Abstract
Purpose
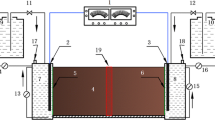
In this study, a novel and ecological alternative have been developed to treat soils contaminated with hexavalent chromium coupling two well-known systems: electrokinetic remediation and permeable reactive biobarriers. The electric field promotes the electromigration of the hexavalent chromium oxyanions towards the anode. The biobarriers were placed before the anode electrode, in order to promote the reduction and retention of the chromium migrating in its direction. Thus, this technology provided a global treatment to soil removal without subsequent treatments of the contaminated effluents.
Methods
The electrokinetic system was coupled with two different permeable reactive biobarriers composed by Arthrobacter viscosus bacteria, supported either in activated carbon or zeolite. An electric field of 10 V was applied and two different treatment times of 9 and 18 days were tested.
Results
Removal values of 60% and 79% were obtained when electrokinetic treatment was coupled with zeolite and activated carbon biobarriers, respectively, for a test period of 18 day. The reduction of hexavalent chromium to trivalent chromium was around 45% for both systems.
Conclusions
In this work, two types of biobarriers were efficiently coupled to electrokinetic treatment to decontaminate soil with Cr(VI). Furthermore, the viability of the new coupling technology developed (electrokinetic + biobarriers) to treat low-permeability polluted soils was demonstrated.






Similar content being viewed by others
References
Alcantara MT, Gomez J, Pazos M, Sanroman MA (2008) Combined treatment of PAHs contaminated soils using the sequence extraction with surfactant-electrochemical degradation. Chemosphere 70:1438–1444
Ali I (2010) The quest for active carbon adsorbent substitutes: inexpensive adsorbents for toxic metal ions removal from wastewater. Sep Purif Rev 39:95–171
Ali I, Gupta VK (2007) Advances in water treatment by adsorption technology. Nat Protoc 1:2661–2667
Ali I, Khan TA, Asim M (2011) Removal of arsenic from water by electrocoagulation and electrodialysis techniques. Sep Purif Rev 40:25–42
Baraud F, Fourcade MC, Tellier S, Astruc M (1997a) Modelling of decontamination rate in an electrokinetic soil processing. Int J Environ Anal Chem 68:105–121
Baraud F, Tellier S, Astruc M (1997b) Ion velocity in soil solution during electrokinetic remediation. J Hazard Mater 56:315–332
Boni MR, Sbaffoni S (2009) The potential of compost-based biobarriers for Cr(VI) removal from contaminated groundwater: column test. J Hazard Mater 166:1087–1095
Candela L, Álvarez-Benedí J, Condesso de Melo MT, Rao PSC (2007) Laboratory studies on glyphosate transport in soils of the Maresme area near Barcelona, Spain: transport model parameter estimation. Geoderma 140:8–16
Cristani M, Naccari C, Nostro A, Pizzimenti A, Trombetta D, Pizzimenti F (2011) Possible use of Serratia marcescens in toxic metal biosorption (removal). Environmen Sci Pollut Res 1–8
Fendorf SE (1995) Surface reactions of chromium in soils and waters. Geoderma 67:55–71
Figueiredo H, Silva B, Quintelas C, Neves IC, Tavares T (2010a) Effect of the supporting zeolite structure on Cr biosorption: performance of a single-step reactor and of a sequential batch reactor—a comparison study. Chem Eng J 163:22–27
Figueiredo H, Silva B, Quintelas C, Pereira MFR, Neves IC, Tavares T (2010b) Biosorption of hexavalent chromium based on modified Y zeolites obtained by alkali-treatment. Environ Eng Manag J 9:305–311
Figueiredo H, Silva B, Quintelas C, Raposo MMM, Parpot P, Fonseca AM, Lewandowska AE, Bañares MA, Neves IC, Tavares T (2010c) Immobilization of chromium complexes in zeolite Y obtained from biosorbents: synthesis, characterization and catalytic behaviour. Appl Catal Environ 94:1–7
Fonseca B, Teixeira A, Figueiredo H, Tavares T (2009) Modelling of the Cr(VI) transport in typical soils of the North of Portugal. J Hazard Mater 167:756–762
Gupta VK, Ali I (2004) Removal of lead and chromium from wastewater using bagasse fly ash—a sugar industry waste. J Colloid Interface Sci 271:321–328
Gupta VK, Rastogi A (2008) Sorption and desorption studies of chromium(VI) from nonviable cyanobacterium Nostoc muscorum biomass. J Hazard Mater 154:347–354
Gupta VK, Rastogi A (2009) Biosorption of hexavalent chromium by raw and acid-treated green alga Oedogonium hatei from aqueous solutions. J Hazard Mater 163:396–402
Gupta VK, Mohan D, Sharma S, Park KT (1999) Removal of chromium(VI) from electroplating industry wastewater using bagasse fly ash—a sugar industry waste material. Environmentalist 19:129–136
Gupta VK, Gupta M, Sharma S (2001) Process development for the removal of lead and chromium from aqueous solutions using red mud—an aluminium industry waste. Water Res 35:1125–1134
Gupta VK, Jain AK, Kumar P, Agarwal S, Maheshwari G (2006) Chromium(III)-selective sensor based on tri-o-thymotide in PVC matrix. Sens Actuators B: Chem 113:182–186
Gupta VK, Carrott PJM, Ribeiro Carrott MML, Suhas (2009) Low-cost adsorbents: growing approach to wastewater treatment—a review. Crit Rev Environ Sci Technol 39:783–842
Gupta VK, Rastogi A, Nayak A (2010) Adsorption studies on the removal of hexavalent chromium from aqueous solution using a low cost fertilizer industry waste material. J Colloid Interface Sci 342:135–141
Gupta VK, Agarwal S, Saleh TA (2011) Chromium removal by combining the magnetic properties of iron oxide with adsorption properties of carbon nanotubes. Water Res 45:2207–2212
Kanagaraj J, Mandal AB (2011) Combined biodegradation and ozonation for removal of tannins and dyes for the reduction of pollution loads. Environ Sci Pollut Res. 1–11
Machado MD, Soares EV, Soares HMVM (2011) Selective recovery of chromium, copper, nickel, and zinc from an acid solution using an environmentally friendly process. Environ Sci Pollut Res 18:1279–1285
Mohan D, Singh KP, Singh VK (2006) Trivalent chromium removal from wastewater using low cost activated carbon derived from agricultural waste material and activated carbon fabric cloth. J Hazard Mater 135:280–295
Ouki SK, Neufeld RD (1997) Use of activated carbon for the recovery of chromium from industrial wastewaters. J Chem Technol Biotechnol 70:3–8
Pazos M, Gouveia S, Sanroman MA, Cameselle C (2008) Electromigration of Mn, Fe, Cu and Zn with citric acid in contaminated clay. J Environ Sci Health, Part A: Tox Hazard Subst Environ Eng 43:823–831
Pazos M, Alcántara MT, Cameselle C, Sanromán MA (2009) Evaluation of electrokinetic technique for industrial waste decontamination. Sep Sci Technol 44:2304–2321
Pazos M, Branco M, Neves IC, Sanromán MA, Tavares T (2010) Removal of Cr(VI) from aqueous solutions by a bacterial biofilm supported on zeolite: Optimisation of the operational conditions and scale-up of the bioreactor. Chem Eng Technol 33:2008–2014
Priyantha N, Bandaranayaka A (2011) Interaction of Cr(VI) species with thermally treated brick clay. Environ Sci Pollut Res 18:75–81
Quintelas C, Tavares T (2001) Removal of chromium(VI) and cadmium(II) from aqueous solution by a bacterial biofilm supported on granular activated carbon. Biotechnol Lett 23:1349–1353
Quintelas C, Fernandes B, Castro J, Figueiredo H, Tavares T (2008) Biosorption of Cr(VI) by three different bacterial species supported on granular activated carbon—a comparative study. J Hazard Mater 153:799–809
Quintelas C, Fonseca B, Silva B, Figueiredo H, Tavares T (2009) Treatment of chromium(VI) solutions in a pilot-scale bioreactor through a biofilm of Arthrobacter viscosus supported on GAC. Bioresour Technol 100:220–226
Reddy KR, Chinthamreddy S (2003) Effects of initial form of chromium on electrokinetic remediation in clays. Adv Environ Res 7:353–365
Silva B, Figueiredo H, Quintelas C, Neves IC, Tavares T (2008) Zeolites as supports for the biorecovery of hexavalent and trivalent chromium. Microporous Mesoporous Mater 116:555–560
Silva B, Figueiredo H, Neves IC, Tavares T (2009) The role of pH on Cr(VI) reduction and removal by Arthrobacter viscous. Int J Chem Biol Eng 2:100–103
Singh AK, Gupta VK, Gupta B (2007) Chromium(III) selective membrane sensors based on Schiff bases as chelating ionophores. Anal Chim Acta 585:171–178
Teutli-León MM, Oropeza MT, González I, Soria A (2005) Mathematical modeling of a galvanostatic soil electroremediation process. AICHE J 51:1822–1833
Toride N, Leij FJ, van Genuchten MT (1995) The CXTFIT code for estimating transport parameters from laboratory or field tracer experiments. U.S. Salinity Laboratory, U.S. Department of Agriculture, Riverside
USEPA (1992) Chromium, hexavalent (colorimetric). 7196A. USEPA
USEPA (1996) Alkaline digestion for hexavalent chromium. 3060A. USEPA
USEPA (2004) Soil and waste pH. Method 9045D. USEPA
USEPA (2007) Microwave assisted acid digestion of sediments, sludges, soils, and oils. 3051. USEPA
van Genuchten MT (1981) Non equilibrium transport parameters from miscible displacement experiments. U.S. Salinity Laboratory, U.S. Department of Agriculture, Riverside
Viamajala S, Peyton BM, Gerlach R, Sivaswamy V, Apel WA, Petersen JN (2008) Permeable reactive biobarriers for in situ Cr(VI) reduction: bench scale tests using Cellulomonas sp. strain ES6. Biotechnol Bioeng 101:1150–1162
Wu D, Sui Y, He S, Wang X, Li C, Kong H (2008) Removal of trivalent chromium from aqueous solution by zeolite synthesized from coal fly ash. J Hazard Mater 155:415–423
Acknowledgments
This work was supported by the Spanish Ministry of Science and Innovation (CTQ2008-03059/PPQ), Xunta de Galicia (08MDS034314PR). The authors are grateful to the Spanish Ministry of Science and Innovation for providing financial support for Marta Pazos under the Ramón y Cajal program and the Fundação para a Ciência e Tecnologia, Ministério da Ciência e Tecnologia, Portugal through the PhD grant of Bruna Fonseca (SFRH/BD/27780/2006).
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor Vinod Kumar Gupta
Rights and permissions
About this article
Cite this article
Fonseca, B., Pazos, M., Tavares, T. et al. Removal of hexavalent chromium of contaminated soil by coupling electrokinetic remediation and permeable reactive biobarriers. Environ Sci Pollut Res 19, 1800–1808 (2012). https://doi.org/10.1007/s11356-011-0694-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-011-0694-y