Abstract
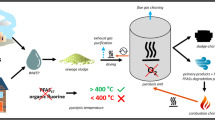
In this study, sewage sludge-derived activated carbon (SDAC) was synthesized, characterized, and tested for its potential as an adsorbent of methyl tert-butyl ether (MTBE). The SDAC was produced by activating the sludge with zinc chloride and subsequently subjected to various ranges of pyrolytic temperatures. It was then characterized using SEM/EDX, BET, and TGA. The SEM-EDX analyses showed that impurities like Fe, Al, Mg, Mn, Ca, and Na of the raw sludge were removed by the higher pyrolytic temperature and acid-washing procedures. TGA showed the thermal stability of the produced material. Results of the BET revealed a significant increase in surface area of the sludge from 1.5 m2/g to 385 m2/g after acid washing. The MTBE removal efficiency of 70% was achieved after 60 min with 2 g/L of SDAC at pH 6, and initial MTBE concentration of 1 ppm. The adsorption kinetics of SDAC fitted into pseudo-second-order reactions. This work demonstrated a beneficial use of a bio-waste material (sewage sludge) in water treatment technologies.












Similar content being viewed by others
Abbreviations
- Ct (mg/l):
-
Equilibrium concentration of MTBE at time t.
- Ce (mg/l):
-
Equilibrium concentration of MTBE obtained at the end of experiment.
- Co (mg/L):
-
Initial concentration of MTBE, (mg/l).
- Dp (Å):
-
Average pore diameter.
- Kf :
-
Freundlich isotherm constant
- KL (L/mg):
-
Langmuir constant related to the energy of adsorption.
- m (g):
-
mass of the adsorbed sample
- qe (mg/g):
-
Amount of MTBE adsorbed per unit mass of adsorbent at equilibrium.
- qt (mg/g):
-
Amount of MTBE adsorbed at a given time.
- qm (mg/g):
-
Maximum adsorption capacity of MTBE per unit mass of adsorbent.
- SB (m2/g):
-
BET surface area of the adsorbent
- Se (m2/g):
-
External pore surface area
- t (min):
-
Time of running the isotherm experiment
- V (mL):
-
Volume of the solution.
- Vm (cm3/g):
-
Micropores volume
- Vt (cm3/g):
-
Total pore volume
References
Adebayo SB, Tawabini BS, Atieh MA, Abuilaiwi FA, Alfadul S (2016) Investigating the removal of methyl tertiary butyl ether (MTBE) from water using raw and modified fly ash waste materials. Desalin Water Treat 57:26307–26312
Al-Qodah Z, Shawabkah R (2009) Production and characterization of granular activated carbon from activated sludge. Braz J Chem Eng 26:127–136
Bagreev A, Bandosz TJ (2002) H2S adsorption/oxidation on materials obtained using sulfuric acid activation of sewage sludge-derived fertilizer. J Colloid Interface Sci 252:188–194
Bagreev A, Bandosz TJ, Locke DC (2001) Pore structure and surface chemistry of adsorbents obtained by pyrolysis of sewage sludge-derived fertilizer. Carbon 39:1971–1979
Chen X, Jeyaseelan S, Graham N (2002) Physical and chemical properties study of the activated carbon made from sewage sludge. Waste Manag 22:755–760
Dai W, Gong R, Hu J, Zhou L-M (2014) Thiophene capture by an oxidation-modified activated carbon derived from bergamot. Sep Sci Technol 49:367–375
Fang P, Cen C, Chen D, Tang Z (2010) Carbonaceous adsorbents prepared from sewage sludge and its application for Hg0 adsorption in simulated flue gas. Chin J Chem Eng 18:231–238
Franus M, Barnat-Hunek D, Wdowin M (2016) Utilization of sewage sludge in the manufacture of lightweight aggregate. Environ Monit Assess 188:10–15
Ganiyu SA, Alhooshani K, Sulaiman KO, Qamaruddin M, Bakare IA, Tanimu A, Saleh TA (2016) Influence of aluminium impregnation on activated carbon for enhanced desulfurization of DBT at ambient temperature: role of surface acidity and textural properties. Chem Eng J 303:489–500
Gu L, Zhu N, Guo H, Huang S, Lou Z, Yuan H (2013) Adsorption and Fenton-like degradation of naphthalene dye intermediate on sewage sludge derived porous carbon. J Hazard Mater 246-247:145–153
Hejazi RF, Husain T, Khan FI (2003) Landfarming operation of oily sludge in arid region—human health risk assessment. J Hazard Mater 99:287–302
Jarrah RT, Siddiqui MK (2012) Sustainability: opportunities and challenges from a construction contractor’s perspective, ICSDEC 2012: developing the frontier of sustainable design, engineering, and construction. American Society of Civil Engineers, Fort Worth
Ji B, Shao F, Hu G, Zheng S, Zhang Q, Xu Z (2009) Adsorption of methyl tert-butyl ether (MTBE) from aqueous solution by porous polymeric adsorbents. J Hazard Mater 161:81–87
Julcour Lebigue C, Andriantsiferana C, N’Guessan K, Ayral C, Mohamed E, Wilhelm A-M, Delmas H, Le Coq L, Gerente C, Smith KM, Pullket S, Fowler GD, Graham NJD (2010) Application of sludge-based carbonaceous materials in a hybrid water treatment process based on adsorption and catalytic wet air oxidation. J Environ Manag 91:2432–2439
Li YH, Liu YL, Deng H, Zhang SL (2012) The adsorption and desorption characteristics of a novel sorbent in groundwater remediation: modified oil sludge. In: Cheng GZAS (ed) Advanced Materials Research, pp 412-417
Lodeiro P, Barriada JL, Herrero R, Sastre de Vicente ME (2006) The marine macroalga Cystoseira baccata as biosorbent for cadmium(II) and lead(II) removal: kinetic and equilibrium studies. Environ Pollut 142:264–273
Lu H, Zhang W, Wang S, Zhuang L, Yang Y, Qiu R (2013) Characterization of sewage sludge-derived biochars from different feedstocks and pyrolysis temperatures. J Anal Appl Pyrolysis 102:137–143
Martin MJ, Adriana A, Dolors BM, Miquel R (2002) Towards waste minimisation in WWTP: activated carbon from biological sludge and its application in liquid phase adsorption. J Chem Technol Biotechnol 77:825–833
Molina-Sabio M, RodRíguez-Reinoso F, Caturla F, Sellés MJ (1995) Porosity in granular carbons activated with phosphoric acid. Carbon 33:1105–1113
Nilsson C, Dahlström H (2005) Treatment and disposal methods for wastewater sludge in the area of Beijing, China. Lund University, Sweden
Rengaraj S, Moon S-H, Sivabalan R, Arabindoo B, Murugesan V (2002) Removal of phenol from aqueous solution and resin manufacturing industry wastewater using an agricultural waste: rubber seed coat. J Hazard Mater 89:185–196
Rio S, Le Coq L, Faur C, Lecomte D, Le Cloirec P (2006) Preparation of adsorbents from sewage sludge by steam activation for industrial emission treatment. Process Saf Environ Prot 84:258–264
Salimi A, Vaghar-Moussavi M, Seydi E, Pourahmad J (2016) Toxicity of methyl tertiary-butyl ether on human blood lymphocytes. Environ Sci Pollut Res 23:8556–8564
Salleh MAM, Mahmoud DK, Karim WAWA, Idris A (2011) Cationic and anionic dye adsorption by agricultural solid wastes: a comprehensive review. Desalination 280:1–13
Selmani A, Špadina M, Plodinec M, Delač Marion I, Willinger MG, Lützenkirchen J, Gafney HD, Redel E (2015) An experimental and theoretical approach to understanding the surface properties of one-dimensional TiO2 nanomaterials. J Phys Chem C 119:19729–19742
Senthil Kumar P, Ramalingam S, Senthamarai C, Niranjanaa M, Vijayalakshmi P, Sivanesan S (2010) Adsorption of dye from aqueous solution by cashew nut shell: studies on equilibrium isotherm, kinetics and thermodynamics of interactions. Desalination 261:52–60
Sethia G & Sayari A (2016) Activated carbon with optimum pore size distribution for hydrogen storage. Carbon 99:289–294
Shah I, Adnan R, Wan Ngah WS, Mohamed N (2015) Iron impregnated activated carbon as an efficient adsorbent for the removal of methylene blue: regeneration and kinetics studies. PLoS One 10:e0122603
Shahidi F, Ambigaipalan P (2015) Phenolics and polyphenolics in foods, beverages and spices: antioxidant activity and health effects—a review. J Funct Foods 18:820–897
Shahidi Bonjar GH (2004) Potential ecotoxicological implication of methyl tert-butyl ether (MTBE) spills in the environment. Ecotoxicology 13:631–635
Shawabkah R, Al-Qodah Z, Al-Bsoul A (2015) Bio-adsorption of triadimenol pesticide from aqueous solutions using activated sludge of dairy plants. Desalin Water Treat 53:2555–2564
Shawabkeh RA, Abu-Nameh ESM (2007) Absorption of phenol and methylene blue by activated carbon from pecan shells. Colloid J 69:355–359
Sheha D, Khalaf H, Daghestani N (2013) Experimental design methodology for the preparation of activated carbon from sewage sludge by chemical activation process. Arab J Sci Eng 38:2941–2951
Shih TC, Wangpaichitr M, Suffet M (2003) Evaluation of granular activated carbon technology for the removal of methyl tertiary butyl ether (MTBE) from drinking water. Water Res 37:375–385
Squillace PJ, Pankow JF, Korte NE, Zogorski JS (1997) Review of the environmental behavior and fate of methyl tert-butyl ether. Environ Toxicol Chem 16:1836–1844
Tawabini BS (2014) Simultaneous removal of MTBE and benzene from contaminated groundwater using ultraviolet-based ozone and hydrogen peroxide. Int J Photoenergy 2014:7
Waqas M, Li G, Khan S, Shamshad I, Reid BJ, Qamar Z, Chao C (2015) Application of sewage sludge and sewage sludge biochar to reduce polycyclic aromatic hydrocarbons (PAH) and potentially toxic elements (PTE) accumulation in tomato. Environ Sci Pollut Res 22:12114–12123
Wu F-C, Tseng R-L, Juang R-S (2005) Preparation of highly microporous carbons from fir wood by KOH activation for adsorption of dyes and phenols from water. Sep Purif Technol 47:10–19
Yu L, Adams C, Ludlow D (2005) Adsorption isotherms for methyl <i>Tert</i>-butyl ether and other fuel oxygenates on two bituminous-coal activated carbons. J Environ Eng 131:983–987
Zhang F-S, Nriagu JO, Itoh H (2005) Mercury removal from water using activated carbons derived from organic sewage sludge. Water Res 39:389–395
Zou J, Dai Y, Wang X, Ren Z, Tian C, Pan K, Li S, Abuobeidah M, Fu H (2013) Structure and adsorption properties of sewage sludge-derived carbon with removal of inorganic impurities and high porosity. Bioresour Technol 142:209–217
Acknowledgements
The authors acknowledge King Fahd University of Petroleum & Minerals (KFUPM) for providing an enabling environment, necessary tools and financial support to carry out this study.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Tito Roberto Cadaval Jr
Rights and permissions
About this article
Cite this article
Liadi, M., Tawabini, B., Shawabkeh, R. et al. Treating MTBE-contaminated water using sewage sludge-derived activated carbon. Environ Sci Pollut Res 25, 29397–29407 (2018). https://doi.org/10.1007/s11356-018-2737-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-018-2737-0