Abstract
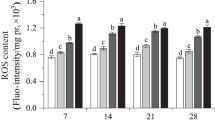
Di-n-butyl phthalates (DBP) are recognized as ubiquitous contaminants in soil and adversely impact the health of organisms. Changes in the activity of antioxidant enzymes and levels of glutathione-S-transferase (GST), glutathione (GSH), and malondialdehyde (MDA) were used as biomarkers to evaluate the impact of DBP on earthworms (Eisenia fetida) after exposure to DBP for 28 days. DBP was added to artificial soil in the amounts of 0, 5, 10, 50, and 100 mg kg−1 of soil. Earthworm tissues exposed to each treatment were collected on the 7th, 14th, 21st, and 28th day of the treatment. We found that superoxide dismutase (SOD) and catalase (CAT) levels were significantly inhibited in the 100 mg kg−1 treatment group on day 28. After 21 days of treatment, GST activity in 10–50 mg kg−1 treatment groups was markedly stimulated compared to the control group. MDA content in treatment groups was higher than in the control group throughout the exposure time, suggesting that DBP may lead to lipid peroxidation (LPO) in cells. GSH content increased in the treatment group that received 50 mg kg−1 DBP from 7 days of exposure to 28 days. These results suggest that DBP induces serious oxidative damage on earthworms and induce the formation of reactive oxygen species (ROS) in earthworms. However, DBP concentration in current agricultural soil in China will not constitute any threat to the earthworm or other animals in the soil.






Similar content being viewed by others

References
Aly MA, Schröder P (2008) Effect of herbicides on glutathione S-transferases in the earthworm, Eisenia fetida. Environ Sci Pollut Res Int 15(2):143–149
Booth LH, Heppelthwaite V, Mcglinchy A (2000) The effect of environmental parameters on growth, cholinesterase activity and glutathione S-transferase activity in the earthworm Apporectodea caliginosa. Biomarkers 5:46–55
Booth LH, Hodge S, O’Halloran K (2001) Use of biomarkers in earthworms to detect use and abuse of field applications of a model organophosphate pesticide. Bull Environ Contam Toxicol 67:633–640
Borch J, Axelstad M, Vinggaard AM, Dalgaard M (2006) Diisobutyl phthalate has comparable anti-androgenic effects to di-n-butyl phthalate in fetal rat testis. Toxicol Lett 163(3):183–190
Burgeot T, Bocqufn G, Porte C (1996) Bioindicators of pollutant exposure in the northwestern Mediterranean east Sea. Mar Ecol Prog Ser 131:125–141
Charles AS, Dennis RP, Thomas FP (1997) The environmental fate of phthalate esters: a literature review. Chemosphere 35(4):667–749
Chen XX, Zhang W, Wu Y, Yang X (2010) Oxidative damage induced by dibutyl phthalate to stem and leaves of arabidopsis seedlings. Asian J Ecotoxicol 5(3):402–406 (in Chinese)
Chen C, Zhou Q, Liu S, Xiu Z (2011) Acute toxicity, biochemical and gene expression responses of the earthworm Eisenia fetida exposed to polycyclic musks. Chemosphere 83:1147–1154
Coleman DC, Ingham ER (1988) Carbon, nitrogen, phosphorus and sulfur cycling in terrestrial ecosystems. Biogeochemistry 5:3–6
Cossu C, Doyotte A, Jacquin MC, Babut M, Exinger A, Vasseur P (1997) Glutathione reductase, selenium-dependent glutathione peroxidase, glutathione levels, and lipid peroxidation in freshwater bivalves, Unio tumidus, as biomarkers of aquatic contamination in field studies. Ecotoxicol Environ Saf 38:122–131
Dallinger R (1993) Strategies of metal detoxification in terrestrial invertebrates. In: Dallinger R, Rainbow PS (eds) Ecotoxicology of metals in invertebrates. Lewis Publishers, London, pp 246–281
Di Giulio RT, Habig C, Gallagher EP (1993) Effects of Black Rock Harbor sediments on indices of biotransformation, oxidative stress, and DNA integrity in channel catfish. Aquat Toxicol 26:15–22
Farber JL (1994) Mechanisms of cell injury by activated oxygen species. Environ Health Perspect 102:17–24
Farombi EO, Abarikwu SO, Adedara IA, Oyeyemi MO (2006) Curcumin and kolaviron ameliorate di-n-butyl phthalate-induced testicular damage in rats. Basic Clin Pharmacol Toxicol 100:43–48
Geret F, Serafim A, Bebianno MJ (2003) Antioxidant enzyme activities, metallothioneins and lipid peroxidation as biomarker in Ruditapes decussatus. Ecotoxicology 12:417–426
Flecha BSG, Repetto M, Evelson P, Boveris A (1991) Inhibition of microsomal lipid-peroxidation by alpha-tocopherol and alpha-tocopherol acetate. Xenobiotica 21:1013–1022
Gao YH, Sun ZJ, Sun XS, Bao YZ (2007a) Toxic effect of olaquindox antibiotic on Eisenia fetida. Eur J Soil Biol 43(s):252–255
Gao YH, Sun ZJ, Liu YQ, Sun XS, Li YR, Bao YZ, Wang GC (2007b) Effect of albendazole anthelmintics on the enzyme activities of different tissue regions in Eisenia fetida. Eur J Soil Biol 43(s):246–251
Greanwald RA (1985) Handbook of methods for oxygen radical research. CRC Press, Boca Raton
Grundy JE, Storey KB (1998) Antioxidant defenses and lipid peroxidation damage in estivating toads, Scaphiopus couchii. J Comp Physiol 168:132–142
Gudbrandsen M, Sverdrup LE, Aamodt S, Stenersen J (2007) Short-term preexposure increases earthworm tolerance to mercury. Eur J Soil Biol 43(s):261–267
Habig WH, Pabst MJ, Jakoby WB (1974) Glutathione S-transferases. The first enzymatic step in mercapturic acid formation. J Biol Chem 249:7130–7139
Hallmark N, Walker M, McKinnell C, Mahood IK, Scott H, Bayne R, Coutts S, Anderson RA, Greig I, Morris K, Sharpe RM (2007) Effects of monobutyl and di(n-butyl) phthalate in vitro on steroidogenesis and Leydig cell aggregation in fetal testis explants from the rat: comparison with effects in vivo in the fetal rat and neonatal marmoset and in vitro in the human. Environ Health Perspect 115:390–396
Hartley-Whitaker J, Ainsworth G, Meharg A (2001) Copper and arsenate-induced oxidative stress in Holcus lanatus L. clones with differential sensitivity. Plant Cell Environ 24:713–722
Higuchi TT, Palmer JS, Gray LE, Veeramachaneni DNR (2003) Effects of dibutyl phthalate in male rabbits following in Utero, Adolescent, or postpubertal exposure. Toxicol Sci 72:301–313
Houghton PJ, Zarka R, Heras B, Hoult JRS (1995) Fixed oil of Nigella sativa and derived thymoquinone inhibit eicosanoid generation in leukocytes and membrane lipid peroxidation. Planta Med 61(1):33–36
Hu XY, Wen B, Shan XQ (2003) Survey of phthalate pollution in arable soils in China. J Environ Monit 5:649–653
Hu CW, Lia M, Cui YB, Li DS, Chen J, Yang LY (2010) Toxicological effects of TiO2 and ZnO nanoparticles in soil on earthworm Eisenia fetida. Soil Biol Biochem 42:586–591
Huff JE, Kluwe WM (1984) Phthalate esters carcinogenicity in F344/N rates and B6C3 F mice. Prog Clin Biol Res 141:137–154
International Programme on Chemical Safety (IPCS) (1992) Environmental health criteria 131. Diethylhexyl phthalate. World Health Organization, Geneva
Jafari M (2007) Dose- and time-dependent effects of sulfur mustard on antioxidant system in liver and brain of rat. Toxicology 231:30–39
Jianlong W, Ping L, Yi Q (1996) Biodegradation of phthalic acid esters by acclimated activated sludge. Environ Int 22:737–741
Jobling S, Reynolds T, White R (1995) A variety of environmentally persistent chemicals, including some phthalate plasticizers, are weakly estrogenic. Environ Health Perspect 103:582–587
Kleymenova E, Swanson C, Boekelheide K, Gaido KW (2005) Exposure in utero to di(n-butyl) phthalate alters the vimentin cytoskeleton of fetal rat sertoli cells and disrupts sertoli cell-gonocyte contact. Biol Reprod 73:482–490
Kochba J, Lavee S, Spiegel-Roy P (1977) Differences in peroxidase activity and isoenzymes in embryogenic and non-embryogenic ‘Shamouti’ orange ovular callus lines. Plant Cell Physiol 18:463–467
Kono Y, Fridovich I (1982) Superoxide radical inhibits catalase. J Biol Chem 257:5751–5754
Łaszczyca P, Augustyniak M, Babczyńska A, Bednarska K, Kafel A, Migula P, Wilczek G, Witas I (2004) Profiles of enzymatic activity in earthworms from zinc, lead and cadmium polluted areas near Olkusz (Poland). Environ Int 30:901–910
Lawson PB, Yu MH (2003) Fluoride inhibition of superoxide dismutase (SOD) from the earthworm Eisenia fetida. Fluoride 36:143–151
Lee SK, Veeramachaneni DNR (2005) Subchronic exposure to low concentrations of di-n-butyl phthalate disrupts spermatogenesis in Xenopus laevis frogs. Toxicol Sci 84:394–407
Lee KY, Shibutani M, Takagi H (2004) Diverse developmental toxicity of di-n-butyl phthalate in both sexes of rat offspring after maternal exposure during the period from late gestation through lactation. Toxicology 203:221–238
Leiers B, Kampkötter A, Grevelding CG, Link CD, Johnson TE, Henkle-Dűhrsen K (2003) A stress-responsive glutathione S-transferase confers resistance to oxidative stress in Caenorhabditis elegans. Free Radic Biol Med 34(11):1405–1415
Li MH (2003) Peroxidase and superoxide dismutase activities in fig leaves in response to ambient air pollution in a subtropical city. Arch Environ Contam Toxicol 45:168–176
Li XY, Luo YR, Yun MX, Wang J, Wang JJ (2010) Effects of 1-methyl-3-octylimidazolium bromide on the anti-oxidant system of earthworm. Chemosphere 78:853–858
Lin AJ, Zhang XH, Chen MM, Cao Q (2007) Oxidative stress and DNA damages induced by cadmium accumulation. Chin J Environ Sci 19:596–602
Lqaszczyca P, Augustyniak M, Babczyńska A, Bednarska K, Kafel A, Migula P, Wilczek G, Witas I (2004) Profiles of enzymatic activity in earthworms from zinc, lead and cadmium polluted areas near Olkusz (Poland). Environ Int 30(7):901–910
Lukkari T, Taavitsainen M, Soimasuo M, Oikari A, Haimi J (2004) Biomarker responses of the earthworm Aporrectodea tuberculata to copper and zinc exposure, differences between populations with and without earlier metal exposure. Environ Pollut 129:377–386
Ma LL, Ma C, Shi ZM, Li WM, Xu L, Hu F, Li HX (2012) Effects of fluoranthene on the growth, bioavailability and anti-oxidant system of Eisenia fetida during the ageing process. Eur J Soil Biol 50:21–27
Maity S, Roy S, Chaudhury S, Bhattacharya S (2008) Antioxidant responses of the earthworm Lampito mauritii exposed to Pb and Zn contaminated soil. Environ Pollut 151(1):1–7
Marklund S, Marklund G (1974) Involvement of the superoxide anion radical in the autoxidation of pyrogallol and a convenient assay for superoxide dismutase. Eur J Biochem 47:169–479
Mather-Mihaich F, Di Giulio RT (1993) Antioxidant enzyme activities and malondialdehyde, glutathione and methemoglobin concentrations in channel catfish exposed to DEF and n-butyl mercaptan. Comp Biochem Physiol C 85:427–432
Nandi PK, Agrawal M, Rao DN (1984) SO2 induced enzymatic changes and ascorbic acid oxidation in Oryza sativa L. Water Air Soil Pollut 21:25–32
OECD (1984) Test 207: Earthworm, acute toxicity tests. In: Organization for economic cooperation and development. OECD guidelines for testing of chemicals. Organization for Economic Cooperation and Development (OECD). Paris, France
Ohkawa H, Ohishi N, Yagi K (1979) Assay for lipid peroxides in animal-tissues by thiobarbituric acid reaction. Anal Biochem 95:351–358
Papadimitriou E, Loumbourdis NS (2002) Exposure of the frog Rana nidibunda to copper impact on two biomarkers, lipid peroxidation, and glutathione. Bull Environ Contam Toxicol 69:885–889
Perally S, LaCourse EJ, Campbell AM, Brophy PM (2008) Heme transport and detoxification in nematodes: subproteomics evidence of differential role of glutathione transferases. J Proteome Res 7(10):4557–4565
Qin JF, Chen HG, Cai WG, Yang T, Jia XP (2011a) Effects of di-n-butyl phthalate on the antioxidant enzyme activities and lipid peroxidation level of Perna viridis. Chin J Appl Ecol 22(7):1878–1884 (in Chinese)
Qin JF, Chen HG, Cai WG, Yang T, Jia XP (2011b) Effects of di-n-butyl phthalate (DBP) on activity of enzyms in different tissues of crimson snapper (Lutjanus erythropterus). J Fish Sci Chin 18(5):1125–1131 (in Chinese)
Radi R, Beckman JS, Bush KM, Freeman BA (1991) Peroxynitrite-induced membrane lipid peroxidation: the cytotoxic potential of superoxide and nitric oxide. Arch Biochem Biophys 288:481–487
Rao JV (2006) Sublethal effects of an organophosphorus insecticide (RPR-II) on biochemical parameters of tilapia, Oreochromis mossambicus. Comp Biochem Physiol C 143:492–498
Saint-Denis M, Narbonne JF, Arnaud C, Thybaud E, Ribera D (1999) Biochemical responses of the earthworm Eisenia fetida andrei exposed to contaminated artificial soil: effects of benzo(a)pyrene. Soil Biol Biochem 31:1837–1846
Saint-Denis M, Narbonne JF, Arnaud C, Ribera D (2001) Biochemical responses of the earthworm Eisenia fetida andrei exposed to contaminated artificial soil, effects of lead acetate. Soil Biol Biochem 33(3):395–404
Sandalio LM, Dalurzo HC, Gómez M, Romero-Puertas MC, del Rio LA (2001) Cadmium induces changes in the growth and oxidative metabolism of pea plants. J Exp Bot 52:2115–2126
Sen CK, Marin E, Kretzschmar M, Hanninen O (1992) Skeletal muscle and liver glutathione homeostasis in response to training, exercise, and immobilization. J Appl Physiol 73:1265–1272
Singh S, Eapen S, D’Souza SF (2006) Cadmium accumulation and its influence on lipid peroxidation and antioxidative system in an aquatic plant, Bacopa monnieri L. Chemosphere 62:233–246
Solé M, Porte C, Albaigés J (1995) The use of biomarkers for assessing the effects of organic pollution in mussels. Sci Total Environ 159:147–153
Song Y, Zhu LS, Wang J, Wang JH, Liu W, Xie H (2009) DNA damage and effects on antioxidative enzymes in earthworm (Eisenia fetida) induced by atrazine. Soil Biol Biochem 41:905–909
Stenersen J, Oien N (1981) Glutathione S-transferase in earthworms (Lumbricidae). Substrate specificity, tissue and species distribution and molecular weight. Comp Biochem Physiol C 69:243–252
Stenersen J, Guthenberg C, Mannervik B (1979) Glutathione-S-transferases in earthworms (Lumbricidae). Biochem J 181:47–50
Tyl RW, Myers CB, Marr MC (2004) Reproductive toxicity evaluation of dietary butyl benzyl phthalate (BBP) in rats. Reprod Toxicol 18:241–264
US EPA (1991) Risk Assessment Guidance for Superfund (RAGS): Volume I-Human Health Evaluation Manual (HHEM) (Part B, Development of Risk-Based Preliminary Remediation Goals). Office of Emergency and Remedial Response, Washington DC, EPA /540/R-92/003, OSWER Directive 9285.7-01B, NTIS PB92-963333
Wang JL, Liu P, Qian Y (1995) Microbial degradation of di-n-butyl phthalate. Chemosphere 31:4051–4056
Wang Y, Ma ZM, Wu SJ (2014) Study on the effect of enzymatic activity and acute toxicity of three PAEs on Eisenia foetida. Environ Sci 35(2):770–779
Willie JGM, Peijnenburg JS (2006) Occurrence of phthalate esters in the environment of the Netherlands. Ecotoxicol Environ Saf 63:204–215
Wu SJ, Wu EM, Qiu LQ, Zhong WH, Chen JM (2011) Effects of phenanthrene on the mortality, growth, and anti-oxidant system of earthworms (Eisenia fetida) under laboratory conditions. Chemosphere 83:429–434
Wu SJ, Zhang HX, Zhao SL, Wang JL, Li HL, Chen JM (2012) Biomarker responses of earthworms (Eisenia fetida) exposured to phenanthrene and pyrene both singly and combined in microcosms. Chemosphere 87:285–293
Xiao N, Jing B, Ge F, Liu X (2006) The fate of herbicide acetochlor and its toxicity to Eisenia fetida under laboratory conditions. Chemosphere 62:1366–1373
Xu G, Li FH, Wang QH (2008) Occurrence and degradation characteristics of dibutyl phthalate (DBP) and di-(2-ethylhexyl) phthalate (DEHP) in typical agricultural soils of China. Sci Total Environ 393(2–3):333–340
Yu M, Li SM, Li XY, Zhang BJ, Wang JJ (2008) Acute effects of 1-octyl-3-methylimidazolium bromide ionic liquid on the antioxidant enzyme system of mouse liver. Ecotoxicol Environ Saf 71:903–908
Zeng Q, Cai FY, Wang J, Li L, Yang X (2010) A study on the oxidative damage of Esisenia foelide cells induced by DEHP. In: Proceedings of the 2010 4th International Conference on Bioinformatics and Biomedical Engineering (iCBBE). Chengdu, China: IEEE, 1–4
Zheng Z, He PJ, Fu Q, Shao LM, Lee DJ (2008) Partition of six phthalic acid esters in soluble and solid residual fractions of wastewater sludges. Environ Technol 29:343–350
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Philippe Garrigues
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(XLS 51 kb)
Rights and permissions
About this article
Cite this article
Du, L., Li, G., Liu, M. et al. Biomarker responses in earthworms (Eisenia fetida) to soils contaminated with di-n-butyl phthalates. Environ Sci Pollut Res 22, 4660–4669 (2015). https://doi.org/10.1007/s11356-014-3716-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-014-3716-8