Abstract
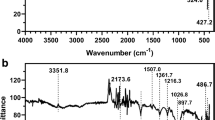
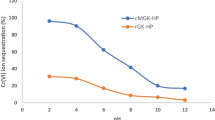
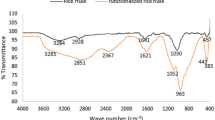
Heavy metal pollution in watercourses is a major environmental problem throughout the world due to rapid population growth, industrialization, and economic development. Considering this, the present study aimed to develop a new adsorbent from pumpkin husk (PH) by KOH modification to remove copper (Cu2+) ions and to explore its adsorptive potential. The sorption studies of Cu2+ on KOH-modified PH were carried out as functions of particle size, solution pH, adsorbent dose, temperature, initial metal concentration, and contact time. The sorption capacity of KOH-modified PH was found to be higher than that of raw PH, as 19.4 and 10.2 mg g−1, respectively. Morphology and surface structures of adsorbents were characterized by determination of zero point charge, a Fourier transform infrared spectrometer (FTIR–ATR) spectra, and a scanning electron microscopy (SEM) of PH powders before and after the sorption of Cu2+. The pHzpc of PH was found to be 5.0. FTIR–ATR analyses indicated that amino, amide, hydroxyl, carboxyl, and oxygenated groups of PH play an important role in the sorption process. Sorption isotherm, kinetic, and thermodynamic parameters of Cu2+ on KOH-modified PH were studied. The kinetic process was well represented by the Logistic model. The maximum sorption was found as 73.16 mg g−1 according to the well-fitting of Langmuir isotherm. Results of sorption and thermodynamic studies indicated that the process was exothermic, being feasible, and spontaneous. KOH-modified PH as an eco-friendly adsorbent had great potential to remove Cu2+ ions from aquatic system.




Similar content being viewed by others
References
Akpomie KG, Dawodua FA, Adebowale KO (2015) Mechanism on the sorption of heavy metals from binary-solution by a low cost montmorillonite and its desorption potential. Alexandria Eng J 54(3):757–767. https://doi.org/10.1016/j.aej.2015.03.025
Asuquo ED, Martin AD (2016) Sorption of cadmium (II) ion from aqueous solution onto sweet potato (Ipomoea batatas L.) peel adsorbent: characterization, kinetic and isotherm studies. J Environ Chem Eng 4(4):4207–4228. https://doi.org/10.1016/j.jece.2016.09.024
Awwad AM, Salem NM (2012) Biosorption of copper(II) and lead(II) ions from aqueous solutions by modified loquat (Eriobotrya japonica) leaves (MLL). J Chem Eng Mater Sci 3:7–17
Aydın H, Bulut Y, Yerlikaya C (2008) Removal of copper (II) from aqueous solution by adsorption onto low-cost adsorbents. J Environ Manag 87(1):37–45. https://doi.org/10.1016/j.jenvman.2007.01.005
Bakhtiari N, Azizian S (2015) Adsorption of copper ion from aqueous solution by nanoporous MOF-5: a kinetic and equilibrium study. J Molecul Liquids 206:114–118. https://doi.org/10.1016/j.molliq.2015.02.009
Balkaya A, Özbakir M, Kurtar ES (2010) The phenotypic diversity and fruit characterization of winter squash (Cucurbita Maxima) populations from the Black Sea region of Turkey. Afr J Biotechnol 9(2):152–162
Başçı N, Kocadağıstan E, Kocadağıstan B (2004) Biosorption of copper (II) from aqueous solutions by wheat shell. Desalination 164(2):135–140. https://doi.org/10.1016/S0011-9164(04)00172-9
Benaissa H, Elouchdi MA (2007) Removal of copper ions from aqueous solutions by dried sunflower leaves. Chem Eng Process 46(7):614–622. https://doi.org/10.1016/j.cep.2006.08.006
Ben-Ali S, Jaouali I, Souissi-Najar S, Ouederni A (2017) Characterization and adsorption capacity of raw pomegranate peel biosorbent for copper removal. J Clean Prod 142:3809–3821. https://doi.org/10.1016/j.jclepro.2016.10.081
Bilal M, Shah JA, Ashfaq T, Mubashar S, Gardazi H, Tahir AA, Pervez A, Haroon H, Mahmood Q (2013) Wastewater biomass adsorbents for copper removal from industrial wastewater-a review. J Hazard Mater 263:322–333. https://doi.org/10.1016/j.jhazmat.2013.07.071
Chen X, Chen G, Chen L, Chen Y, Lehmann J, McBride MB, Hay AG (2011) Adsorption of copper and zinc by biochars produced from pyrolysis of hardwood and corn straw in aqueous solution. Bioresour Technol 102(19):8877–8884. https://doi.org/10.1016/j.biortech.2011.06.078
Çelekli A, Bozkurt H (2013) Predictive modeling of an azo metal complex dye sorption by pumpkin husk. Environ Sci Pollut Res 20(10):7355–7366. https://doi.org/10.1007/s11356-013-1751-5
Çelekli A, Çelekli F, Çiçek E, Bozkurt H (2014) Predictive modeling of sorption and desorption of reactive azo dye by pumpkin husk. Environ Sci Pollut Res 21(7):5086–5097. https://doi.org/10.1007/s11356-013-2452-9
Çelekli A, Küçükgüner B, Bozkurt H (2016) Diazo dye sorption by Ni-modified pumpkin husk. Desalin Water Treat 57(52):25242–25255. https://doi.org/10.1080/19443994.2016.1149740
Çelekli A, Yavuzatmaca M, Bozkurt H (2010) An eco-friendly process: predictive modeling of copper adsorption from aqueous solution on Spirulina platensis. J Hazard Mater 173(1-3):123–129. https://doi.org/10.1016/j.jhazmat.2009.08.057
Dundar M, Nuhoglu Ç, Nuhoglu Y (2008) Biosorption of cu(II) ions onto the litter of natural trembling poplar forest. J Hazard Mater 151(1):86–95. https://doi.org/10.1016/j.jhazmat.2007.05.055
Elaigwu SE, Rocher V, Kyriakou G, Greenway GM (2014) Removal of Pb2+ and Cd2+ from aqueous solution using chars from pyrolysis and microwave-assisted hydrothermal carbonization of Prosopis africana shell. J Ind Eng Chem 20(5):3467–3473. https://doi.org/10.1016/j.jiec.2013.12.036
Esmaeili A, Najmeh K (2016) Optimization of process parameters for removal of heavy metals by biomass of cu and co-doped alginate-coated chitosan nanoparticles. Bioresour Technol 218:650–658. https://doi.org/10.1016/j.biortech.2016.07.005
FAO (2010) FAO–food and agriculture organization of the United Nations. Statistical Database, Rome
Feng N, Guo X, Liang S (2009) Adsorption study of copper (II) by chemically modified orange peel. J Hazard Mater 164(2–3):1286–1292. https://doi.org/10.1016/j.jhazmat.2008.09.096
Fernandez ME, Ledesma B, Román S, Bonelli PR, Cukierman AL (2015) Development and characterization of activated hydrochars from orange peels as potential adsorbents for emerging organic contaminants. Bioresour Technol 183:221–228. https://doi.org/10.1016/j.biortech.2015.02.035
Fouodjouo M, Fotouo-Nkaffo H, Laminsi S, Cassini FA, de Brito-Benetoli LO, Debacher NA (2017) Adsorption of copper (II) onto cameroonian clay modified by non-thermal plasma: characterization, chemical equilibrium and thermodynamic studies part a. Appl Clay Sci 142:136–144. https://doi.org/10.1016/j.clay.2016.09.028
Freundlich H (1906) Over the adsorption in solution. J Phys Chem 57:385–471
Guo XY, Liang S, Tian QH (2011) Removal of heavy metal ions from aqueous solutions by adsorption using modified orange peel as adsorbent. Adv Mater Res 236–238:237–240
Gupta VK, Agarwal S, Saleh TA (2011) Synthesis and characterization of alumina-coated carbon nanotubes and their application for lead removal. J Hazard Mater 185(1):17–23. https://doi.org/10.1016/j.jhazmat.2010.08.053
Gupta VK, Ali I, Saleh TA, Nayak A, Agarwal S (2012) Chemical treatment technologies for waste-water recycling—an overview. RSC Adv 2(16):6380–6388. https://doi.org/10.1039/c2ra20340e
Gupta VK, Nayak A (2012) Cadmium removal and recovery from aqueous solutions by novel adsorbents prepared from orange peel and Fe2O3 nanoparticles. Chem Eng J 180:81–90. https://doi.org/10.1016/j.cej.2011.11.006
Gupta VK, Saleh TA (2013) Sorption of pollutants by porous carbon, carbon nanotubes and fullerene - an overview. Environ Sci Pollut Res 20(5):2828–2843. https://doi.org/10.1007/s11356-013-1524-1
Gupta VK, Nayak A, Agarwal S (2015) Bioadsorbents for remediation of heavy metals: current status and their future prospects. Environ Eng Res 20(1):001–018. https://doi.org/10.4491/eer.2015.018
Hesas RH, Arami-Niya A, Wan Daud WMA, Sahu JN (2013) Preparation and characterization of activated carbon from apple waste by microwave-assisted phosphoric acid activation: application in methylene blue adsorption. Bioresources 8:2950–2966
Ho YS, McKay G (1999) Pseudo-second order model for sorption processes. Process Biochem 34(5):451–465. https://doi.org/10.1016/S0032-9592(98)00112-5
Iqbal M, Saeed A, Kalim I (2009) Characterization of adsorptive capacity and investigation of mechanism of Cu2+, Ni2+ and Zn2+ adsorption on mango peel waste from constituted metal solution and genuine electroplating effluent. Sep Sci Technol 44(15):3770–3791. https://doi.org/10.1080/01496390903182305
Izquierdo M, Marzal P, Lens PNL (2013) Effect of organic ligands on copper(II) removal from metal plating wastewater by orange peel-based biosorbents. Water Air Soil Pollut 224:1–15
İmamoğlu M, Tekir O (2008) Removal of copper (II) and lead (II) ions from aqueous solutions by adsorption on activated carbon from a new precursor hazelnut husks. Desalination 228(1-3):108–113. https://doi.org/10.1016/j.desal.2007.08.011
Johnson PD, Watson MA, Brown J, Jefcoat IA (2002) Peanut hull pellets as a single use sorbent for the capture of cu(II) from wastewater. Waste Manag 22(5):471–480. https://doi.org/10.1016/S0956-053X(01)00036-8
Kazemipour M, Ansari M, Tajrobehkar S, Majdzadeh M, Kermani HR (2008) Removal of Pb(II), cadmium, zinc, and copper from industrial wastewater by carbon developed from walnut, hazelnut, almond, pistachio shell, and apricot stone. J Hazard Mater 150(2):322–327. https://doi.org/10.1016/j.jhazmat.2007.04.118
Kilic M, Yazici H, Solak M (2009) A comprehensive study on removal and recovery of copper(II) from aqueous solutions by NaOH-pretreated Marrubium globosum sp. globosum leaves powder: potential for utilizing the copper(II) condensed desorption solutions in agricultural applications. Bioresour Technol 100(7):2130–2137. https://doi.org/10.1016/j.biortech.2008.11.002
Krishnani KK, Meng X, Christodoulatos C, Boddu VM (2008) Biosorption mechanism of nine different heavy metals onto biomatrix from rice husk. J Hazard Mater 153(3):1222–1234. https://doi.org/10.1016/j.jhazmat.2007.09.113
Labidi A, Salaberria AM, Fernandes SCM, Labidi J, Abderrabba M (2016) Adsorption of copper on chitin-based materials: kinetic and thermodynamic studies. J Taiwan Inst Chem Eng 65:140–148. https://doi.org/10.1016/j.jtice.2016.04.030
Langmuir I (1918) The adsorption of gases on plane surfaces of glass, mica and platinum. J Am Chem Soc 4:1361–1403
Liang S, Guo X, Feng N, Tian Q (2010) Isotherms, kinetics and thermodynamic studies of adsorption of Cu2+ from aqueous solutions by Mg2+/K+ type orange peel adsorbents. J Hazard Mater 174(1-3):756–762. https://doi.org/10.1016/j.jhazmat.2009.09.116
Liu Y, Cao Q, Luo F, Chen J (2009) Biosorption of Cd2+, Cu2+, Ni2+ and Zn2+ ions from aqueous solutions by pretreated biomass of brown algae. J Hazard Mater 163(2-3):931–938. https://doi.org/10.1016/j.jhazmat.2008.07.046
Pfeiffer CC, Mailloux R (1987) Excess copper as a factor in human diseases. J Orthomol Med 2(3):171–182
Regmi P, Moscoso JLG, Kumar S, Cao X, Mao J, Schafran G (2012) Removal of copper and cadmium from aqueous solution using switchgrass biochar produced via hydrothermal carbonization process. J Environ Manag 109:61–69. https://doi.org/10.1016/j.jenvman.2012.04.047
Romero-Cano LA, Gonzalez-Gutierrez LV, Baldenegro-Perez LA (2016) Biosorbents prepared from orange peels using instant controlled pressure drop for cu(II) and phenol removal. Ind Crop Prod 84:344–349. https://doi.org/10.1016/j.indcrop.2016.02.027
Sabela MI, Kunene K, Kanchi S, Xhakaza NM, Bathinapatla A, Mdluli P, Sharma D, Bisetty K (2018) Removal of copper (II) from wastewater using green vegetable waste derived activated carbon: an approach to equilibrium and kinetic study. Arab J Chem. https://doi.org/10.1016/j.arabjc.2016.06.001
Saleh TA, Gupta VK (2014) Processing methods, characteristics and adsorption behavior of tire derived carbons: a review. Adv Colloid Interf Sci 211:93–101. https://doi.org/10.1016/j.cis.2014.06.006
Semerciöz S (2015) Development of new carbonaceous material from grapefruit peel for the adsorption of Cu+2 ions. Master Thesis, Graduate School of Natural and Apllied Science, University of Gaziantep, Turkey
Semerciöz AS, Göğüş F, Çelekli A, Bozkurt H (2017) Development of carbonaceous material from grapefruit peel with microwave implemented-low temperature hydrothermal carbonization technique for the adsorption of cu (II). J Clean Prod 165:599e610
Tasaso P (2014) Adsorption of copper using pomelo peel and depectinated pomelo peel. J Clean Energy Technol 2:154–157
Arief VO, Trilestari K, Sunarso J, Indraswati N, Ismadji S (2008) Recent progress on biosorption of heavy metals from liquids using low cost biosorbents: characterization, biosorption parameters and mechanism studies. Clean-Soil Air Water 36(12):937–962. https://doi.org/10.1002/clen.200800167
Zhu B, Fan T, Zhang D (2008) Adsorption of copper ions from aqueous solution by citric acid modified soybean straw. J Hazard Mater 153(1-2):300–308. https://doi.org/10.1016/j.jhazmat.2007.08.050
Acknowledgements
Authors thank Scientific Research Projects Executive Councils of University of Gaziantep and DPT (T.R. Prime Ministry State Planning Organization).
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Guilherme L. Dotto
Rights and permissions
About this article
Cite this article
Çelekli, A., Bozkuş, B. & Bozkurt, H. Development of a new adsorbent from pumpkin husk by KOH-modification to remove copper ions. Environ Sci Pollut Res 26, 11514–11523 (2019). https://doi.org/10.1007/s11356-017-1160-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-017-1160-2