:1–22. https://doi.org/10.1016/j.memsci.2010.12.036
Le-Minh N, Khan SJ, Drewes JE, Stuetz RM (2010) Fate of antibiotics during municipal water recycling treatment processes. Water Res 44:4295–4323. https://doi.org/10.1016/j.watres.2010.06.020
Li D (2013) A porous granulated biological carrier for nitrogen removal of low carbon to nitrogen ratio wastewater under aerobic conditions and its preparation processes. China, CN201310093411
Li B, Sun K, Guo Y, Tian J, Xue Y, Sun D (2013) Adsorption kinetics of phenol from water on Fe/AC. Fuel 110:99–106. https://doi.org/10.1016/j.Fuel.2012.10.043
Li P, Liu Z, Wang X, Guo Y, Wang L (2017) Enhanced decolorization of methyl orange in aqueous solution using iron–carbon micro-electrolysis activation of sodium persulfate. Chemosphere 180:100–107. https://doi.org/10.1016/j.chemosphere.2017.04.019
Liu Y, Li S, Chen Z, Megharaj M, Naidu R (2014) Influence of zero-valent iron nanoparticles on nitrate removal by Paracoccus sp. Chemosphere 108:426–432. https://doi.org/10.1016/j.chemosphere.2014.02.045
Lubphoo Y, Chyan J-M, Grisdanurak N, Liao C-H (2015) Nitrogen gas selectivity enhancement on nitrate denitrification using nanoscale zero-valent iron supported palladium/copper catalysts. J Taiwan Inst Chem E 57:143–153. https://doi.org/10.1016/j.jtice.2015.05.005
Luo J, Song G, Liu J, Qian G, Xu ZP (2014) Mechanism of enhanced nitrate reduction via micro-electrolysis at the powdered zero-valent iron/activated carbon interface. J Colloid Interface Sci 435:21–25. https://doi.org/10.1016/j.jcis.2014.08.043
Metcalf Eddy I (2004) Wastewater engineering: Treatment, disposal, reuse. McGraw-Hill, New York
Ozgun H, Dereli RK, Ersahin ME, Kinaci C, Spanjers H, van Lier JB (2013) A review of anaerobic membrane bioreactors for municipal wastewater treatment: integration options, limitations and expectations. Sep Purif Technol 118:89–104. https://doi.org/10.1016/j.seppur.2013.06.036
Qin L, Zhang G, Meng Q, Xu L, Lv B (2012) Enhanced MBR by internal micro-electrolysis for degradation of anthraquinone dye wastewater. Chem Eng J 210:575–584. https://doi.org/10.1016/j.cej.2012.09.006
Rodríguez-Maroto JM, García-Herruzo F, García-Rubio A, Gómez-Lahoz C, Vereda-Alonso C (2009) Kinetics of the chemical reduction of nitrate by zero-valent iron. Chemosphere 74:804–809. https://doi.org/10.1016/j.chemosphere.2008.10.020
Schmidt CA, Clark MW (2012) Efficacy of a denitrification wall to treat continuously high nitrate loads. Ecol Eng 42:203–211. https://doi.org/10.1016/j.ecoleng.2012.02.006
Shi D, Zhang X, Wang J, Fan J (2018) Highly reactive and stable nanoscale zero-valent iron prepared within vesicles and its high-performance removal of water pollutants. Appl Catal B Environ 221:610–617. https://doi.org/10.1016/j.apcatb.2017.09.057
Song X, Chen Z, Wang X, Zhang S (2017) Ligand effects on nitrate reduction by zero-valent iron: role of surface complexation. Water Res 114:218–227. https://doi.org/10.1016/j.watres.2017.02.040
Tang S, X-m W, H-w Y, Xie YF (2013) Haloacetic acid removal by sequential zero-valent iron reduction and biologically active carbon degradation. Chemosphere 90:1563–1567. https://doi.org/10.1016/j.Chemosphere.2012.09.046
Wang X, Wang J, Li K, Zhang H, Yang M (2018) Molecular characterization of effluent organic matter in secondary effluent and reclaimed water: comparison to natural organic matter in source water. J Environ Sci 63:140–146. https://doi.org/10.1016/j.jes.2017.03.020
Weast L (1989) CRC Handbook of chemisity and physics. CRC Press, Boca Raton
Westerhoff P, James J (2003) Nitrate removal in zero-valent iron packed columns. Water Res 37:1818–1830. https://doi.org/10.1016/s0043-1354(02)00539-0
Xing W, Li D, Li J, Hu Q, Deng S (2016) Nitrate removal and microbial analysis by combined micro-electrolysis and autotrophic denitrification. Bioresour Technol 211:240–247. https://doi.org/10.1016/j.biortech.2016.03.044
Xu H, Li Y, Ding M, Chen W, Wang K, Lu C (2018) Simultaneous removal of dissolved organic matter and nitrate from sewage treatment plant effluents using photocatalytic membranes. Water Res 143:250–259. https://doi.org/10.1016/j.watres.2018.06.044
Yang Z, Ma Y, Liu Y, Li Q, Zhou Z, Ren Z (2017) Degradation of organic pollutants in near-neutral pH solution by Fe-C micro-electrolysis system. Chem Eng J 315:403–414. https://doi.org/10.1016/j.cej.2017.01.042
Ying D, Peng J, Xu X, Li K, Wang Y, Jia J (2012) Treatment of mature landfill leachate by internal micro-electrolysis integrated with coagulation: a comparative study on a novel sequencing batch reactor based on zero valent iron. J Hazard Mater 229-230:426–433. https://doi.org/10.1016/j.jhazmat.2012.06.037
Zhang Y, Douglas GB, Pu L, Zhao Q, Tang Y, Xu W, Luo B, Hong W, Cui L, Ye Z (2017) Zero-valent iron-facilitated reduction of nitrate: chemical kinetics and reaction pathways. Sci Total Environ 598:1140–1150. https://doi.org/10.1016/j.scitotenv.2017.04.071
Zhang Y, Li J, Bai J, Li X, Shen Z, Xia L, Chen S, Xu Q, Zhou B (2018) Total organic carbon and total nitrogen removal and simultaneous electricity generation for nitrogen-containing wastewater based on the catalytic reactions of hydroxyl and chlorine radicals. Appl Catal B Environ 238:168–176. https://doi.org/10.1016/j.apcatb.2018.07.036
Zhou H, Lv P, Shen Y, Wang J, Fan J (2013) Identification of degradation products of ionic liquids in an ultrasound assisted zero-valent iron activated carbon micro-electrolysis system and their degradation mechanism. Water Res 47:3514–3522. https://doi.org/10.1016/j.watres.2013.03.057
Funding
This work was supported by the Fundamental Research Funds for the Central Universities (No. 2018YJS123) and the National Natural Science Foundation of China (No. 51778040).
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Bingcai Pan
First author: Zhifeng Hu
Electronic supplementary material
ESM 1
(DOCX 17 kb)
Rights and permissions
About this article
Cite this article
Hu, Z., Li, D., Deng, S. et al. Combination with catalyzed Fe(0)-carbon microelectrolysis and activated carbon adsorption for advanced reclaimed water treatment: simultaneous nitrate and biorefractory organics removal . Environ Sci Pollut Res 26, 5693–5703 (2019). https://doi.org/10.1007/s11356-018-3919-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-018-3919-5