Abstract
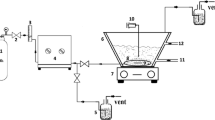
For this work, a phenol solution model was treated by an advanced oxidation process (AOPs), using the heterogeneous catalyst TiO2/BiPO4 and hydrogen peroxide combined with UVA for 240 min. An annular reactor containing a UVA lamp (80 W) was employed. A central composite rotacional design was developed employing a TiO2/BiPO4 concentration of 87 mg L−1 and a hydrogen peroxide concentration of 1800 mg L−1, being evaluated by the degradation percentage and phenol mineralization percentage as responses; 94.30 and 67.00 % were obtained for the phenol degradation and total organic carbon (TOC) conversion, respectively. The lumped kinetic model (LKM) was applied and a satisfactory profile of the residual fractions of the organic compounds present in the liquid phase as a time function with a determination coefficient (R 2 = 0.9945). The toxicity tests employing microbiological species indicated that the organisms tested for the evaluation of the toxic compounds present in the contaminated samples presented a practical low cost test, rapid execution, and high sensibility as an indicator of the presence of toxic substances in liquid effluents.











Similar content being viewed by others
References
Avisar D, Horovitz I, Lozzi L, Ruggieri Baker M, Abel M, Mamane H (2013) Impact of water quality on removal of carbamazepine in natural waters by N-doped TiO2 photo-catalytic thin film surfaces. J Hazard Mater 244–245:463–471
Ayodele OB, Lim JK, Hameed BH (2012) Degradation of phenol in photo-Fenton process by phosphoric acid modified kaolin supported ferric-oxalate catalyst: optimization and kinetic modeling. Chem Eng J 197:181–192
Azevedo EB, Tôrres AR, Aquino NFR, Dezotti M (2009) TiO2-Photocatalyzed degradation of phenol in saline media in an annular reactor: hydrodynamics, lumped kinetics, intermediates, and acute toxicity. Braz J Chem Eng 26:75–87
Bothwell JM, Scott W, Krabbez W, Mohan RS (2011) Applications of bismuth(III) compounds in organic synthesis. Chem Soc Rev 40:4649–4707
Cao J, Xu B, Lin H, Chen S (2013) Highly improved visible light photocatalytic activity of BiPO4 through fabricating a novel p–n heterojunction BiOI/BiPO4 nanocomposite. Chem Eng J 228:482–488
Carpio E, Zuniga P, Ponce S, Solis J, Rodriguez J, Estrada W (2005) Photocatalytic degradation of phenol using TiO2 nanocrystals supported on activated carbon. J Mol Catal A Chem 228:293–298
Chiou C, Juang R (2007) Photocatalytic degradation of phenol in aqueous solutions by Pr-doped TiO2 nanoparticles. J Hazard Mater 149:1–7
Karunakaran C, Dhanalakshmi R, Gomathisankar P (2012) Phenol-photodegradation on ZrO2. Enhancement by semiconductors. Spectrochim Acta A Mol Biomol Spectrosc 92:201–206
Keav S, Monteros AE, Barbier J Jr, Duprez D (2014) Wet air oxidation of phenol over Pt and Ru catalysts supported on cerium-based oxides: resistance to fouling and kinetic modelling. Appl Catal B Environ 150–151:402–410
Laoufi NA, Tassalit D, Bentahar F (2008) The degradation of phenol in water solution by TiO2 photocatalyst in a chemical reactor. Glob NEST J 404–418
Lin H, Ye H, Xu B, Cao J, Chen S (2013) Ag3PO4 quantum dot sensitized BiPO4: a novel p–n junction Ag3PO4/BiPO4 with enhanced visible-light photocatalytic activity. Catal Commun 37:55–59
Liu Y, Zhu Y, Bai X, Zong R, Zhu Y (2013) Degradation and mineralization mechanism of phenol by BiPO4 photocatalysis assisted with H2O2. Appl Catal B Environ 142–143:562–567
Mcmanamon C, Holmes JD, Morris MA (2011) Improved photocatalytic degradation rates of phenol achieved using novel porous ZrO2-doped TiO2 nanoparticulate powders. J Hazard Mater 193:120–127
Pan L, Liu X, Sun Z, Sun C (2013) Nanophotocatalysis via microwave-assisted solution-phase synthesis for efficient photocatalysis. J Mater Chem 1:8299–8326
Pintar A, Bercic G, Besson M, Gallezot P (2014) Catalytic wet-air oxidation of industrial effluents: total mineralization of organics and lumped kinetic modelling. Appl Catal B Environ 47:143–152
Prabha I, Lathasree S (2014) Photodegradation of phenol by zinc oxide, titânia and zinc oxide–titania composites: nanoparticle synthesis, characterization and comparative photocatalytic efficiencies. Mater Sci Semicond Process 26:603–613
Rego E, Marto J, São Marcos P, Labrincha JA (2009) Decolouration of Orange II solutions by TiO2 and ZnO active layers screen-printed on ceramic tiles under sunlight irradiation. Appl Catal A: Gen 355:109–114
Rodríguez-Díaz JM, García JOP, Sánchez LRB, da Silva MGC, da Silva VL, Arteaga-Pérez LE (2015) Comprehensive characterization of sugarcane bagasse ash for its use as an adsorbent. Bioenerg Res 8(4):1885–1895
Royaee SJ, SohrabI M, Barjesteh PJ (2012) Performance evaluation of a continuous flow photo-impinging streams cyclone reactor for phenol degradation. Chem Eng Res Des 90:1923–1929
Sanchez-Dominguez M, Morales-Mendoza G, Rodriguez-Vargas MJ, Ibarra-Malo CC, Rodriguez-Rodriguez AA, Vela-Gonzalez AV, Perez-Garcia SA, Gomez R (2015) Synthesis of Zn-doped TiO2 nanoparticles by the novel oil-in-water (O/W) microemulsion method and their use for the photocatalytic degradation of phenol. J Environ Chem Eng
Satyapaul AS, Madras G (2013) Photocatalytic degradation with combustion synthesized WO3 and WO3/TiO2 mixed oxides under UV and visible light. Sep Purif Technol 105:79–89
Seabra MP, Rego E, Ribeiro A, Labrincha JA (2011) Photodegradation of Orange II solutions by TiO2 active layers jet sprayed on aluminium sheets. Chem Eng J 171:175–180
Shee D, Deo G, Hirt AM (2010) Characterization and reactivity of sol–gel synthesized TiO2–Al2O3 supported vanadium oxide catalysts. J Catal 273:221–228
Silva SW, Klauck CR, Siqueira MA, Bernardes AM (2015) Degradation of the commercial surfactant nonylphenolethoxylate by advanced oxidation processes. J Hazard Mater 282:241–248
Song C, Wang L, Ren J, Lv B, Sun Z, Yan J, Li X, Liu J (2015) Comparative study of diethyl phthalate degradation by UV/H2O2 and UV/TiO2: kinetics, mechanism, and effects of operational parameters. Environ Sci Pollut Res. doi:10.1007/s11356-015-5481-8
Suzuki H, Araki S, Yamamoto H (2015) Evaluation of advanced oxidation processes (AOPs) using O3, UV, and TiO2 for the degradation of phenol in water. J Water Proc Eng 7:54–60
Todan L, Dascalescu T, Preda S, Andronescu C, Munteanu C, Culita AD, Rusu A, State R, Zaharescu M (2014) Porous nanosized oxide poders in the MgO-TiO2 binary system obtained by sol–gel method. Ceram Int 40:15693–15701
Vallejo M, Fresnedo SRM, Ortiz I, Irabien A (2015) Overview of the PCDD/Fs degradation potential and formation risk in the application of advanced oxidation processes (AOPs) to wastewater treatment. Chemosphere 118:44–56
Vermelho AB, Ferreira PA, Rodrigues CRR, Sauto-Padrón T (2006) Microbiologia. Guanabara: Rio de Janeiro
Xu J, Li L, Guo C, Zhang Y, Meng W (2013) Photocatalytic degradation of carbamazepine by tailored BiPO4: Efficiency, intermediates and pathway. Appl Catal B Environ 130–131:285–292
Zhang Y, Xin Q, Cong Y, Wang Q, Jiang B (2013) Application of TiO2 nanotubes with pulsed plasma for phenol degradation. Chem Eng J 215–216:261–268
Zhang Y, Selvaraj R, Sillanpää M, Kim Y, Tai C-W (2014) The influence of operating parameters on heterogeneous photocatalytic mineralization of phenol over BiPO4. Chem Eng J 245:117–23
Acknowledgments
The authors would like to express their upmost gratitude to PRH-28, CNPq/APQ, CNPq/INCTAA, FACEPE/NUQAAPE, CAPES/FCT/MES for financial support.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Santiago V. Luis
Rights and permissions
About this article
Cite this article
Zaidan, L.E.M.C., de Lima Sales, R.V., de Almeida Salgado, J.B. et al. Photodegradation applied to the treatment of phenol and derived substances catalyzed by TiO2/BiPO4 and biological toxicity analysis. Environ Sci Pollut Res 24, 6002–6012 (2017). https://doi.org/10.1007/s11356-015-5952-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-015-5952-y