Abstract
The Buffalo River and its dams are major surface water sources used for fresh produce irrigation, raw water abstraction and recreation in parts of the Eastern Cape Province in South Africa. Over a 12-month period (August 2010 to July 2011), we assessed the bacteriological qualities of water from the river and 3 source water dams along its course. Faecal indicator bacteria (FIB), including total coliform (TC), faecal coliform (FC) and enterococci (ENT) counts, were high and ranged as follows: 1.9 × 102–3.8 × 107, 0–3.0 × 105 and 0–5.3 × 105 cfu/100 ml for TC, FC and ENT, respectively. Significantly (P < 0.05) higher concentrations of FC and ENT were observed at the sampling sites located at the lower reaches of the river compared to the upper reaches, and at Bridle Drift Dam compared to the other two dams. FIB counts mostly exceeded the recommended maximum values suggested by national and international guidelines for safe fresh produce irrigation, domestic applications, full-contact recreation and livestock watering. These results show that the bacteriological qualities of the Buffalo River and dams were poor, and suggest that sewage was dumped into the Buffalo River during the study period. Urban runoffs and effluents of wastewater treatment plants appear to be important sources of faecal contamination in the river. We conclude that these water bodies represent significant public health hazards. Provision of adequate sanitary infrastructure will help prevent source water contamination, and public health education aimed at improving personal, household and community hygiene is imperative.
Similar content being viewed by others
Explore related subjects
Find the latest articles, discoveries, and news in related topics.Avoid common mistakes on your manuscript.
Introduction
Surface waters, including dams, rivers and streams, constitute an important source of water for drinking, domestic, agricultural, recreational and other purposes. However, they are vulnerable to pollution and are frequently contaminated with faecal matter (Effler et al. 2001; Kistemann et al. 2002). Nonpoint sources of such contamination include domestic and wild animal defecation, malfunctioning sewage and septic systems, storm water drainage and urban runoff (Kistemann et al. 2002; Chigor et al. 2012). Point sources include municipal wastewater treatment plants (Shuval 1990; Okoh et al. 2007; Igbinosa and Okoh 2009; Lata et al. 2009: Chigor et al. 2010a; Odjadjare et al. 2010), and drainage from areas where livestock are handled (Williams et al. 2012).
Coliforms and enterococci are indicator organisms used worldwide to monitor water quality (Toranzos and McFeters 1997; APHA 1998; Anderson et al. 2005; Harwood et al. 2005; Gersberg et al. 2006). The detection of these indicators in water signifies faecal pollution, which could have detrimental effects on public health, the economy, and on ecological balance and functioning (Gourmelon et al. 2007; Abdelzaher et al. 2010; MacIntyre and de Villiers 2010). The public health risks associated with faecal pollution include the introduction of microbial pathogens (APHA 1998; Pruss et al. 2002; Okoh et al. 2007; WHO 2008) and antibiotic-resistant strains of bacterial pathogens, which could result in the transfer of resistance to previously susceptible strains or species in aquatic environments (Ash et al. 2002; Zhang et al. 2009; Chigor et al. 2010b).
Contamination of surface water bodies with pathogenic agents (including bacteria, viruses and protozoa) could result in the transmission of waterborne and water-related diseases to people using the water for domestic purposes (Fong and Lipp 2005; World Health Organization 2008), to swimmers (Gersberg et al. 2006; Abdelzaher et al. 2010), and to agricultural workers and the consumers of crops irrigated with polluted waters (Shuval 1990; Mohanty et al. 2002; Gemmell and Schmidt 2012). Many viral, bacterial and parasitic diseases have been associated with waterborne transmission (Hunter 2003). Such infections contribute significantly to the global disease burden (Payment and Riley 2002; Pruss et al. 2002). Among the bacterial pathogens, toxigenic Vibrio cholerae, the aetiological agent of cholera, has caused several pandemics and still represents a serious problem, causing repeated epidemics especially in developing countries (Stewart-Tull 2001; Zahid et al. 2008). Salmonella and Shigella species pose serious public health problems to the developing world (Mills-Robertson et al. 2003; Deering et al. 2012), and the threat from Escherichia coli pathotypes is a rising global challenge (Chigor et al. 2010b; Bielaszewska et al. 2011). Human enteric viruses are the major cause of water-related disease and have been estimated to cause about 30–90 % of gastroenteritis cases worldwide (Fong and Lipp 2005; Bosch et al. 2008) and protozoans such as Cryptosporidium and Giardia have been implicated in outbreaks involving recreational water use and contaminated municipal water (Wilczynski et al. 2012).
In South Africa, although water infrastructures are well developed in urban areas, in rural communities, they are either poorly developed or nonexistent (Obi et al. 2004). Available data (2008) reveals that more than 40 % of the South African populations dwell in rural areas (DWAF 2010). In many rural areas, over 75 % of poor households have no access to treated tap water (DWAF 2004). Consequently, many households (approximately 74 % of all rural households) rely solely on untreated stream or river water (DWAF 2004; Obi et al. 2004; RHP 2004). Only 13.6 % of the Eastern Cape population of about 7.3 million has access to pipe-borne water either in their dwelling place or within 200 m (MDB 2010). Numerous studies have, however, shown that such water sources are susceptible to pollution, are contaminated and constitute serious public health risks in South Africa (Jagals 1997; Morrison et al. 2001; Muller et al. 2001; Obi et al. 2002; Diergaardt et al. 2004; Igbinosa and Okoh 2009; Odjadjare and Okoh 2010).
Buffalo River, located in the Eastern Cape Province, is important as the major water source for urban, rural, industrial and irrigation consumers, as well as for recreational purposes in one of the most populous areas on the East coast of southern Africa. Despite its importance, there appears to be no report on the microbial quality of this river. The 2004 River Health Programme (RHP) report on the Buffalo River was based on such indices as diversity of habitats, geomorphology and riparian vegetation that served as indicators of ecological health (RHP 2004). Although the report says that blockages in the sewerage systems, inadequate treatment capacity and poor management result in the discharge of partially treated and untreated sewage into the river and dams, no bacteriological data was presented. The paucity of reports on the bacteriological quality of Buffalo River and the source water dams located along its course calls for attention. It was therefore important to carry out this study with the primary goal of determining the bacteriological quality of these essential surface waters to assess the public health risks attendant to their uses.
Materials and methods
Study area and samples collection
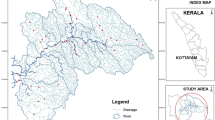
The Buffalo River (Fig. 1) is located in the Eastern Cape Province of South Africa. Rising at an altitude of 1,200 m in the Amathola Mountains of the Eastern Cape, it flows south-eastwards for about 126 km before emptying into the Indian Ocean at East London harbour. The climate is warm and temperate, and temperatures are moderate in the coastal zone (8 to 39 °C) with a warm mean annual value of 21 °C while inland temperatures vary between −2 and 42 °C with a mean annual value of 18 °C (RHP 2004). The mainly summer rainfall in the Buffalo River catchment ranges from 400 to more than 1,000 mm per year with an annual mean value of about 700 mm (RHP 2004). Precipitation measurements were beyond the scope of the present study. Measurements of the river dimensions and flow were also beyond the scope of this study. Recorded average width of the highly meandering river ranged from about 4 m in the upper reaches to between 40 and 50 m at the lower reaches (RHP 2004). South Africa’s only river port, the Port of East London, is located at the mouth of the Buffalo River. Along the river there are four dams that serve as raw water sources for drinking water production by water treatment works (WTW), including King William’s Town WTW, Schornville WTW and East London WTW, supplying water to the urban areas of King William’s Town, Zwelitsha, Mdantsane and East London and the surrounding settlements. Urban built-up and industrial areas cover about 12 % of its 1,287-km2 catchment. Agriculture is widespread in the middle reaches of the catchment, from the foothill zone downstream of Rooikrantz Dam to King William’s Town and as far downstream as Bridle Drift Dam. Goat, cattle and sheep farming prevail. Although subsistence farming predominates, local areas of intensive irrigation provide fresh produce and other crops. The coastal zone is commercially important for tourism, fishing and related activities (RHP 2004). There are at least nine different wastewater treatment plants (WTPs) in the Buffalo River catchment discharging effluents either directly or indirectly (via major tributaries like Ngqokweni and Yellowwoods rivers) into the Buffalo River (Table 1).
The study area and sampling sites, S1–S6 Maden Dam, Rooikrantz Dam, King William’s Town, Eluxolzweni, Bridle Drift Dam and Parkside. With kind permission from Springer Science+Business Media: Food and Environmental Virology, Quantitative Detection and Characterization of Human Adenoviruses in the Buffalo River in the Eastern Cape Province of South Africa, 4, 2012, 200, VN Chigor and AI Okoh, Fig. 1
A total of six different sampling sites, S-1 to S-6 (Fig. 1) were identified using a Global Positioning System (GPS) instrument (eTrex Legend H; Garmin, Olathe, KS, USA), on the river course including three dam sites (S–1, S–2 and S-5) and three non-dam sites (S-3, S-4 and S-6). The geographical coordinates and description of the sites are given in ESM 1. The sites were selected based on a number of factors including geographical location, anthropogenic activity/major water use, rural/urban status and access. Although there are four source water dams along the Buffalo River course, samples were collected from only three dams because the bridge leading to Laing Dam was closed during the study period.
From August 2010 to July 2011, between 8 a.m. and 1 p.m., duplicate water samples were collected once monthly, from spatially discrete points at each sampling site, at a depth of approximately 15 cm below the water surface, using sterile 1.75-l screw-capped bottles. Ample air space was left in the bottles to facilitate mixing by shaking before examination. The samples were immediately placed in a lightproof insulated box containing ice-packs and transported to the Applied and Environmental Microbiology Research Group (AEMREG) Laboratory at the University of Fort Hare, Alice, South Africa, through a journey of about 2 h. Upon arrival, the samples were immediately stored at 4 °C until processing. All the samples were processed within 8 h of collection as recommended by American Public Health Association (APHA 1998).
Enumeration of water quality indicators
Equal volumes (500 ml) of the duplicate samples were mixed and the homogenate analysed. The total coliforms (TC), faecal coliforms (FC) and enterococci (ENT) counts were determined by membrane filtration according to standard methods (APHA 1998). For TC, samples were processed by making tenfold serial dilutions with 100 ml of each composite and filtering 100 ml of water through membrane filters (47-mm diameter, 0.45 μm pore size; Millipore, County Cork, Ireland). Thereafter, the Millipore filter papers were placed on m-Endo agar (Merck, Wadeville, South Africa) and incubated at 37 °C for 24 h. Typical red colonies with a metallic sheen were enumerated and reported as colony forming units (cfu)/100 ml surface water. For FC, composite samples were processed by making serial dilutions as described above and filtering 100 ml of water through membrane filters (47-mm diameter, 0.45 μm pore size). The Millipore filter papers were then placed on m-FC agar (Merck, Wadeville, South Africa) and incubated at 44.5 °C for 24 h. Colonies exhibiting any shades of blue were counted and reported as cfu/100 ml surface water. E. coli (ATCC 29522) was used as a positive control in both the TC and FC tests. For the enumeration of ENT, water samples were diluted and filtered as described above and the Millipore filter paper was placed on Enterococcus Selective Agar (Merck, Wadeville, South Africa). After incubation at 37 °C for 48 h, all brown to black colonies with a typical dark halo were counted as faecal enterococci and reported as cfu/100 ml surface water. Enterococcus faecalis (ATCC 29212) was used as a positive control. Analysis per sample per parameter was done in triplicate.
Statistical analysis
Using SPSS (IBM SPSS Statistics 19), one-way analysis of variance and Tukey’s multiple range tests were used to compare the mean values of the tested parameters for all the different sampling sites, months and seasons. Statistical significance was set at P values < 0.05.
Results and discussion
Faecal indicator bacteria (FIB) concentrations observed in this study were high across all sites. Total coliforms (TC), faecal coliforms (FC) and enterococci concentrations varied widely and ranged from 1.9 × 102–3.8 × 107 cfu/100 ml, 0–3.0 × 105 cfu/100 ml and 0–5.3 × 105 cfu/100 ml, respectively. These concentrations and wide variations are similar to findings elsewhere (Schets et al. 2008; Lata et al. 2009; USEPA 2010; Chigor et al. 2012).
The average concentrations of the FIB were compared per sampling site across the four South Africa seasons including spring (September, October and November), summer (December, January and February), autumn (March, April and May) and winter (June, July and August). No seasonal trend was observed. This is not surprising considering that rainfall and storm events occurred across the seasons during the study period and previous reports have shown that extreme rainfall and runoff result in significant increases in microbial loads of surface waters (Kistemann et al. 2002; Chigor et al. 2012). The continuous faecal contamination of the river appears to emanate also from the WTPs in the catchment (Table 1). There is a prevalence of overloading and recorded microbiological non-compliance amongst the WTPs (RHP 2004; DWAF 2012). Five of the 9 WTPs are currently overloaded with operational capacities ranging from 133.3 to 237 %. Of the 9 WTPs, whose effluents are discharged directly into Buffalo River, only one (West Bank) currently records a satisfactory microbiological compliance (MC). The compliance level of the other nine ranged from 0 to 68 %, with 77.8 % of the plants recording MC values below 50 %. While the MC level at the Postdam WTP was as low as 2 %, the Schornville and Mdantsane East plants showed zero compliance (DWAF 2012). Consequently, untreated or inadequately treated sewage is discharged of into the river and dams.
The contribution of individual point sources to the microbial load of surface water is variable. Inefficient WTPs will discharge final effluents with unacceptable microbial counts into the receiving water bodies (Casadio et al. 2010; Odjadjare et al. 2010). Even in cases where the WTPs are efficient and there is significant reduction of enteric microbes, heavy rainfall events may still result in flooding of these plants and the washing off of raw sewage into surface waters. Reports have continued to associate faecal pollution and waterborne disease with heavy rainfall (Hunter 2003; Drayna et al. 2010). The study period was preceded by drought (Clarke et al. 2012) in which the study area experienced very low rainfall, and during reconnaissance visits and selection of sampling sites in May–June, 2010, water levels at the dams were observed to be very low. The heavy rainfall that returned before the onset of sampling in August 2010 did continue, with varying intensity, throughout the study period. The results presented in this study agree with other reports that storm events can lead to high counts of indicator bacteria in river waters (Kistemann et al. 2002; Hunter 2003; Chigor et al. 2012) and suggest that people swimming in Buffalo River are at an increased risk of illness.
Figure 2 shows the spatial variation in the counts of the FIB in the Buffalo River and compares the 12-month mean values for the three indicators assessed in this study. The significantly higher (P < 0.05) mean concentrations of the indicator bacteria recorded at Bridle Drift Dam (TC, 3.4 × 104 cfu/100 ml; FC, 1.9 × 103 cfu/100 ml; ENT, 2.9 × 102 cfu/100 ml) compared to the two other dams, Maden (TC, 3.9 × 103 cfu/100 ml; FC, 3.8 × 102 cfu/100 ml; ENT, 1.5 × 102 cfu/100 ml) and Rooikrantz Dam (TC, 6.0 × 103 cfu/100 ml; FC, 2.0 × 102 cfu/100 ml; ENT, 7.1 × 101 cfu/100 ml) indicate that this is the most contaminated dam.
Spatial variation in mean concentrations of faecal indicator bacteria at the six sites (S1–S6) located on the Buffalo River. Composite samples were collected monthly at the each site for a total of 12 months (August 2010 to July 2011) and each sample was analysed in triplicate. Reported values are the average counts for the entire 12-month period
A uniform trend was observed for the three bacteriological parameters tested. Significantly higher (P < 0.05) mean concentrations of FIB were recorded at the sampling sites located at the lower reaches (King William’s Town, Eluxolzweni, Bridle Drift Dam and Parkside) of the river compared to those at the upper reaches (Maden Dam and Rooikrantz Dam). For TC, mean concentrations observed at Maden Dam and Roikrantz were 3.9 × 103 cfu/100 ml and 6.0 × 103 cfu/100 ml, respectively, while at the lower reaches mean concentrations ranged from 3.4 × 104 cfu/100 ml recorded at Bridle Drift Dam to 3.2 × 106 cfu/100 ml observed at Parkside. Both the FC and enterococci showed trends similar to that of TC with the lower-reaches sites yielding significantly (P < 0.05) higher mean concentrations ranging from 1.9 × 103–2.7 × 104 cfu/100 ml for FC, and from 2.9 × 102–4.8 × 104 cfu/100 ml for enterococci. This could be attributable to anthropogenic activities and increased populations in the different catchments.
A recent report (Williams et al. 2012) highlighted the need for land-use types associated with particular areas of a watercourse to be considered as a central factor in models that aim to predict pathogen risk in environmental waters. In this study, the least counts of FIB were detected at Maden and Rooikrantz dams. Although there are a few sparsely populated settlements, much of the catchment upstream of this area is a protected state forest, so pressures from human activity are limited to forest management and recreational activities. The significantly higher counts recorded at the lower reaches including at King William’s Town, Eluxolzweni and Bridle Drift Dam could be attributed to catchment conditions and land-use patterns, which our data suggests to have remained unchanged nearly a decade since the RHP studies (RHP 2004). This area is heavily impacted by dense rural and urban populations, and WTPs (Table 1) which are reported to be overloaded and spilling effluents that are either untreated or insufficiently treated into the river (DWAF 2012). Irrigation agriculture along the river catchment, even on steep slopes, is extensive and in situ herd watering is common. Downstream of Bridle Drift Dam, the Buffalo River passes through the Umtiza Nature Reserve where anthropogenic impacts are low and the river’s self-purification process is therefore enhanced. The impact of this, in addition to high salinity of the estuary would have been very low bacterial counts at Parkside. Conversely, higher counts (Fig. 2) were recorded, and this could be attributed to the impact of the Amalinda Central WTP effluents and stormwater runoff from the East London city centre (RHP 2004).
Expectedly, the general trend at all the sites was that TC concentrations were significantly (P < 0.05) higher than FC concentrations. It is also evident from Fig. 2 that across the sites, FC concentrations recorded in this study were always higher than the ENT concentrations, except at Parkside where the mean ENT concentration was higher than that of FC. Previous studies have reported that FC shows greater persistence in freshwater than ENT (Sinton et al. 2002; Anderson et al. 2005). In their report on the persistence and differential survival of faecal indicator bacteria in subtropical waters and sediments, Anderson et al. (2005), who measured persistence by decay rates (change in culturable concentrations over time), showed that faecal coliform decay rates were significantly lower than those of ENT in freshwater. This higher persistence of FC has been attributed, in part, to the sensitivity of ENT to photooxidation (Bernier et al. 2009) that results in ENT surviving less easily, compared to faecal coliforms, in river water. The higher ENT concentrations observed at Parkside could be attributable to the fact that ENT have been shown to survive harsh environments that is associated with river estuaries (He and Jiang 2005) and characterized by extremes of salinity as observed at Parkside (range, 32.47–33.62). Longer persistence of ENT than of FC in saline waters has been documented (Davies et al. 1995).
Figures 3 to 5 show the monthly variation in counts of faecal indicator bacteria observed at the six sites on the Buffalo River.
Monthly variation in concentrations of total coliforms in water samples collected from the six sites (S1–S6) located on the Buffalo River. Each composite sample, collected monthly at each site, was analysed in triplicate. The triplicate values obtained for each sample were averaged to obtain the results reported
The FC counts were significantly (P < 0.05) higher at King William’s Town in 8 of the 12 months than at all the other sites. In August 2010 and September 2010, although FC counts at King William’s Town and Eluxolzweni were significantly (P < 0.05) higher than the counts recorded at the rest of the sites, the difference between the means at both sites was not significant. For ENT, significantly higher counts were recorded at King William’s Town than at all the other sites throughout the study period, except in June and July during which counts at Parkside were significantly higher. The significantly higher counts of the indicator bacteria recorded at King William’s Town compared to the other sites suggests that this is the most contaminated site.
The general trend suggests that Buffalo River is continuously being polluted with faecal matter from a variety of sources; resulting in for example, severe eutrophication and extensive growth of water hyacinths. The level of algal growth observed at Eluxolzweni stretch of the Buffalo River should not be overlooked. Besides representing the ecological risks attendant to faecal pollution of surface waters, blooms of various planktonic species have been shown to release cyanobacterial toxins into waters thereby presenting an additional water supply hazard (Hitzfeld et al. 2000). The very high counts of indicator bacteria recorded at Parkside (FC, 3.0 × 105 cfu/100 ml and enterococci, 5.3 × 105 cfu/100 ml) in June, which were also signicantly (P < 0.05) higher than counts encountered that month at all the other sites, suggest that sewage was dumped into the Buffalo River around that site.
A part of the spatial variations in FIB concentrations recorded in this study could be attributable to the time of sample collection during each sampling trip (Whitman and Nevers 2004). According to a recent report by the USEPA, previous studies have identified diurnal variation in indicator density in freshwater environments including rivers, streams, and non-flowing inland waters (USEPA 2010). The report shows that all other factors being equal, when measured by culture methods, faecal indicator bacteria demonstrate a predictable pattern of highest density in the morning, decreasing density during the day (often by several orders of magnitude), reaching the lowest density in the mid-afternoon (USEPA 2010). The decrease of indicator bacteria during daylight hours results from inactivation of organisms by incident solar radiation among other factors (Sinton et al. 2002). In this study, sampling at Maden Dam and Rooikrantz Dam were always done between 8 a.m. and 9 a.m., while collection of samples at the remaining sites occurred around noon. This could help explain why, despite limited anthropogenic influences at both dams, the FIB counts at both upper catchment dams (Maden and Rooikrantz) were in some cases not significantly different from those at the other three freshwater sampling sites located in the lower reaches of the Buffalo River, as evident in Figs. 3, 4 and 5, and typified by the concentrations recorded in September, October and June for FC, ENT and TC respectively. Further, despite its location in the lower catchment, Bridle Drift Dam recorded lower FIB counts compared to Maden and Rooikrantz dams at the upper catchment in some of the months (e.g. October for TC and ENT and April for TC and FC). This, however, may not be as a result of reduced faecal pollution, but could be due to dilution in higher water volumes at the Bridle Drift Dam.
Monthly variation in concentrations of faecal coliforms in water samples collected from the six sites (S1–S6) located on the Buffalo River. Each composite sample, collected monthly at each site, was analysed in triplicate. The triplicate values obtained for each sample were averaged to obtain the results reported
Monthly variation in concentrations of enterococci in water samples collected from the six sites (S1–S6) located on the Buffalo River. Each composite sample, collected monthly at each site, was analysed in triplicate. The triplicate values obtained for each sample were averaged to obtain the results reported
Contamination of watercourses with faecal matter represents a significant risk to public health due to the associated risk from human pathogens, and the concentration of indicator microorganisms in a body of water is used to estimate the health risk to users for domestic, irrigational and recreational purposes (Abdelzaher et al. 2010). Table 2 shows the results of the evaluation of the pollution level of the Buffalo River based on bacteriological standards and guidelines. Faecal coliform and even total coliform counts should be zero (per 100 ml) of sample in domestic water supplies, piped or unpiped, treated or untreated (DWAF 1996a; WHO 2008). While all the samples (100 %) exceeded this acceptable limit for TC count, 89 % of the samples yielded counts that were above the FC standard limit, indicating that the quality of Buffalo River water is very poor. Poor water quality poses a serious health risk for rural communities, since many households rely solely on untreated river water for domestic purposes (RHP 2004). The river water poses greater health risks for infants, some of the elderly, and people with severely compromised immune systems. The potential impact is more profound considering the high number of people, in the Eastern Cape, whose immune systems are compromised by HIV/AIDS (Obi et al. 2006).
By the United States Environmental Protection Agency (USEPA) limits, recreational waters with concentrations exceeding the maximum contaminant limit of 33 or 200 cfu/100 ml for ENT or FC respectively presents a health risk (USEPA 1986; Abdelzaher et al. 2010). The South Africa Department of Water Affairs (SA-DWAF) sets target water quality ranges for both parameters of 0–30 cfu/100 ml for ENT and 0–130 cfu/100 ml for FC (DWAF 1996b). The percentage of water samples in this study that exceeded these limits were 71–79 % for FC and 82–85 % for ENT. At Parkside, where recreational activities are most pronounced on the Buffalo River, unacceptable ENT concentrations were observed in 92–100 % of the water samples.
Agriculture is widespread in the middle reaches of Buffalo River catchment (from the foothill zone downstream of Rooikrantz Dam to King William’s Town and as far as Bridle Drift Dam), with local areas of intensive irrigation that provide fresh produce (RHP 2004). Irrigation water used for fruit and vegetable crops can be a potential cause of contamination with microbial pathogens. For unrestricted irrigation (that is, for uses that include crops likely to be eaten uncooked), only 32 % of all the 72 water samples analysed in this study exceeded the WHO guideline for faecal coliform bacteria (≤1000 FC/100 ml) (Blumenthal et al. 2000). This, however, gives an underestimation of the potential hazards that may arise from the use the Buffalo River water in fresh produce irrigation. The USEPA and SA-DWAF have recommended strict guidelines of no detectable faecal coliform bacteria being allowed in 100 ml of water for fresh produce irrigation (USEPA 1992; DWAF 1996c, e). In this study, FC concentrations exceeding this zero limit were detected in 89 % of the water samples.
The bacterial water quality represents significant threats to the health of not only agricultural workers but also the consumers of fresh produce irrigated with the Buffalo River water, as previous studies have demonstrated the presence and prolonged survival of excreted pathogens on the surface of vegetables irrigated with faecally contaminated water (Beuchat 2002; Gemmell and Schmidt 2012). Considering that cattle farming and in situ herd watering are extensive in the catchment, it is worth noting that the carriage of E. coli O157 in cattle (LeJeune et al. 2004) and irrigation with water contaminated by animal faeces were the vital factors in the emergence of this pathogen in South Africa (Effler et al. 2001). The risk attendant to irrigation with contaminated water is heightened by the demonstrated ability of this pathogen and Salmonella to migrate to internal locations in plant tissue and thus gain protection from the action of sanitising agents by virtue of inaccessibility (Solomon et al. 2002; Deering et al. 2012).
Of the three dams serving as source raw water for drinking water production, the biggest, Bridle Drift Dam (with a capacity of 101.6 million cubic metres) is the most polluted, with 58 and 25 % of all samples collected this dam yielding TC and FC counts, respectively, that exceeded international limits. The European Union guide limits for surface waters used as raw water for normal full physical and chemical treatment with disinfection suggests maximum concentrations of <5.0 × 103 TC/100 ml and <2.0 × 103 FC/100 ml, respectively (Tebbut 1992). It is known that most waterborne pathogens have low infective doses (WHO 2008). Knowing also that inadequately treated water may contain pathogens, and that an accidental water treatment failure may pose significant risk to public health, there is an urgent need for provision of adequate sanitary infrastructure that will help prevent source water contamination. People from low socio-economic rural communities in the Buffalo River catchment draw water directly from the river for domestic use which they often drink without any form of treatment like boiling or filtering, as long as it “appears clean”. The USEPA source water quality coliform limits for filtration avoidance required that the FC concentration in water prior to disinfection must not exceed 20/100 ml in at least 90 % of the samples, or that the TC concentration must not exceed 100/100 ml in at least 90 % of the samples (USEPA 2004). The data presented in this study by far exceed these limits that seek to prevent waterborne diseases. The Buffalo River water therefore constitutes a potential health hazard to consumers.
It should be pointed out that specific pathogens were not evaluated in this study. The implication is that there is a probability that a part of the data presented could represent false-negatives (in which FIB were absent in water samples and pathogens were present) or false-positives (FIB were present in the samples and pathogens were absent) results. The risks therefore could have been overestimated or underestimated. In addition, some studies on the relationships between indicator microbes and pathogens have shown that bacteriological indicators might not estimate, reliably, the sanitary risk related to faecal contamination and viral particles in water (Jurzik et al. 2010). However, although in a recent study Abdelzaher et al. (2010) assessed the presence of selected members all three classes of pathogens (viral, protozoan, and bacterial) as well as indicator microbes, the assessment of all possible pathogens is not economically, technologically, or practically feasible. The determination of faecal indicators, such as faecal coliforms or E. coli, as a means for assessing faecal pollution in environmental freshwaters in temperate regions like Europe and North America is widely accepted (Toranzos and McFeters 1997). Enterococci are recommended for assessing coastal water quality (Ahmed et al. 2009; Abdelzaher et al. 2010).
Conclusions
The distinct increase in bacterial indicators as the Buffalo River flowed from its source downstream through settlements reveals the deterioration of the water quality and reflects the degradating impact of settlements and anthropogenic activities on the quality of the river. Our results indicate that the bacteriological water quality of the Buffalo River and dams are poor. The use of these source waters for fresh produce irrigation, full-contact recreation, domestic and herd watering purposes therefore represent significant public health hazards. Future research should focus on the assessment of these surface waters for the presence of bacterial and viral pathogens in the interest of public health. Provision of adequate sanitary infrastructure will help prevent source water contamination, and public health education aimed at improving personal, household and community hygiene is imperative.
References
Abdelzaher AM, Wright ME, Ortega C, Solo-Gabriele HM, Miller G, Elmir S, Newman X, Bonilla JA, Bonilla TD et al (2010) Presence of pathogens and indicator microbes at a non-point source subtropical recreational marine beach. Appl Environ Microbiol 76:724–732
Ahmed W, Goonetilleke A, Powell D, Chauhan K, Gardner T (2009) Comparison of molecular markers to detect fresh sewage in environmental waters. Water Res 43:4908–4917
American Public Health Association (APHA) (1998) Standard methods for the examination of water and wastewater. 20thEdn. American Public Health Association, American Water Works Association, Water Environment Federation: Washington DC, USA
Anderson KL, Whitlock JE, Harwood VJ (2005) Persistence and differential survival of fecal indicator bacteria in subtropical waters and sediments. Appl Environ Microbiol 71:3041–3048
Ash RJ, Mauck B, Morgan M (2002) Antibiotic resistance of gram-negative bacteria in rivers, United States. Emerg Infect Dis 8:713–716
Bernier J-LT, Maheux AF, Boissinot M, Picard FJ, Bissonnette L, Martin D, Dewailly E, Bergeron MG (2009) Onsite microbiological quality monitoring of raw source water in Cree community of Mistissini. Water Quality Research Journal of Canada 44:345–354
Beuchat LR (2002) Ecological factors influencing survival and growth of human pathogens on raw fruits and vegetables. Microbes and Infection 4:413–423
Bielaszewska M, Mellmann A, Zhang W, Kock R, Fruth A, Bauwens A, Peters G, Karch H (2011) Characterisation of the Escherichia coli strain associated with an outbreak of haemolytic uraemic syndrome in Germany, 2011: a microbiological study. Lancet Infect Dis 11:671–76
Blumenthal UJ, Mara DD, Peasey A, Ruiz-Palacios G, Stott R (2000) Guidelines for the microbiological quality of treated wastewater used in agriculture: recommendations for revising WHO guidelines. Bulletin of the World Health Organization 78:1104–1116
Bosch A, Guix S, Sano D, Pinto RM (2008) New tools for the study and direct surveillance of viral pathogens in water. Current Opinions in Biotechnology 19:1–7
Casadio A, Maglionico M, Bolognesi A, Artina S (2010) Toxicity and pollutant impact analysis in an urban river due to combined sewer overflows loads. Water Sci Technol 61:207–215
Chigor VN, Umoh VJ, Okuofu CA, Ameh JB, Igbinosa EO, Okoh AI (2012) Water quality assessment: surface water sources used for drinking and irrigation in Zaria, Nigeria are a public health hazard. Environ Monit Assess 184:3389–3400
Chigor VN, Umoh VJ, Smith SI (2010a) Occurrence of Escherichia coli O157 in a river used for fresh produce irrigation in Nigeria. Afr J Biotechnol 9:178–182
Chigor VN, Umoh VJ, Smith SI, Igbinosa EO, Okoh AI (2010b) Multidrug resistance and plasmid patterns of Escherichia coli O157 and other E. coli isolated from diarrhoeal stools and surface waters from some selected sources in Zaria, Nigeria. International Journal of Environmental Research and Public Health 7:3831–3841
Clarke CL, Shackleton SE, Powell M (2012) Climate change perceptions, drought responses and views on carbon farming amongst commercial livestock and game farmers in the semiarid Great Fish River Valley, Eastern Cape province, South Africa. African Journal of Range & Forage Science 29:13–23
Davies CM, Long JA, Donald M, Ashbolt NJ (1995) Survival of fecal microorganisms in marine and freshwater sediments. Appl Environ Microbiol 61:1888–1896
Deering AJ, Mauer LJ, Pruitt RE (2012) Internalization of E. coli O157:H7 and Salmonella spp. in plants: A review. Food Res Int 45:567–575
Department of Water Affairs (DWAF) (2010) Strategic Plan 2010/11–2012. DWAF, Pretoria, South Africa. http://www.dwaf.gov.za/documents/Other/Strategic%20Plan/StrategicPlan2010-2013.pdf
Department of Water Affairs (DWAF) (2012) 2012 Green Drop Progress Report. Department of Water Affairs; Pretoria, Republic of South Africa
Department of Water Affairs and Forestry (DWAF) (1996a) South African water quality guidelines (2nd Edn.). Vol. 1: Domestic Use. Pretoria: Department of Water Affairs and Forestry
Department of water affairs and forestry (DWAF) (1996b) South African water quality guidelines (2nd Edn.). Vol. 2: Recreational Use. Pretoria: Department of Water Affairs and Forestry
Department of water affairs and forestry (DWAF) (1996c) South African water quality guidelines (2nd Edn.). Vol. 4: Agricultural Use: Irrigation. Pretoria: Department of Water Affairs and Forestry
Department of Water Affairs and Forestry (DWAF) (1996d) South African water quality guidelines (2nd Edn.). Vol. 5: Agricultural Use: Livestock Watering. Pretoria: Department of Water Affairs and Forestry
Department of Water Affairs and Forestry (DWAF) (1996e) South African water quality guidelines (1st Edn.). Vol. 8: Field Guide Pretoria: Department of Water Affairs and Forestry
Department of Water Affairs and Forestry (DWAF) (2004) Annual Report 2002/2003. DWAF, Pretoria, South Africa
Diergaardt SM, Venter SN, Spreeth A, Theron J, Brozel VS (2004) The occurrence of campylobacters in water sources in South Africa. Water Res 38:2589–2595
Drayna P, McLellan SL, Simpson P, Li S-H, Gorelick MH (2010) Association between rainfall and pediatric emergency department visits for acute gastrointestinal illness. Environ Heal Perspect 118:1439–1443
Effler P, Isaacson M, Arntzen L, Heenan R, Canter P, Barrett T, Lee L, Mamba C, Levine W, Zaidi A, Griffin PM (2001) Factors contributing to the emergence of Escherichia coli O157:H7 in Africa. Emerg Infect Dis 7:812–819
Fong TT, Lipp EK (2005) Enteric viruses of human and animals in aquatic environments: heath risks, detection and potential water quality assessment tools. Appl Environ Microbiol 69:357–371
Gemmell ME, Schmidt S (2012) Microbiological assessment of river water used for the irrigation of fresh produce in a sub-urban community in Sobantu, South Africa. Food Res Int 47:300–305
Gersberg RM, Rose MA, Robles-Sikisaka R, Dhar AK (2006) Quantitative detection of hepatitis A virus and enteroviruses near the United States–Mexico border and correlation with levels of fecal indicator bacteria. Appl Environ Microbiol 72:7438–7444
Gourmelon M, Caprais MP, Segura R, Le Mennec C, Lozach S, Piriou JY, Rince A (2007) Evaluation of two library-independent microbial source tracking methods to identify sources of fecal contamination in French estuaries. Appl Environ Microbiol 73:4857–4866
Harwood VJ, Levine AD, Scott TM, Chivukula V, Lukasik J, Farrah SR, Rose JB (2005) Validity of the indicator organism paradigm for pathogen reduction in reclaimed water and public health protection. Appl Environ Microbiol 71:6.3163–3170
He JW, Jiang S (2005) Quantification of enterococci and human adenoviruses in environmental samples by real-time PCR. Appl Environ Microbiol 71:2250–2255
Hitzfeld, B.C., Hoger, S.J. and Dietrich, D.R (2000). Cyanobacterial toxins: removal during drinking water treatment and risk assessment. Environmental Health Perspectives 108, 113S1-122S1
Hunter PR (2003) Climate change and waterborne and vector-borne disease. J Appl Microbiol 94:37S–46S
Igbinosa EO, Okoh AI (2009) Impact of discharge wastewater effluents on the physico-chemical qualities of a receiving watershed in a typical rural community. Int J Environ Sci Technol 6:175–182
Jagals P (1997) Stormwater runoff from typical developed and developing South African urban developments: Definitely not for swimming. Water Sci Technol 35:133–140
Jurzik L, Hamzaa IA, Puchertb W, Überlac K, Wilhelma M (2010) Chemical and microbiological parameters as possible indicators for human enteric viruses in surface water. Int J Hyg Environ Health 213:210–216
Kistemann T, Claßen T, Koch C, Dagendorf F, Fischeder R, Gebel J, Vacata V, Exner M (2002) Microbial load of drinking water reservoir tributaries during extreme rainfall and runoff. Appl Environ Microbiol 68:2188–2187
Lata P, Ram S, Agrawal M, Shanker R (2009) Enterococci in river Ganga surface waters: propensity of species distribution, dissemination of antimicrobial-resistance and virulence-markers among species along landscape. BMC Microbiol 9:140
LeJeune JT, Besser TE, Rice DH, Berg JL, Stilborn RP, Hancock DD (2004) Longitudinal study of fecal shedding of Escherichia coli O157:H7 in feedlot cattle: predominance and persistence of specific clonal types despite massive cattle population turnover. Applied And Environmental Microbiology 70:377–384
MacIntyre UE, de Villiers FPR (2010) The economic burden of diarrheal disease in a tertiary level hospital, Gauteng, South Africa. J Infect Dis 202:S116–S125
Mills-Robertson F, Crupper SS, Addy ME, Mensah P (2003) Antibiotic resistance and genotyping of clinical group B Salmonella isolated in Accra, Ghana. J Appl Microbiol 94:289–294
Mohanty JC, Ford TE, Harrington JJ, Laksmipathy V (2002) A cross-sectional study of enteric disease risks associated with water quality and sanitation in Hyderabad City. J Water Supply Res Technol 51:239–251
Morrison G, Fatoki OS, Persson L, Ekberg A (2001) Assessment of the impact of point source pollution from the Keiskammahoek sewage treatment plant on the Keiskamma River - pH, electrical conductivity, oxygen- demanding substance (COD) and nutrients. Water SA 27:475–480
Muller EE, Ehlers MM, Grabow WOK (2001) The occurrence of E. coli O157 in South African water sources intended for direct and indirect human consumption. Water Res 35:085–3088
Municipal Demarcation Board (MDB) (2010). Municipal demarcation board statistics. http://www.demarcation.org.za Retrieved 13/07/2010
Obi CL, Green E, Bessong PO, Villiers B, Hoosen AA, Igumbor EO, Potgieter N (2004) Gene encoding virulence markers among Escherichia coli isolates from diarrhoeic stool samples and river sources in rural Venda communities of South Africa. Water SA 30:37–42
Obi CL, Onabolu B, Momba MNB, Igumbor JO, Ramalivahna J, van Rensburg EJ, Lukoto M, Bessong PO, Green E, Mulaudzi TB (2006) The interesting cross-paths of HIV/AIDS and water in Southern Africa with special reference to South Africa. Water SA 32:323–344
Obi CL, Potgieter N, Bessong PO, Matsaung G (2002) Assessment of the microbial quality of river water sources in rural Venda communities in South Africa. Water SA 28:287–292
Odjadjare EEO, Okoh AI (2010) Prevalence and distribution of Listeria pathogens in the final effluents of a rural wastewater treatment facility in the Eastern Cape Province of South Africa. World J Microbiol Biotechnol 26:297–307
Odjadjare EEO, Obi LC, Okoh AI (2010) Municipal wastewater effluents as a source of listerial pathogens in the aquatic milieu of the Eastern Cape Province of South Africa: a concern of public health importance. International Journal of Environmental Research and Public Health 7:2376–2394
Okoh AI, Odjadjare EE, Igbinosa EO, Osode AN (2007) Wastewater treatment plants as a source of microbial pathogens in the receiving watershed. Africa Journal of Biotechnology 6:2932–2944
Payment, P. and Riley, S.M. (2002) Resolving the global burden of gastrointestinal illness: A call to action. American Academy of Microbiology. Washington DC, United States of America 1–25.
Pruss A, Kay D, Fewtrell L, Bartram J (2002) Estimating the burden of disease from water sanitation and hygiene at a global level. Environ Heal Perspect 110:537–542
River Health Programme (2004) State-of-Rivers Report: Buffalo River System. Department of Water Affairs and Forestry, Pretoria, South Africa, 1–41
Schets FM, van Wijnen JH, Schijven JF, Schoon H, de Roda Husman AM (2008) Monitoring of waterborne pathogens in surface waters in Amsterdam, The Netherlands, and the potential health risk associated with exposure to Cryptosporidium and Giardia in these waters. Appl Environ Microbiol 74:2069–2078
Shuval, H I (1990) Wastewater irrigation in developing countries: health effects and technical solutions. Summary of World Bank Technical Paper No. 51. The World Bank, Washington DC, United States of America, 1-19.
Sinton LW, Hall CH, Lynch PA, Davies-Colley RJ (2002) Sunlight inactivation of fecal indicator bacteria and bacteriophages from waste stabilization pond effluent in fresh and saline waters. Appl Environ Microbiol 68:1122–1131
Solomon EB, Yaron S, Mathews KR (2002) Transmission of Escherichia coli 0157: H7 from contaminated manure and irrigation water to lettuce plant tissue and its subsequent internalization. Appl Environ Microbiol 68:397–400
Stewart-Tull DES (2001) Vaba, hariza, kholera, foklume or cholera: in any language, still the disease of seven pandemics. J Appl Microbiol 91:580–581
Tebbut THY (1992) Principles of water quality. Pergamon, Oxford, England, pp 1–251
Toranzos GA, McFeters GA (1997) Detection of indicator microorganisms in environmental fresh waters. In: Hurst CJ, Knudsen GR, McInerney MJ, Stetzenbach LD, Walter MV (eds) Manual of environmental microbiology. American Society for Microbiology, Washington, D.C., pp 184–194
United States Environmental Protection Agency (USEPA) (1986) Ambient water quality criteria for bacteria. EPA440/5-84-002, U.S. Environmental Protection Agency Office of Water. Washington, DC
United States Environmental Protection Agency (USEPA) (1992) Guidelines for water reuse. EPA/625/R-92/004, U.S. Environmental Protection Agency Office of Wastewater Enforcement and Compliance. Washington, DC
United States Environmental Protection Agency (USEPA) (2004) Comprehensive surface water treatment rules quick reference guide: unfiltered systems. EPA 816-F-04-001, U.S. Environmental Protection Agency Office of Water. Washington, DC
United States environmental protection agency (USEPA) (2010) Sampling and consideration of variability (temporal and spatial) for monitoring of recreational waters. EPA-823-R-10-005 U.S. Environmental Protection Agency Office of Water, Washington, DC
Whitman RL, Nevers MB (2004) Escherichia coli sampling reliability at a frequently closed Chicago beach: monitoring and management implications. Environ Sci Technol 38:4241–4246
Wilczynski JA, Peters S, House J, Hill V, Schneeberger C, Xiao L, Dearen T, Webeck J (2012) Outbreak of cryptosporidiosis associated with a firefighting response—Indiana and Michigan, June 2011. MMWR Morb Mortal Wkly Rep 61:153–156
Williams AP, Quilliam RS, Thorn CE, Cooper D, Reynolds B, Jones DL (2012) Influence of land use and nutrient flux on metabolic activity of E. coli O157 in river water. Water Air Soil Pollut 223:3077–3083
World Health Organization (2008) Guidelines to drinking water quality (3rd Edn). Vol. 1 World Health Organization. Geneva, Switzerland pp.1-666
Zahid MSH, Udden SMN, Faruque ASG, Calderwood SB, Mekalanos JJ, Faruque SM (2008) Effect of phage on the infectivity of Vibrio cholerae and emergence of genetic variants. Infect Immun 76:5266–5273
Zhang XX, Zhang T, Fang HHP (2009) Antibiotic resistance genes in water environment. Appl Microbiol Biotechnol 82:397–414
Acknowledgements
The authors are grateful to the Water Research Commission (WRC) of South Africa for funding this research and the Education Trust Fund of Nigeria for study bursary for VNC.
Open Access
This article is distributed under the terms of the Creative Commons Attribution License which permits any use, distribution, and reproduction in any medium, provided the original author(s) and the source are credited.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Robert Duran
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOC 4589 kb)
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution License which permits any use, distribution, and reproduction in any medium, provided the original author(s) and the source are credited.
About this article
Cite this article
Chigor, V.N., Sibanda, T. & Okoh, A.I. Studies on the bacteriological qualities of the Buffalo River and three source water dams along its course in the Eastern Cape Province of South Africa. Environ Sci Pollut Res 20, 4125–4136 (2013). https://doi.org/10.1007/s11356-012-1348-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-012-1348-4