Abstract
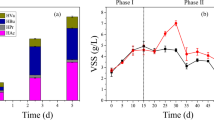
The main objective of this work was to investigate the influence of different oxygen supply patterns on poly-β-hydroxybutyrate (PHB) yield and bacterial community diversity. The anaerobic–aerobic (A/O) sequencing batch reactors (SBR1) and feast–famine (F/F) SBR2 were used to cultivate activated sludge to produce PHB. The mixed microbial communities were collected and analyzed after 3 months cultivation. The PHB maximum yield was 64 wt% in SBR1 and 53 wt% in SBR2. Pyrosequencing analysis 16S rRNA gene of two microbial communities indicated there were nine and four bacterial phyla in SBR1 and SBR2, respectively. Specifically, Proteobacteria (36.4 % of the total bacterial community), Actinobacteria (19.7 %), Acidobacteria (14.1 %), Firmicutes (4.4 %), Bacteroidetes (1.7 %), Cyanobacteria/Chloroplast (1.5 %), TM7 (0.8 %), Gemmatimonadetes (0.2 %), and Nitrospirae (0.1 %) were present in SBR1. Proteobacteria (94.2 %), Bacteroidetes (2.9 %), Firmicutes (1.9 %), and Actinobacteria (0.7 %) were present in SBR2. Our results indicated the SBR1 fermentation system was more stable than that of SBR2 for PHB accumulation.





Similar content being viewed by others
References
APHA (2001) Standard methods for the examination of water and wastewater, 21st ed. American Public Health Association, Washington, DC
Chang HF, Chang WC, Tsai CY (2012) Synthesis of poly(3-hydroxybutyrate/3-hydroxyvalerate) from propionate-fed activated sludge under various carbon sources. Bioresour Technol 113:51–57
Chen G (2010) Plastics from bacteria: natural functions and applications. In: Chen G (ed) Plastics completely synthesized by bacteria: polyhydroxyalkanoates. Springer, Berlin Heidelberg, pp 17–37
Chen GQ, Hajnal I, Wu H, Lv L, Ye JW (2015) Engineering biosynthesis mechanisms for diversifying polyhydroxyalkanoates. Trends Biotechnol 33(10):565–574
Choi J, Lee SY (1999) Efficient and economical recovery of poly(3-hydroxybutyrate) from recombinant Escherichia coli by simple digestion with chemicals. Biotechnol Bioeng 62:546–553
Ciesielski S, Możejko J, Pisutpaisal N (2014) Plant oils as promising substrates for polyhydroxyalkanoates production. J Clean Prod. doi:10.1016/j.jclepro.2014.09.040
Dai J, Gliniewicz K, Settles ML, Coats ER, McDonald AG (2015) Influence of organic loading rate and solid retention time on polyhydroxybutyrate production from hybrid poplar hydrolysates using mixed microbial cultures. Bioresour Technol 175:23–33
Dalal J, Priyangshu MS, Lavania M, Mandal AK, Lal B (2013) Evaluation of bacterial strains isolated from oil-contaminated soil for production of polyhydroxyalkanoic acids (PHA). Pedobiologia 54:25–30
Dietrich D, Illman B, Crooks C (2013) Differential sensitivity of polyhydroxyalkanoate producing bacteria to fermentation inhibitors and comparison of polyhydroxybutyrate production from Burkholderia cepacia and Pseudomonas pseudoflava. BMC Res 219:1–4
Fradinho JC, Oehmen A, Reis MAM (2014) Photosynthetic mixed culture polyhydroxyalkanoate (PHA) production from individual and mixed volatile fatty acids (VFAs): substrate preferences and co-substrate uptake. J Biotechnol 185:19–27
Haba E, Vidal-Mas J, Bassas M, Espuny MJ, Llorens J, Manresa A (2007) Poly 3-(hydroxyalkanoates) produced from oily substrates by Pseudomonas aeruginosa 47T2 (NCBIM 40044): effect of nutrients and incubation temperature on polymer composition. Biochem Eng J 35(2):99–106
Helena MG, Robbert K, Anuska MC, Mark CM, Loosdrecht (2013) Impact of oxygen limitation on glycerol-based biopolymer production by bacterial enrichments. Water Res 47:1209–1217
Jiang Y, Marang L, Kleerebezem R, Muyzer G, Loosdrecht M (2011) Polyhydroxybutyrate production from lactate using a mixed microbial culture. Biotechnol Bioeng 108:2022–2035
Johnson K, Kleerebezem R, Mark CML (2010) Influence of ammonium on the accumulation of polyhydroxybutyrate (PHB) in aerobic open mixed cultures. J Biotechnol 147:73–79
Lemos P, Levantesi C, Serafim L, Rossetti S, Reis M, Tandoi V (2008) Microbial characterization of polyhydroxyalkanoates storing populations selected under different operating conditions using a cell-sorting RT-PCR approach. Appl Microbiol Biotechnol 78:351–360
Lim CP, Zhang S, Zhou Y, Ng WJ (2015) Enhanced carbon capture biosorption through process manipulation. Biochem Eng J 93:128–136
Liu C, Wang H, Xing W, Wei L (2013) Composition diversity and nutrition conditions for accumulation polyhydroxyalkanoate (PHA) in a bacterial community from activated sludge. Appl Microbiol Biotechnol 97(21):9377–9387
Mohan SV, Srikanth S, Velvizhi G., Babu ML (2013) Microbial fuel cells for sustainable bioenergy generation: principles and perspective applications. Biofuel Technol. 335–368
Moita R, Lemos PC (2012) Biopolymers production from mixed cultures and pyrolysis by-products. J Biotechnol 157:578–583
Moita R, Freches A, Lemos PC (2014) Crude glycerol as feedstock for polyhydroxyalkanoates production by mixed microbial cultures. Water Res 58:9–21
Morgan SF, Hjort M, Cirne D (2015) Integrated production of polyhydroxyalkanoates (PHAs) with municipal wastewater and sludge treatment at pilot scale. Bioresour Technol 181:78–89
Queiros D, Rossetti S, Serafim LS (2014) PHA production by mixed cultures: a way to valorize wastes from pulp industry. Bioresour Technol 157:197–205
Reddy MV, Mohan SV (2012) Effect of substrate load and nutrients concentration on the polyhydroxyalkanoates (PHA) production using mixed consortia through wastewater treatment. Bioresour Technol 114:573–582
Schloss PD, Westcott SL (2009) Introducing Mothur: open-source, platform independent, community supported software for describing and comparing microbial communities. Appl Environ Microbiol 75:7537–7541
Takaichi S, Takashi M, Kazuto T, Satoshi H (2010) Carotenoids of Gemmatimonas aurantiaca (Gemmatimonadetes): identification of a novel carotenoid, deoxyoscillol 2-rhamnoside, and proposed biosynthetic pathway of oscillol 2,2-dirhamnoside. Microbiol 156:757–763
Tanaka K, Miyawai K, Yamaguchi A, Khosravi-Darani K, Matsusaki H (2011) Cell growth and P(3HB) accumulation from CO2 of a carbon monoxide-tolerant hydrogen-oxidizing bacterium, Ideonella sp. O-1. Appl Microbiol Biotechnol 92:1161–1169
Thuoc DV, Quillaguaman J (2014) Improving culture conditions for poly(3-hydroxybutyrate- co-3-hydroxyvalerate) production by Bacillus sp. ND153, a bacterium isolated from a mangrove forest in Vietnam. Ann Microbiol 64:991–997
Wang Y, Geng J, Guo G, Wang C, Liu S (2011) N2O production in anaerobic/anoxic denitrifying phosphorus removal process: the effects of carbon sources shock. Chem Eng J 172:999–1007
Wang DB, Chen YG, Zheng X, Li X, Feng LY (2013a) Short-chain fatty acid production from different biological phosphorus removal sludges: the influences of PHA and gram-staining bacteria. Environ Sci Technol 47(6):2688–2695
Wang YP, Cai JY, Lan JH, Liu ZG, He N, Shen L, Li QB (2013b) Biosynthesis of poly(hydroxybutyrate-hydroxyvalerate) from the acclimated activated sludge and microbial characterization in this process. Bioresour Technol 148:61–69
Wang DB, Zeng GM, Chen YG, Li XM (2015a) Effect of polyhydroxyalkanoates on dark fermentative hydrogen production from waste activated sludge. Water Res 73:311–322
Wang DB, Zhao JW, Zeng GM, Chen YG, Bond PL, Li XM (2015b) How does poly(hydroxyalkanoate) affect methane production from the anaerobic digestion of waste-activated sludge? Environ Sci Technol 49(20):12253–12262
Yang Y, Quensen J, Mathieu J, Wang Q, Wang J, Li M, James MT, Pedro JJ (2014) Alvarez Pyrosequencing reveals higher impact of silver nanoparticles than AgD on the microbial community structure of activated sludge. Water Res 48:317–325
Yoram B, Jaap R (2000) Atypical polyphosphate accumulation by the denitrifying bacterium Paracoccus denitrificans. Appl Environ Microbiol 66(3):1209–1212
Zakaria MM, Ariffin H, Hassan MA (2014) Bio-based production of crotonic acid by pyrolysis of poly(3-hydroxybutyrate) inclusions. J Clean Prod 83:463–472
Acknowledgments
This work was supported by NEFU biology base scientific research training and ability project (J1210053, J0106).
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Gerald Thouand
Highlights
• The maximum PHB yields were 64 wt% in SBR1 and 53 wt% in SBR2.
• The stability of microbial community in SBR1 was better than SBR2.
• The diversity of microbial community in SBR1 was richer than SBR2.
Rights and permissions
About this article
Cite this article
Liu, C., Liu, D., Qi, Y. et al. The effect of anaerobic–aerobic and feast–famine cultivation pattern on bacterial diversity during poly-β-hydroxybutyrate production from domestic sewage sludge. Environ Sci Pollut Res 23, 12966–12975 (2016). https://doi.org/10.1007/s11356-016-6345-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-016-6345-6