Abstract
Purpose
Parkinson’s disease (PD) is one of the most common neurodegenerative disorders. This study compared the effects of two types of exercises on motor dysfunction in characterizing adult male Wistar rats with unilateral 6-hydroxydopamine lesions placed in the median forebrain bundle.
Methods
Sixty adult male Wistar rats were randomly assigned to six groups: the control intact and Parkinson group (no exercise), the forced group (sham, PD), the voluntary group (sham, PD). The effects of 4 weeks of forced treadmill running and voluntary wheel running on motor behavior were investigated in the open field, Rotarod and hanging tests.
Results
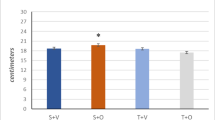
The outcomes showed that although the length moved by the voluntary exercise group was more than that in the forced exercise, the forced exercise resulted in better performance in Rotarod (P = 0.01), whereas voluntary exercise was found to increase rearing behavior (P = 0.01). Both exercise groups were the same in the open field (p > 0.05), grooming frequency (P = 0.12), and hanging test (P = 0.181).
Conclusion
We found that wheel running and treadmill running exercise protocols improved the motor impairments in the 6-OHDA-lesioned rat model of Parkinson’s disease; therefore, our result suggests that both exercise methods could be beneficial for PD patients to reduce their motor dysfunction.







Similar content being viewed by others
References
Aguiar AS et al (2009) Physical exercise improves motor and short-term social memory deficits in reserpinized rats. Brain Res Bull 79(6):452–457
de Rijk MD et al (1997) Prevalence of parkinsonism and Parkinson’s disease in Europe: the EUROPARKINSON Collaborative Study. European Community Concerted Action on the Epidemiology of Parkinson’s disease. Journal of Neurology Neurosurgery. Psychiatry 62(1):10–15
Huang Y et al (2004) Genetic contributions to Parkinson’s disease. Brain Res Rev 46(1):44–70
Fahn S (2003) Description of Parkinson’s disease as a clinical syndrome. Ann N Y Acad Sci 991(1):1–14
Smith BA, Goldberg NR, Meshul CK (2011) Effects of treadmill exercise on behavioral recovery and neural changes in the substantia nigra and striatum of the 1-methyl-4-phenyl-1,2, 3, 6-tetrahydropyridine-lesioned mouse. Brain Res 1386:70–80
Petzinger GM et al (2013) Exercise-enhanced neuroplasticity targeting motor and cognitive circuitry in Parkinson’s disease. Lancet Neurol 12(7):716–726
Paillé V et al (2007) Rat model of Parkinson’s disease with bilateral motor abnormalities, reversible with levodopa, and dyskinesias. Mov Disord 22(4):533–539
Julien C et al (2006) Postmortem brain fatty acid profile of levodopa-treated Parkinson disease patients and parkinsonian monkeys. Neurochem Int 48(5):404–414
Samadi P et al (2006) Docosahexaenoic acid reduces levodopa-induced dyskinesias in 1-methyl-4-phenyl-1, 2, 3, 6-tetrahydropyridine monkeys. Annals of neurology 59(2):282–288
Schapira AH et al (2006) Novel pharmacological targets for the treatment of Parkinson’s disease. Nat Rev Drug Discovery 5(10):845–854
Kordower JH et al (2013) Disease duration and the integrity of the nigrostriatal system in Parkinson’s disease. Brain 136(8):2419–2431
Wang Z et al (2013) Functional reorganization of motor and limbic circuits after exercise training in a rat model of bilateral parkinsonism. PLoS ONE 8(11):e80058
Schallert T et al (2000) CNS plasticity and assessment of forelimb sensorimotor outcome in unilateral rat models of stroke, cortical ablation, parkinsonism and spinal cord injury. Neuropharmacology 39(5):777–787
Tillerson JL et al (2001) Forced limb-use effects on the behavioral and neurochemical effects of 6-hydroxydopamine. J Neurosci 21(12):4427–4435
Kim ST et al (2010) Vertical grid test and modified horizontal grid test are sensitive methods for evaluating motor dysfunctions in the MPTP mouse model of Parkinson’s disease. Brainresearch 1306:176–183
Moscatelli F et al (2016) Relationship between blood lactate and cortical excitability between taekwondo athletes and non-athletes after hand-grip exercise. Somatosens Motor Res 33(2):137–144
Sunvisson H et al (1997) Changes in motor performance in persons with Parkinson’s disease after exercise in a mountain area. J Neurosci Nurs 29(4):255–260
Chen H et al (2005) Physical activity and the risk of Parkinson disease. Neurology 64(4):664–669
Kuroda K et al (1992) Effect of physical exercise on mortality in patients with Parkinson’s disease. Acta Neurol Scand 86(1):55–59
Goodwin VA et al (2008) The effectiveness of exercise interventions for people with Parkinson’s disease: a systematic review and meta-analysis. Mov Disord 23(5):631–640
Herman T, Giladi N, Hausdorff J (2009) Treadmill training for the treatment of gait disturbances in people with Parkinson’s disease: a mini-review. J Neuraltransmission 116(3):307–318
Kwakkel G, De Goede C, Van Wegen E (2007) Impact of physical therapy for Parkinson’s disease: a critical review of the literature. Parkinsonism Relat Disord 13:S478–S487
Tillerson J et al (2003) Exercise induces behavioral recovery and attenuates neurochemical deficits in rodent models of Parkinson’s disease. Neuroscience 119(3):899–911
Petzinger GM et al (2010) Enhancing neuroplasticity in the basal ganglia: the role of exercise in Parkinson’s disease. Mov Disord 25(S1):S141–S145
Tillerson JL et al (2002) Forced nonuse in unilateral parkinsonian rats exacerbates injury. J Neurosci 22(15):6790–6799
Cohen AD et al (2003) Neuroprotective effects of prior limb use in 6-hydroxydopamine-treatedrats: possible role of GDNF. J Neurochem 85(2):299–305
Bezard E, Gross CE, Brotchie JM (2003) Presymptomatic compensation in Parkinson’s disease is not dopamine-mediated. Trends Neurosci 26(4):215–221
Faherty CJ et al (2005) Environmental enrichment in adulthood eliminates neuronal death in experimental parkinsonism. Mol Brain Res 134(1):170–179
Anstrom KK et al (2007) Repetitive vibrissae-elicited forelimb placing before and immediately after unilateral 6-hydroxydopamine improves outcome in a model of Parkinson’s disease. Behav Brain Res 179(2):183–191
Kim D-H et al (2010) Treadmill exercise inhibits traumatic brain injury-induced hippocampalapoptosis. Physiol Behav 101(5):660–665
Kim S-E et al (2010) Treadmill exercise prevents aging-induced failure of memory through an increase in neurogenesis and suppression of apoptosis in rat hippocampus. Exp Gerontol 45(5):357–365
Sim Y-J et al (2004) Treadmill exercise improves short-term memory by suppressing ischemia-induced apoptosis of neuronal cells in gerbils. Neurosci Lett 372(3):256–261
Bove J, Perier C (2012) Neurotoxin-based models of Parkinson’s disease. Neuroscience 211:51–76
Farbood Y et al (2015) Ellagic acid protects the brain against 6-hydroxydopamine induced neuroinflammation in a rat model of Parkinson’s disease. Basic Clin Neurosci 6(2):83–90
Watson C, Paxinos G, Kayalioglu G (2009) The spinal cord: a Christopher and Dana Reeve Foundation text and atlas. Academic press, New York
Sarkaki A, Eidypour Z, Motamedi F (2012) Motor disturbances and thalamic electrical power of frequency bands’ improve by grape seed extract in animal model of Parkinson’s disease. Avicenna J Phytomed 2(4):222
Razavinasab M et al (2013) Pharmacological blockade of TRPV1 receptors modulates the effects of 6-OHDA on motor and cognitive functions in a rat model of Parkinson’s disease. Fundam Clin Pharmacol 27(6):632–640
Shabani M et al (2012) Profound destructive effects of adolescent exposure to vincristine accompanied with some sex differences in motor and memory performance. Can J Physiol Pharmacol 90(4):379–386
Al-Jarrah M et al (2007) Endurance exercise promotes cardiorespiratory rehabilitation without neurorestoration in the chronic mouse model of parkinsonism with severeneurodegeneration. Neuroscience 149(1):28–37
Gorton LM et al (2010) Exercise effects on motor and affective behavior and catecholamine neurochemistry in the MPTP-lesioned mouse. Behav Brain Res 213(2):253–262
Pan Q et al (2015) Impaired voluntary wheel running behavior in the unilateral 6-hydroxydopamine rat model of Parkinson’s disease. J Korean Neurosurg Soc 57(2):82–87
Howells FM et al (2005) Stress reduces the neuroprotective effect of exercise in a rat model for Parkinson’s disease. Behav Brain Res 165(2):210–220
Sconce M et al (2015) Intervention with exercise restores motor deficits but not nigrostriatal loss in a progressive MPTP mouse model of Parkinson’s disease. Neuroscience 299:156174
Mabandla M et al (2004) Voluntary running provides neuroprotection in rats after 6-hydroxydopamine Injection into the medial forebrain bundle. Metabolicbraindisease 19(1–2):43–50
Baatile J et al (2000) Effect of exercise on perceived quality of life of individuals with Parkinson’s disease. J Rehabil Res Dev 37(5):529
Ahlskog JE (2011) Does vigorous exercise have a neuroprotective effect in Parkinson disease? Neurology 77(3):288–294
Nieuwboer A et al (2009) Motor learning in Parkinson’s disease: limitations and potential for rehabilitation. Parkinsonism Relat Disord 15:S53–S58
Deumens R, Blokland A, Prickaerts J (2002) Modeling Parkinson’s disease in rats: an evaluation of 6-OHDA lesions of the nigrostriatal pathway. Exp Neurol 175(2):303–317
Burghardt PR et al (2004) The effects of chronic treadmill and wheel running on behavior in rats. Brain Res 1019(1):84–96
Leasure J, Jones M (2008) Forced and voluntary exercise differentially affect brain and behavior. Neuroscience 156(3):456–465
Kinni H et al (2011) Cerebral metabolism after forced or voluntary physical exercise. Brainresearch 1388:48–55
Lambert M et al (1996) Tests of running performance do not predict subsequent spontaneous running in rats. Physiol Behav 60(1):171–176
Tarr BA et al (2004) Voluntary running distance is negatively correlated with striatal dopamine release in untrained rats. Behav Brain Res 154(2):493–499
Wang Z et al (2015) Exercise alters resting-state functional connectivity of motor circuits in parkinsonian rats. Neurobiol Aging 36(1):536–544
Fornaguera J, Schwarting RK (1999) Early behavioral changes after nigro-striatal system damage can serve as predictors of striatal dopamine depletion. Progress NeuroPsychopharmacol Biol Psychiatry 23(8):1353–1368
Reglodi D et al (2004) Pituitary adenylate cyclase activating polypeptide protects dopaminergic neurons and improves behavioral deficits in a rat model of Parkinson’s disease. Behav Brain Res 151(1):303–312
Yuede CM et al (2009) Effects of voluntary and forced exercise on plaque deposition, hippocampal volume, and behavior in the Tg2576 mouse model of Alzheimer’s disease. Neurobiol Dis 35(3):426–432
Hoffmann P, Thorén P, Ely D (1987) Effect of voluntary exercise on open-field behavior and on aggression in the spontaneously hypertensive rat (SHR). Behav Neural Biol 47(3):346–355
Acknowledgements
The valuable help and support of the Neuroscience Research Center of Kerman University of Medical Sciences, Iran, was much appreciated. Also, we would like to thank Dr. Khadije Esmaeilpour and Dr. Sina Kakoei for their technical assistance in this project.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interests
The authors declare no conflict of interests.
Ethical approval
The study protocol was submitted to the neuroscience research center of medical university of Kerman, (ethical code: KNRC/94/59), and it was determined that the research activity was not human research as defined by guidelines. All the experimental protocols and treatments were approved by the Ethics Committee of Kerman Neuroscience Research Center (EC: KNRC/94/59) that was completely in agreement with the “NIH Guide for the Care and Use of Laboratory Animals”.
All procedures performed were in accordance with the ethical standards of the institutional neuroscience research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
For this type of study, formal consent is not required.
Rights and permissions
About this article
Cite this article
Sheibani, V., Rafie, F., Shahbazi, M. et al. Comparison of voluntary and forced exercise effects on motor behavior in 6-hydroxydopamine-lesion rat model of Parkinson’s disease. Sport Sci Health 13, 203–211 (2017). https://doi.org/10.1007/s11332-017-0354-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11332-017-0354-9