Abstract
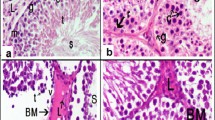
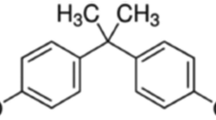
The current study presents the bisphenol A exposure and the ameliorative effects of Adiantum capillus-veneris on testicular toxicity induced by bisphenol A. Adult male albino rats were divided into five groups of five animals each: A (control), B (vehicle control), C (toxic), D (protective), and E (ameliorative) were served distilled water, olive oil, bisphenol A (BPA) at 100 mg/kg body weight, A. capillus-veneris plant extract at 25 mg/kg body weight, and BPA + A. capillus-veneris, respectively. All of the doses were administered orally for 15 days, and the rats were then sacrificed. Blood samples for the testosterone assay and both testes were collected for histological examination. The body weight, paired testes weight, relative tissue weight index, Johnsen scoring of tubules, and level of serum testosterone decreased in BPA-treated rats. Similarly, histological examination of the testes in BPA-treated animals revealed a lower number of Leydig cells, an irregular basement membrane, sloughing of germinal layers, vacuolization, a lower number of spermatocytes, and debris in the lumen. However, co-administration of A. capillus-veneris with BPA increased the total antioxidative capacity (330.82 ± 22.46 μmol/mg protein) of the testes and restored the serum testosterone level (1.70 ng/ml); histological features showed restoration in the stages of spermatogenesis. Conclusively, A. capillus-veneris plant extract overcomes the estrogenic effects of BPA on the reproductive system of rats and protects rats’ testes against BPA-induced injury/damage via an antioxidative mechanism that appears to be conciliated.





Similar content being viewed by others
References
Abbasi AM, Khan MA, Ahmad M, Qureshi R, Arshad M, Jahan S, Zafar M, Sultan S (2010) Ethnobotanical study of wound healing herbs among the tribal communities in Northern Himalaya ranges District Abbottabad, Pakistan. Pak J Bot 42:3747–3753, http://www.pakbs.org/pjbot/PDFs/42%286%29/PJB42%286%293747.pdf
Abd El Tawab AM, Shahin NN, AbdelMohsen MM (2014) Protective effect of Satureja montana extract on cyclophosphamide-induced testicular injury in rats. Chem Biol Interact 224:196–205. doi:10.1016/j.cbi.2014.11.001
Abdel-Halim OB, Ibraheim ZZ, Shiojima K (2002) Oleanane triterpenes from Adiantum capillus-veneris growing in Egypt. Alex J Pharm Sci 16:87–92, http://www.scopus.com/record/display.url?eid=2-s2.0-79952702832&origin=inward&txGid=C47B1BFD6AA064659618EB34B6E7FFED.kqQeWtawXauCyC8ghhRGJg%3a8
Ahmad I, Hussain M, Ahmad MSA, Ashraf MY, Ahmad R, Ali A (2008) Spatio-temporal variations in physiochemical attributes of Adiantum capillus-veneris from Soone valley of salt range (Pakistan). Pak J Bot 40:1387–1398, https://www.researchgate.net/profile/Muhammad_Ashraf18/publication/26541879_SPATIO-TEMPORAL_VARIATIONS_IN_PHYSIOCHEMICAL_ATTRIBUTES_OF_ADIANTUM_CAPILLUS_VENERIS_FROM_SOONE_VALLEY_OF_SALT_RANGE_%28PAKISTAN%29/links/02e7e5178a45467117000000.pdf
Ahmed A, Wadud A, Jahan N, Bilal A, Hajera S (2013) Efficacy of Adiantum Capillus veneris Linn in chemically induced urolithiasis in rats. J Ethnopharmacol 146:411–416. doi:10.1016/j.jep.2013.01.011
Akingbemi BT (2005) Estrogen regulation of testicular function. Reprod Biol Endocrinol 3:51. doi:10.1186/1477-7827-3-51
Akingbemi BT, Sottas CM, Koulova AI, Klinefelter GR, Hardy MP (2004) Inhibition of testicular steroidogenesis by the xenoestrogen bisphenol A is associated with reduced pituitary luteinizing hormone secretion and decreased steroidogenic enzyme gene expression in rat Leydig cells. Endocrinology 145:592–603. doi:10.1210/en.2003-1174
Al-Hiyasat AS, Darmani H, Elbetieha AM (2002) Effects of bisphenol A on adult male mouse fertility. Eur J Oral Sci 110:163–167. doi:10.1034/j.1600-0722.2002.11201.x
Ansari R, Ekhlasi-Kazaj K (2012) Adiantum capillus-veneris. L: phytochemical constituents, traditional uses and pharmacological properties: a review. J Adv Sci Res 3:15–20
Bancroft JD, Gamble M (2008) Theory and practice of histological techniques, 6th edn. Elsevier Health Sciences, London
Beeton C, Garcia A, Chandy KG (2007) Drawing blood from rats through the saphenous vein and by cardiac puncture. J Vis Exp 266. doi:10.3791/266
Benzie IF, Strain JJ (1996) The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”: the FRAP assay. Anal Biochem 239:70–76
Bhuiyan AS, Badrun N, Quamrun N (2001) Effects of Sumithion on the histological changes of spotted Murrel, Channa punctatus (bloch). Pak J Biol Sci 4:1288–1290. doi:10.3923/pjbs.2001.1288.1290
Biedermann S, Tschudin P, Grob K (2010) Transfer of bisphenol A from thermal printer paper to the skin. Anal Bioanal Chem 398:571–576. doi:10.1007/s00216-010-3936-9
Biles JE, McNeal TP, Begley TH, Hollifield HC (1997) Determination of bisphenol-A in reusable polycarbonate food-contact plastics and migration to food-simulating liquids. J Agric 45:3541–3544. doi:10.1021/jf970072i
Bindhumol V, Chitra KC, Mathur PP (2003) Bisphenol A induces reactive oxygen species generation in the liver of male rats. Toxicology 188:117–124. doi:10.1016/S0300-483X(03)00056-8
Brotons J a, Olea-Serrano MF, Villalobos M, Pedraza V, Olea N (1995) Xenoestrogens released from lacquer coatings in food cans. Environ Health Perspect 103:608–612. doi:10.1289/ehp.95103608
Camejo-Rodrigues J, Ascensão L, Bonet MÀ, Vallès J (2003) An ethnobotanical study of medicinal and aromatic plants in the Natural Park of “Serra de São Mamede” (Portugal). J Ethnopharmacol 89:199–209. doi:10.1016/S0378-8741(03)00270-8
Chapin RE, Delaney J, Wang Y, Lanning L, Davis B, Collins B, Mintz N, Wolfe G (1999) The effects of 4-nonylphenol in rats: a multigeneration reproduction study. Toxicol Sci 52:80–91, http://toxsci.oxfordjournals.org/content/52/1/80.full.pdf±html
Chitra KC, Latchoumycandane C, Mathur PP (2003) Induction of oxidative stress by bisphenol A in the epididymal sperm of rats. Toxicology 185:119–127. doi:10.1016/S0300-483X(02)00597-8
Dastagir G, Imtiaz-ul-Haq, Malik ZH (1999) Medicinal plants of Mai Dhani hill Muzaffarabad (AJK), Pakistan. Pak J Bio Sci 2:185–191, http://docsdrive.com/pdfs/ansinet/pjbs/1999/185-191.pdf
De Natale A, Pollio A (2007) Plants species in the folk medicine of Montecorvino Rovella (inland Campania, Italy). J Ethnopharmacol 109:295–303. doi:10.1016/j.jep.2006.07.038
Doshi T, D’Souza C, Vanage G (2013) Aberrant DNA methylation at Igf2-H19 imprinting control region in spermatozoa upon neonatal exposure to bisphenol A and its association with post implantation loss. Mol Biol Rep 40:4747–4757. doi:10.1007/s11033-013-2571-x
El-Ghazzawy IF, Meleis AE, Farghaly EF, Solaiman A (2011) Histological study of the possible protective effect of pomegranate juice on bisphenol-A induced changes of the caput epididymal epithelium and sperms of adult albino rats. Alex J Med 47:125–137. doi:10.1016/j.ajme.2011.06.006
El-Tantawy M, El-Sakhawy F, El-Deeb K, Fathy M, Hassan A (1994) A phytochemical and pharmacological study of Adiantum capillus-veneris L. growing in Egypt. Zagazig J Pharm Sci 3:97–103
European Food Safety Authority (EFSA), 2013. Public consultation on the draft opinion on bisphenol A (BPA)—exposure assessment 314 pages, http://www.efsa.europa. eu/en/consultations/call/130725.htm (accessed 23 August 2013)
European Union (EU) (2003) European Union Risk Assessment Report. Bisphenol A, CAS No: 80-05-7. Institute for Health and Consumer Protection, European Chemicals Bureau, European Commission Joint Research Centre, 3rd Priority List. Luxembourg: Office for Official Publications of the European Communities
Gaikwad K, Dhande S, Joshi YM, Kadam V (2013) Protective Effect of Adiantum capillus against chemically induced oxidative stress by cisplatin. J Appl Pharm Sci 3:65–68. doi:10.7324/JAPS.2013.30211
Geens T, Dirtu AC, Dirinck E, Malarvannan G, Van Gaal L, Jorens PG, Covaci A (2015) Daily intake of bisphenol A and triclosan and their association with anthropometric data, thyroid hormones and weight loss in overweight and obese individuals. Environ Int 76:98–105. doi:10.1016/j.envint.2014.12.003
Ghorbani A (2005) Studies on pharmaceutical ethnobotany in the region of Turkmen Sahra, north of Iran (Part 1): general results. J Ethnopharmacol 102:58–68. doi:10.1016/j.jep.2005.05.035
Guarrera PM, Lucchese F, Medori S (2008) Ethnophytotherapeutical research in the high Molise region (Central-Southern Italy). J Ethnobiol Ethnomed 4:7. doi:10.1186/1746-4269-4-7
Gurmeet KSS, Rosnah I, Normadiah MK, Das S, Mustafa AM (2014) Detrimental effects of bisphenol A on development and functions of the male reproductive system in experimental rats. EXCLI J 13:151–160
Haider S, Nazreen S, Alam MM, Gupta A, Hamid H, Alam MS (2011) Anti-inflammatory and anti-nociceptive activities of ethanolic extract and its various fractions from Adiantum capillus veneris Linn. J Ethnopharmacol 138:741–747. doi:10.1016/j.jep.2011.10.012
Haider S, Kharbanda C, Alam MS, Hamid H, Ali M, Alam M, Nazreen S, Ali Y (2013) Anti-inflammatory and anti-nociceptive activities of two new triterpenoids from Adiantum capillus-veneris Linn. Nat Prod Res 27:2304–2310. doi:10.1080/14786419.2013.828292
Hamayun M, Khan SA, Sohn EY, Lee IJ (2006) Folk medicinal knowledge and conservation status of some economically valued medicinal plants of District Swat, Pakistan. Lyonia 11:101–113, http://www.lyonia.org/downloadPDF.php?pdfID=262.497.1
Hassan AH, Ismail AA, Khudir AN (2013) Effects of pre-and postnatal exposure to bisphenol-A on the reproductive efficacy in male albino rats. J Kerbala Univ 11:158–172, http://www.iasj.net/iasj?func=fulltext&aId=79084
Hinton DE, Baumann PC, Gardener WE, Hawkins WE, Hendricks JD (1992) Histopathologic biomarkers. In: Huggett RJ, KImerie RA, Mehrie PM, Bergman HL (eds) Biochemical, physiological and histological markers of anthropogenic stress. Lewis Publishers, Boaco Rato, pp 155–209
Honma S, Suzuki A, Buchanan DL, Katsu Y, Watanabe H, Iguchi T (2002) Low dose effect of in utero exposure to bisphenol A and diethylstilbestrol on female mouse reproduction. Reprod Toxicol 16:117–122. doi:10.1016/S0890-6238(02)00006-0
Hulak M, Gazo I, Shaliutina A, Linhartova P (2013) In vitro effects of bisphenol A on the quality parameters, oxidative stress, DNA integrity and adenosine triphosphate content in sterlet (Acipenser ruthenus) spermatozoa. Comp Biochem Physiol C Toxicol Pharmacol 158:64–71. doi:10.1016/j.cbpc.2013.05.002
Ibraheim ZZ, Ahmed AS, Gouda YG (2011) Phytochemical and biological studies of Adiantum. Saudi Pharm J 19:65–74. doi:10.1016/j.jsps.2011.01.007
Johnsen SG (1970) Testicular biopsy score count—a method for registration of spermatogenesis in human testes: normal values and results in 335 hypogonadal males. Hormones 1:2–25. doi:10.1159/000178170
Kabuto H, Hasuike S, Minagawa N, Shishibori T (2003) Effects of bisphenol A on the metabolisms of active oxygen species in mouse tissues. Environ Res 93:31–35. doi:10.1016/S0013-9351(03)00062-8
Kabuto H, Amakawa M, Shishibori T (2004) Exposure to bisphenol A during embryonic/fetal life and infancy increases oxidative injury and causes underdevelopment of the brain and testis in mice. Life Sci 74:2931–2940. doi:10.1016/j.lfs.2003.07.060
Korkmaz A, Ahbab M a, Kolankaya D, Barlas N (2010) Influence of vitamin C on bisphenol A, nonylphenol and octylphenol induced oxidative damages in liver of male rats. Food Chem Toxicol 48:2865–2871. doi:10.1016/j.fct.2010.07.019
Kumar A (2009) antioxidant effect of Adiantum capillus veneris Linn. on human lymphocyte : an in vitro study 9, 1899–1902.
Kumar A, Aushik P (1999) Antibacterial effect of Adiantum capillus-veneris Linn. Ind Fern J 16:72–74
Maamar MB, Lesné L, Desdoits-Lethimonier C, Coiffec I, Lassurguère J, Lavoué V, Deceuninck Y, Antignac J-P, Le Bizec B, Perdu E, Zalko D, Pineau C, Chevrier C, Dejucq-Rainsford N, Mazaud-Guittot S, Jégou B (2015) An Investigation of the endocrine-disruptive effects of bisphenol A in human and rat fetal testes. PLoS One 10:e0117226. doi:10.1371/journal.pone.0117226
Machholz E, Mulder G, Ruiz C, Corning BF, Pritchett-Corning KR (2012) Manual restraint and common compound administration routes in mice and rats. J Vis Exp 1–8. doi:10.3791/2771
McGaw LJ, Lall N, Meyer JJM, Eloff JN (2008) The potential of South African plants against Mycobacterium infections. J Ethnopharmacol 119:482–500. doi:10.1016/j.jep.2008.08.022
Meyers TR, Hendricks JD (1985) Histopathology. In: Rand GM, Petrocelli SR (eds) Fundamental of aquatic toxicology. Hemisphere, New York, pp 283–331
Miao S, Gao Z, Kou Z, Xu G, Su C, Liu N (2008) Influence of bisphenol A on Developing rat estrogen receptors and some cytokines in rats: a two-generational study. J Toxicol Environ Health A 71:1000–8. doi:10.1080/15287390801907467
Mihail CM, Marghitas L Al, Bobisi O, Dezmireanl D (2010) Estimation of flavonoid content in propolis by two different colorimetric methods. Journal of food and analysis 43, 407–410.
Mourad IM, Khadrawy YA (2012) The sensitivity of liver, kidney and testis of rats to oxidative stress induced by different doses of bisphenol A. Int J Life Sci Pharma Res 2(2):19–28, http://www.ijlpr.com/admin/php/uploads/61_pdf.pdf
Nagao T, Saito Y, Usumi K, Yoshimura S, Ono H (2002) Low-dose bisphenol A does not affect reproductive organs in estrogen-sensitive C57BL/6N mice exposed at the sexually mature, juvenile, or embryonic stage. Reprod Toxicol 16:123–130. doi:10.1016/S0890-6238(02)00003-5
Nanjappa MK, Simon L, Akingbemi BT (2012) The industrial chemical bisphenol A (BPA) interferes with proliferative activity and development of steroidogenic capacity in rat Leydig cells. Biol Reprod 86:135–135. doi:10.1095/biolreprod.111.095349
Norazit A, Mohamad J, Razak SA, Abdulla MA, Azmil A, Mohd MA (2012) Effects of soya bean extract, bisphenol A and 17β-estradiol on the testis and circulating levels of testosterone and estradiol among peripubertal juvenile male Sprague-Dawley rats. Sains Malaysiana 41:63–69
Noubarani M, Rostamkhani H, Erfan M, Kamalinejad M, Eskandari MR, Babaeian M, Salamzadeh J (2014) Effect of Adiantum capillus veneris Linn on an animal model of testosterone-induced hair loss. Iran J Pharm Res 13:113–118
Nyarko HD, Barku VYA, Batama J (2012) Antimicrobial examinations of Cymbopogon citratus and Adiatum capillus-veneris used in Ghanaian folkloric medicine. Int J Life Sci Pharma Res 2:115–121
Olea N, Pulgar IR, Pdrez P, Olea-serrano F, Rivas A, Soto AM, Sonnenschein C (1996) Estrogenicity of resin-based composites and sealants used in dentistry. Environ Health Perspect 104:298–305
Oliva A, Giami A, Multigner L (2002) A study in a consulting population. J Androl 23:546–50
Peretz J, Vrooman L, Ricke W a, Hunt P a, Ehrlich S, Hauser R, Padmanabhan V, Taylor HS, Swan SH, Vandevoort C a, Flaws J a (2014) Bisphenol A and reproductive health: update of experimental and human evidence, 2007–2013. Environ Health Perspect 122:775–786. doi:10.1289/ehp.1307728
Pourmorad F, Hosseinimehr SJ, Shahabimajd N (2006) Antioxidant activity, phenol and flavonoid contents of some selected Iranian medicinal plants. Afr J Biotechnol 5:1142–1145. doi:10.1055/s-2007-987042
PR (2015) Plant and flower: a comprehensive plants and flower database. Adiantum capillus-veneris, Plant Rescue. http://www.plantsrescue.com/adiantum-capillus-veneris/. Assessed 01 June, 2015
Rahman MS, Kwon W-S, Lee J-S, Yoon S-J, Ryu B-Y, Pang M-G (2015) Bisphenol-A affects male fertility via fertility-related proteins in spermatozoa. Sci Rep 5:9169. doi:10.1038/srep09169
Richter CA, Birnbaum LS, Farabollini F, Newbold RR, Rubin BS, Talsness CE, Vandenbergh JG, Walser-Kuntz DR, vom Saal FS (2007) In vivo effects of bisphenol A in laboratory rodent studies. Reprod Toxicol 24:199–224
Sangai NP, Verma RJ (2011) Quercetin alleviates bisphenol A-induced changes in nucleic acid and protein contents in mice. Acta Pol Pharm Drug Res 68:867–873
Shapiro GD, Dodds L, Arbuckle TE, Ashley-Martin J, Fraser W, Fisher M, Taback S, Keely E, Bouchard MF, Monnier P, Dallaire R, Morisset A, Ettinger AS (2015) Exposure to phthalates, bisphenol A and metals in pregnancy and the association with impaired glucose tolerance and gestational diabetes mellitus: the MIREC study. Environ Int 83:63–71. doi:10.1016/j.envint.2015.05.016
Singleton VL,Orthofer R, Lamuela-Raventós RM (1999) [14] Analysis of total phenols and other oxidation substrates and antioxidants by means of folin-ciocalteu reagent. Methods in Enzymology 299:152–178 doi:10.1016/S0076-6879(99)99017-1; http://www.sciencedirect.com/science/article/pii/S0076687999990171
Shinozaki J, Shibuya M, Masuda K, Ebizuka Y (2008) Squalene cyclase and oxidosqualene cyclase from a fern. FEBS Lett 582:310–318. doi:10.1016/j.febslet.2007.12.023
Singh M, Singh N, Khare PB, Rawat AKS (2008) Antimicrobial activity of some important Adiantum species used traditionally in indigenous systems of medicine. J Ethnopharmacol 115:327–329. doi:10.1016/j.jep.2007.09.018
Soto AM, Sonnenschein C (2010) Environmental causes of cancer: endocrine disruptors as carcinogens. Nat Rev Endocrinol 6:363–370. doi:10.1038/nrendo.2010.87
Staples CA, Dom PB, Klecka GM, Sandra TO, Harris LR (1998) A review of the environmental fate, effects, and exposures of bisphenol A. Chemosphere 36:2149–2173. doi:10.1016/S0045-6535(97)10133-3
Takahashi O, Oishi S (2003) Testicular toxicity of dietarily or parenterally administered bisphenol A in rats and mice. Food Chem Toxicol 41:1035–1044. doi:10.1016/S0278-6915(03)00031-0
Takao T, Nanamiya W, Nagano I, Asaba K, Kawabata K, Hashimoto K (1999) Exposure with the environmental estrogen bisphenol A disrupts the male reproductive tract in young mice. Life Sci 65:2351–2357. doi:10.1016/S0024-3205(99)00502-0
Tan BLL, Kassim NM, Mohd MA (2003) Assessment of pubertal development in juvenile male rats after sub-acute exposure to bisphenol A and nonylphenol. Toxicol Lett 143:261–270. doi:10.1016/S0378-4274(03)00172-3
TDS (2010) Technical Data Sheet 866, chemistry beyond the ordinary. Polysciences, Inc. http://www.polysciences.com/skin/frontend/default/polysciences/pdf/866.pdf
Tiwari D, Vanage G (2013) Mutagenic effect of bisphenol A on adult rat male germ cells and their fertility. Reprod Toxicol 40:60–68. doi:10.1016/j.reprotox.2013.05.013
Toppari J, Larsen JC, Christiansen P, Giwercman A, Grandjean P, Guillette LJ, Jégou B, Jensen TK, Jouannet P, Keiding N, Leffers H, McLachlan JA, Meyer O, Müller J, Rajpert-De Meyts E, Scheike T, Sharpe R, Sumpter J, Skakkebaek NE (1996) Male reproductive health and environmental xenoestrogens. Environ Health Perspect 104(Suppl 4):741–803. doi:10.1289/ehp.96104s4741
Tyl RW, Myers CB, Marr MC, Thomas BF, Keimowitz AR, Brine DR, Veselica MM, Fail PA, Chang TY, Seely JC, Joiner RL, Butala JH, Dimond SS, Cagen SZ, Shiotsuka RN, Stropp GD, Waechter JM (2002) Three-generation reproductive toxicity study of dietary bisphenol A in CD Sprague–Dawley rats. Toxicol Sci 68:121–146. doi:10.1093/toxsci/68.1.121
Vandenberg LN, Hauser R, Marcus M, Olea N, Welshons WV (2007) Human exposure to bisphenol A (BPA). Reprod Toxicol 24:139–177. doi:10.1016/j.reprotox.2007.07.010
Victor B, Maridass M, Ramesh U, Prabhu JMA (2003) Antibacterial activity of essential oils from the leaves of Adiantum capillus-veneris Linn. Malaysian J Sci 22:65–66, http://e-journal.um.edu.my/public/article-view.php?id=1917
Vijayalakshmi A, Kiran Kumar Y (2013) Evaluation of goitrogenic and antithyroidal effect of the fern Adiantum capillus-veneris. Rev Bras Farmacogn 23:802–810. doi:10.1590/S0102-695X2013000500013
Yuan Q, Zhang X, Liu Z, Song S, Xue P, Wang J, Ruan J (2013) Ethanol extract of Adiantum capillus-veneris L. suppresses the production of inflammatory mediators by inhibiting NF-κB activation. J Ethnopharmacol 147:603–611. doi:10.1016/j.jep.2013.03.046
Zade VS, Dabhadkar DK, Thakare VG, Pare SR (2013) Effect of aqueous extract of Moringa oleifera seed on sexual activity of male albino rats. Biol Forum An Int J 5:129–140
Acknowledgments
The authors greatly acknowledged the Higher Education Commission of Pakistan (HEC) for financial support for this study. The World Academy of Science and Chinese Academy of Science are also greatly acknowledged for the CAS-TWAS President fellowship (CAS-TWAS 2014-179). Special thanks are given to the laboratory assistants for their help during experiment and analyses. We would also like to thank Nellie S. and Elizabeth H. for their thoughtful comments and valuable suggestions to improve the language of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Philippe Garrigues
Rights and permissions
About this article
Cite this article
Yousaf, B., Amina, Liu, G. et al. Bisphenol A exposure and healing effects of Adiantum capillus-veneris L. plant extract (APE) in bisphenol A-induced reproductive toxicity in albino rats. Environ Sci Pollut Res 23, 11645–11657 (2016). https://doi.org/10.1007/s11356-016-6330-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-016-6330-0