Abstract
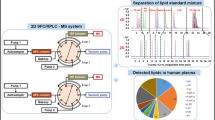
The aim of this study was to develop a method based on ultra high performance liquid chromatography coupled with mass spectrometry (UHPLC-MS) for lipid profiling in human placental choriocarcinoma (JEG-3) cells. Lipids were solid–liquid extracted from JEG-3 cells using a solution of chloroform/methanol (2:1, v/v) in a simple procedure requiring minimal sample alteration. Simultaneous separation of complex lipid mixtures in their major classes was achieved with a reversed-phase (C8) UHPLC column and a mobile phase containing methanol with 1 mM ammonium formate and 0.2 % formic acid (A)/water with 2 mM ammonium formate and 0.2 % formic acid (B). Lipids were characterized using time-of-flight (TOF) and Orbitrap under full scan and positive electrospray ionization mode with both analyzers. A total of 178 species of lipids, including 37 phosphatidylcholines (PC), 32 plasmalogen PC, 9 lyso PC, 4 lyso plasmalogen PC, 30 triacylglycerols, 22 diacylglycerols, 7 cholesterol esters, 25 phosphatidylethanolamines, and 12 sphingomyelins, were identified using TOF and Orbitrap. The identification of all lipid classes was based on exact mass characterization with an error < 5 ppm. The developed methodology was applied to study lipid alterations in human placental cells against the exposure to perfluorinated chemicals (PFCs) and tributyltin (TBT).





Similar content being viewed by others
References
Bennett RD, Heftmann E (1962) Thin-layer chromatography of sterols. J Chromatogr A 9:359–362
Bligh EG, Dyer WJ (1959) A rapid method of total lipid extraction and purification. Can J Biochem Phys 37:911–917
Castro-Perez JM, Kamphorst J, Degroot J, Lafeber F, Goshawk J, Yu K, Shockcor JP, Vreeken RJ, Hankemeier T (2010) Comprehensive LC-MSE lipidomic analysis using a shotgun approach and its application to biomarker detection and identification in osteoarthritis patients. J Proteome Res 9:2377–2389
Christie WW (1985) Rapid separation and quantification of lipid classes by high performance liquid chromatography and mass (light-scattering) detection. J Lipid Res 26:507–512
Fahy E, Subramaniam S, Murphy RC, Nishijima M, Raetz CRH, Shimizu T, Spener F, Van Meer G, Wakelam MJO, Dennis EA (2009) Update of the LIPID MAPS comprehensive classification system for lipids. J Lipid Res 50:S9–S14
Folch J, Lees M, Sloane Stanley GH (1957) A simple method for the isolation and purification of total lipids from animal tissues. J Biol Chem 226:497–509
Garanto A, Mandal NA, Egido-Gabás M, Marfany G, Fabriàs G, Anderson RE, Casas J, Gonzàlez-Duarte R (2013) Specific sphingolipid content decrease in Cerkl knockdown mouse retinas. Exp Eye Res 110:96–106
Gilliland FD, Mandel JS (1996) Serum perfluorooctanoic acid and hepatic enzymes, lipoproteins, and cholesterol: a study of occupationally exposed men. Am J Ind Med 29:560–568
Gorrochategui E, Pérez-Albaladejo E, Casas J, Lacorte S, Porte C (2014) Perfluorinated chemicals: differential toxicity, inhibition of aromatase activity and alteration of cellular lipids in human placental cells. Toxicol Appl Pharmacol 277(2):124–130. doi:10.1016/j.taap.2014.03.012
Grün F, Blumberg B (2006) Environmental obesogens: organotins and endocrine disruption via nuclear receptor signaling. Endocrinol 147:S50–S55
Grün F, Watanabe H, Zamanian Z, Maeda L, Arima K, Cubacha R, Gardiner DM, Kanno J, Iguchi T, Blumberg B (2006) Endocrine-disrupting organotin compounds are potent inducers of adipogenesis in vertebrates. Mol Endocrinol 20:2141–2155
Hughes MA, Brash AR (1991) Investigation of the mechanism of biosynthesis of 8-hydroxyeicosatetraenoic acid in mouse skin. BBA- Lipid Lipid. Met 1081:347–354
Janer G, Navarro JC, Porte C (2007) Exposure to TBT increases accumulation of lipids and alters fatty acid homeostasis in the ramshorn snail Marisa cornuarietis. Comp Biochem Phys C 146:368–374
Nelson JW, Hatch EE, Webster TF (2010) Exposure to polyfluoroalkyl chemicals and cholesterol, body weight, and insulin resistance in the general U.S. population. Environ Health Persp 118:197–202
Oresic M, Hänninen VA, Vidal-Puig A (2008) Lipidomics: a new window to biomedical frontiers. Trends Biotechnol 26:647–652
Pulfer M, Murphy RC (2003) Electrospray mass spectrometry of phospholipids. Mass Spectrom Rev 22:332–364
Ruggieri S (1962) Separation of the methyl esters of fatty acids by thin layer chromatography. Nature 193:1282–1283
Shi Y, Burn P (2004) Lipid metabolic enzymes: emerging drug targets for the treatment of obesity. Nat Rev Drug Discov 3:695–710
Shui G, Bendt AK, Pethe K, Dick T, Wenk MR (2007) Sensitive profiling of chemically diverse bioactive lipids. J Lipid Res 48:1976–1984
Sommer U, Herscovitz H, Welty FK, Costello CE (2006) LC-MS-based method for the qualitative and quantitative analysis of complex lipid mixtures. J Lipid Res 47:804–814
Van Meer G (2005) Cellular lipidomics. EMBO J 24:3159–3165
Wang S, Li J, Shi X, Qiao L, Lu X, Xu G (2013) A novel stop-flow two-dimensional liquid chromatography-mass spectrometry method for lipid analysis. J Chromatogr A 1321:65–72
Watson AD (2006) Thematic review series: systems biology approaches to metabolic and cardiovascular disorders. Lipidomics: a global approach to lipid analysis in biological systems. J Lipid Res 47:2101–2111
Acknowledgments
This study was financed by the INNPACTO project (IPT-2011-0709-060000). Dr. R. Chaler, D. Fanjul, and Eva Dalmau are acknowledged for TOF-MS support and Dr. Alberto Adeva for Orbitrap-MS support.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Philippe Garrigues
Electronic supplementary material
Below is the link to the electronic supplementary material.
Table S1
Elemental composition of glycerophospholipid, glycerolipid, sterol lipid, and sphingolipid species found in human placental choriocarcinoma JEG-3 cells, calculated by mass accuracy within error of 5 ppm, with atom constraints and with −0.5 ≤ DBE ≤ 15.0. DBE double-bond equivalent. Elemental composition of PC, Plasmalogen PC, Lyso PC, Lyso plasmalogen PC, PE, and SM refer to the [M + H]+ ions, whereas TAG, DAG, and CE refer to ammonium adducts [M + NH4]+. Lipid species were detected under ESI (+) using an UHPLC system coupled to a TOF analyzer with an Acquity UPLC BEH C8 column (1.7 μm particle size, 100 mm × 2.1 mm), with the exception of lipid species highlighted with symbol (*), which were identified using an UHPLC-Orbitrap system with the same column (DOCX 65 kb)
Rights and permissions
About this article
Cite this article
Gorrochategui, E., Casas, J., Pérez-Albaladejo, E. et al. Characterization of complex lipid mixtures in contaminant exposed JEG-3 cells using liquid chromatography and high-resolution mass spectrometry. Environ Sci Pollut Res 21, 11907–11916 (2014). https://doi.org/10.1007/s11356-014-3172-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-014-3172-5