Abstract
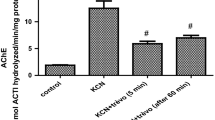
Our study designed to study the potential of potassium dichromate (K2Cr2O7) oral exposure to induce damage in male rat brain and to compare the possible protective role of vitamin C (VC) either pre and/or concurrent supply against (K2Cr2O7) induced changes. Thirty male rats were divided into five groups. First control group received distilled water (C), second received 120 mg/kg b.wt (VC), third received 25 mg/kg b.wt K2Cr2O7 (Cr), fourth group received VC together with K2Cr2O7 by the same former doses (VC + Cr), and the fifth group received the same oral doses of VC 2 weeks prior to and along with K2Cr2O7 for 6 weeks (VC + Cr pro/co treated). The obtained results revealed that K2Cr2O7 induced a significant decrease in cholinergic activity, glutathione reductase GR activity, reduced glutathione content GSH and ATP levels. Furthermore, K2Cr2O7 induced a significant increase in oxidative DNA damage indicated by 8-hydroxy 2′-deoxyguanosine (8OH2′dG) and formation of apoptotic DNA ladders, significant increase in malondialdehyde (MDA), protein carbonyl, and lactate dehydrogenase enzyme. Increased mRNA expression of pro-apoptotic genes, including caspase-3, p53, and Bax, unlike Bcl-2 expression, was decreased. K2Cr2O7 increased caspase-3 and decreased Bcl-2 immuno-labeling. VC supply noticeably ameliorates K2Cr2O7-induced changes which were more significantly in VC pro and concurrent supplement rather than VC concurrent supply only. Finally, it is concluded that K2Cr2O7 oral administration induced oxidative apoptotic changes in rat brain and confirms the usefulness of VC pre and concurrent supply for the amelioration of K2Cr2O7-induced events more significantly than VC concurrent supply only.






Similar content being viewed by others
References
Bagchi D, Bagchi M, Stohs SJ (2001) Chromium (VI)-induced oxidative stress, apoptotic cell death and modulation of p53 tumor suppressor gene. Mol Cell Biochem 222:149–158
Bagchi D, Stohs SJ, Downs BW, Bagchi M, Preuss HG (2002) Cytotoxicity and oxidative mechanisms of different forms of chromium. Toxicology 180:5–22
Banu SK, Stanley JA, Lee J, Stephen SD, Arosh JA, Hoyer PB, Burghardt RC (2011) Hexavalent chromium-induced apoptosis of granulosa cells involves selective sub-cellular translocation of Bcl-2 members, ERK1/2 and p53. Toxicol Appl Pharmacol 251:253–266
Beutler E (1969) Effect of flavin compounds on glutathione reductase activity: in vivo and in vitro studies. J Clin Investig 48:1957–1966
Bortner CD, Oldenburg NB, Cidlowski JA (1995) The role of DNA fragmentation in apoptosis. Trends Cell Biol 5:21–26
Bradberry SM, Vale JA (1999) Therapeutic review: is ascorbic acid of value in chromium poisoning and chromium dermatitis? J Toxicol Clin Toxicol 37:195–200
Cabaud PG, Wróblewski F, Ruggiero V (1958) Colorimetric measurement of lactic dehydrogenase activity of body fluids. Am J Clin Pathol 30:234–236
Cailla H, De Kaouel CLB, Roux D, Delaage M, Marti J (1982) Monoclonal antibodies to 5′-triphospho-(2′-5′) adenyladenosine oligonucleotides. Proc Natl Acad Sci 79:4742–4746
Chandra J, Samali A, Orrenius S (2000) Triggering and modulation of apoptosis by oxidative stress. Free Radic Biol Med 29:323–333
Costa M, Klein CB (2006) Toxicity and carcinogenicity of chromium compounds in humans. Crit Rev Toxicol 36:155–163
Dashti A, Soodi M, Amani N (2016) Cr (VI) induced oxidative stress and toxicity in cultured cerebellar granule neurons at different stages of development and protective effect of Rosmarinic acid. Environ Toxicol 31:269–277
Ellman GL, Courtney KD, Andres V, Featherstone RM (1961) A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem Pharmacol 7:88IN191–88I9095
Galluzzi L, Morselli E, Kepp O, Tajeddine N, Kroemer G (2008) Targeting p53 to mitochondria for cancer therapy. Cell Cycle 7:1949–1955
Gamble M, Wilson I (2008) The hematoxylins and eosin. Theory Pract Histol Tech 6:121–134
García-Niño WR, Zatarain-Barrón ZL, Hernández-Pando R, Vega-García CC, Tapia E, Pedraza-Chaverri J (2015) Oxidative stress markers and histological analysis in diverse organs from rats treated with a hepatotoxic dose of Cr (VI): effect of curcumin. Biol Trace Elem Res 167:130–145
Goldberg DM, Spooner RJ (1988) 3.7 glutathione reductase. Enzymes 1:258
Haupt S, Berger M, Goldberg Z, Haupt Y (2003) Apoptosis-the p53 network. J Cell Sci 116:4077–4085
Hussein MM, Ahmed MM (2016) The Th1/Th2 paradigm in lambda cyhalothrin-induced spleen toxicity: the role of thymoquinone. Environ Toxicol Pharmacol 41:14–21
Jayaraman T, Kannappan S, Ravichandran M, Anuradha C (2008) Impact of Essentiale L on ethanol-induced changes in rat brain and erythrocytes. Singap Med J 49:320
Jin Y, Liu Z, Liu F, Ye Y, Peng T, Fu Z (2015) Embryonic exposure to cadmium (II) and chromium (VI) induce behavioral alterations, oxidative stress and immunotoxicity in zebrafish (Danio Rerio). Neurotoxicol Teratol 48:9–17
Kesinger NG, Stevens JF (2009) Covalent interaction of ascorbic acid with natural products. Phytochemistry 70:1930–1939
Kim J-H, Kang J-C (2016) Oxidative stress, neurotoxicity, and metallothionein (MT) gene expression in juvenile rock fish Sebastes Schlegelii under the different levels of dietary chromium (Cr 6+) exposure. Ecotoxicol Environ Saf 125:78–84
Kook S-H, Son Y-O, Chung S-W, Lee S-A, Kim J-G, Jeon Y-M, Lee J-C (2007) Caspase-independent death of human osteosarcoma cells by flavonoids is driven by p53-mediated mitochondrial stress and nuclear translocation of AIF and endonuclease G. Apoptosis 12:1289–1298
Krim M, Messaadia A, Maidi I, Aouacheri O, Saka S (2013) Protective effect of ginger against toxicity induced by chromate in rats. Ann Biol Clin 7(2):165–173
Kucharski H, Zajac J (2009) Handbook of vitamin C research: daily requirements, dietary sources and adverse effects. Nova Science Publishers, Inc., New York
Lalaouni A, Henderson C, Kupper C, Grant M (2007) The interaction of chromium (VI) with macrophages: depletion of glutathione and inhibition of glutathione reductase. Toxicology 236:76–81
Lee KB, Kim K-R, Huh T-L, Lee YM (2008) Proton induces apoptosis of hypoxic tumor cells by the p53-dependent and p38/JNK MAPK signaling pathways. Int J Oncol 33:1247–1256
Levine RL, Garland D, Oliver CN, Amici A, Climent I, Lenz A-G, Ahn B-W, Shaltiel S, Stadtman ER (1990) [49] determination of carbonyl content in oxidatively modified proteins. Methods Enzymol 186:464–478
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Nair V, Turner GA (1984) The thiobarbituric acid test for lipid peroxidation: structure of the adduct with malondialdehyde. Lipids 19:804–805
Nickens KP, Patierno SR, Ceryak S (2010) Chromium genotoxicity: a double-edged sword. Chem Biol Interact 188:276–288
Nudler SI, Quinteros FA, Miler EA, Cabilla JP, Ronchetti SA, Duvilanski BH (2009) Chromium VI administration induces oxidative stress in hypothalamus and anterior pituitary gland from male rats. Toxicol Lett 185:187–192
O’Brien TJ, Ceryak S, Patierno SR (2003) Complexities of chromium carcinogenesis: role of cellular response, repair and recovery mechanisms. Mutat Res/Fundam Mol Mech Mutagen 533:3–36
Quinteros FA, Poliandri AH, Machiavelli LI, Cabilla JP, Duvilanski BH (2007) In vivo and in vitro effects of chromium VI on anterior pituitary hormone release and cell viability. Toxicol Appl Pharmacol 218:79–87
Quinteros FA, Machiavelli LI, Miler EA, Cabilla JP, Duvilanski BH (2008) Mechanisms of chromium (VI)-induced apoptosis in anterior pituitary cells. Toxicology 249:109–115
Qureshi IZ, Mahmood T (2010) Prospective role of ascorbic acid (vitamin C) in attenuating hexavalent chromium-induced functional and cellular damage in rat thyroid. Toxicol Ind Health 26:349–359
Rekha C, Poornima G, Manasa M, Abhipsa V, Devi P, Kumar V, Kekuda P (2012) Ascorbic acid, total phenol content and antioxidant activity of fresh juices of four ripe and unripe citrus fruits. Chem Sci Trans 1:303–310
Salnikow K, Zhitkovich A (2007) Genetic and epigenetic mechanisms in metal carcinogenesis and cocarcinogenesis: nickel, arsenic, and chromium. Chem Res Toxicol 21:28–44
Shi S-R, Key ME, Kalra KL (1991) Antigen retrieval in formalin-fixed, paraffin-embedded tissues: an enhancement method for immunohistochemical staining based on microwave oven heating of tissue sections. J Histochem Cytochem 39:741–748
Son Y-O, Hitron JA, Wang X, Chang Q, Pan J, Zhang Z, Liu J, Wang S, Lee J-C, Shi X (2010) Cr (VI) induces mitochondrial-mediated and caspase-dependent apoptosis through reactive oxygen species-mediated p53 activation in JB6 Cl41 cells. Toxicol Appl Pharmacol 245:226–235
Stearns DM, Kennedy LJ, Courtney KD, Giangrande PH, Phieffer LS, Wetterhahn KE (1995) Reduction of chromium (VI) by ascorbate leads to chromium-DNA binding and DNA strand breaks in vitro. Biochemistry 34:910–919
Subramanian S, Rajendiran G, Sekhar P, Gowri C, Govindarajulu P, Aruldhas MM (2006) Reproductive toxicity of chromium in adult bonnet monkeys (Macaca Radiata Geoffrey). Reversible oxidative stress in the semen. Toxicol Appl Pharmacol 215:237–249
Tamari Y, Nawata H, Inoue E, Yoshimura A, Yoshii H, Kashino G, Seki M, Enomoto T, Watanabe M, Tano K (2013) Protective roles of ascorbic acid in oxidative stress induced by depletion of superoxide dismutase in vertebrate cells. Free Radic Res 47:1–7
Trzeciak A, Kowalik J, Malecka-Panas E, Drzewoski J, Wojewódzka M, Iwanenko T, Blasiak J (2000) Genotoxicity of chromium in human gastric mucosa cells and peripheral blood lymphocytes evaluated by the single cell gel electrophoresis (comet assay). Med Sci Monit 6:24–29
Tsangaris C, Papathanasiou E, Cotou E (2007) Assessment of the impact of heavy metal pollution from a ferro-nickel smelting plant using biomarkers. Ecotoxicol Environ Saf 66:232–243
Tsao DA, Tseng WC, Chang HR (2011) The expression of RKIP, RhoGDI, galectin, c-Myc and p53 in gastrointestinal system of Cr (VI)-exposed rats. J Appl Toxicol 31:730–740
Viarengo A, Lowe D, Bolognesi C, Fabbri E, Koehler A (2007) The use of biomarkers in biomonitoring: a 2-tier approach assessing the level of pollutant-induced stress syndrome in sentinel organisms. Comp Biochem Physiol Part C: Toxicol Pharmacol 146:281–300
Wang X-F, Xing M-L, Shen Y, Zhu X, Xu L-H (2006) Oral administration of Cr (VI) induced oxidative stress, DNA damage and apoptotic cell death in mice. Toxicology 228:16–23
Wartelle LH, Marshall WE (2005) Chromate ion adsorption by agricultural by-products modified with dimethyloldihydroxyethylene urea and choline chloride. Water Res 39:2869–2876
Wilbur S, Ingerman L, Citra M, Osier M, Wohlers D (2000) Toxicological profile for chromium. US Department of Health and Human Services. Public health service, Agency for Toxic Substances and Disease Registry, Washington DC, pp 1–419
Zhong L-T, Sarafian T, Kane DJ, Charles AC, Mah SP, Edwards RH, Bredesen DE (1993) bcl-2 inhibits death of central neural cells induced by multiple agents. Proc Natl Acad Sci 90:4533–4537
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
All the experimental protocol is endorsed by the Approval of Ethics Committee of Faculty of Veterinary Medicine, Zagazig University, in accordance with the guiding principles of the National Institutes of Health (NIH) for the Care and Use of Laboratory Animals.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Responsible editor: Philippe Garrigues
Rights and permissions
About this article
Cite this article
Abu Zeid, E.H., Hussein, M.M.A. & Ali, H. Ascorbic acid protects male rat brain from oral potassium dichromate-induced oxdative DNA damage and apoptotic changes: the expression patterns of caspase-3, P 53, Bax, and Bcl-2 genes. Environ Sci Pollut Res 25, 13056–13066 (2018). https://doi.org/10.1007/s11356-018-1546-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-018-1546-9