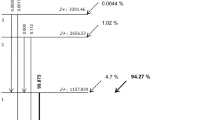
Abstract
Purpose
The increasing interest and availability of non-standard positron-emitting radionuclides has heightened the relevance of radionuclide choice in the development and optimization of new positron emission tomography (PET) imaging procedures, both in preclinical research and clinical practice. Differences in achievable resolution arising from positron range can largely influence application suitability of each radionuclide, especially in small-ring preclinical PET where system blurring factors due to annihilation photon acollinearity and detector geometry are less significant. Some resolution degradation can be mitigated with appropriate range corrections implemented during image reconstruction, the quality of which is contingent on an accurate characterization of positron range.
Procedures
To address this need, we have characterized the positron range of several standard and non-standard PET radionuclides (As-72, F-18, Ga-68, Mn-52, Y-86, and Zr-89) through imaging of small-animal quality control phantoms on a benchmark preclinical PET scanner. Further, the Particle and Heavy Ion Transport code System (PHITS v3.02) code was utilized for Monte Carlo modeling of positron range-dependent blurring effects.
Results
Positron range kernels for each radionuclide were derived from simulation of point sources in ICRP reference tissues. PET resolution and quantitative accuracy afforded by various radionuclides in practicable imaging scenarios were characterized using a convolution-based method based on positron annihilation distributions obtained from PHITS. Our imaging and simulation results demonstrate the degradation of small animal PET resolution, and quantitative accuracy correlates with increasing positron energy; however, for a specific “benchmark” preclinical PET scanner and reconstruction workflow, these differences were observed to be minimal given radionuclides with average positron energies below ~ 400 keV.
Conclusion
Our measurements and simulations of the influence of positron range on PET resolution compare well with previous efforts documented in the literature and provide new data for several radionuclides in increasing clinical and preclinical use. The results will support current and future improvements in methods for positron range corrections in PET imaging.






Similar content being viewed by others
References
Laforest R, Liu X (2008) Image quality with non-standard nuclides in PET. Q J Nucl Med Mol Imaging 52:151–158
Bunka M, Müller C, Vermeulen C, Haller S, Türler A, Schibli R, van der Meulen NP (2016) Imaging quality of (44)Sc in comparison with five other PET radionuclides using Derenzo phantoms and preclinical PET. Appl Radiat Isot 110:129–133
Sanchez-Crespo A (2013) Comparison of gallium-68 and fluorine-18 imaging characteristics in positron emission tomography. Appl Radiat Isot 76:55–62
Sánchez-Crespo A, Andreo P, Larsson SA (2004) Positron flight in human tissues and its influence on PET image spatial resolution. Eur J Nucl Med Mol Imaging 31:44–51
Levin CS, Hoffman EJ (1999) Calculation of positron range and its effect on the fundamental limit of positron emission tomography system spatial resolution. Phys Med Biol 44:781–799. https://doi.org/10.1088/0031-9155/44/3/019
Peng H, Levin CS (2012) Study of PET intrinsic spatial resolution and contrast recovery improvement for PET/MRI systems. Phys Med Biol 57:N101–N115
Cal-González J, Herraiz JL, España S, Corzo PMG, Vaquero JJ, Desco M, Udias JM (2013) Positron range estimations with PeneloPET. Phys Med Biol 58:5127–5152
Champion C, Loirec CL (2006) Positron follow-up in liquid water: I. A new Monte Carlo track-structure code. Phys Med Biol 51:1707–1723
Zur Theorie des Durchgangs schneller Korpuskularstrahlen durch Materie - Bethe - 1930 - Annalen der Physik - Wiley Online Library. https://onlinelibrary.wiley.com/doi/abs/10.1002/andp.19303970303. Accessed 4 Oct 2018
Phelps ME, Hoffman EJ, Huang S-C, Ter-Pogossian MM (1975) Effect of positron range on spatial resolution. J Nucl Med 16:649–652
Cho ZH, Chan JK, Ericksson L, Singh M, Graham S, MacDonald N, Yano Y (1975) Positron ranges obtained from biomedically important positron-emitting radionuclides. J Nucl Med 16:1174–1176
Derenzo SE (1979) Precision measurement of annihilation point spread distributions for medically important positron emitters. Proceedings of the fifth international conference on positron annihilation
Sato T, Niita K, Matsuda N, Hashimoto S, Iwamoto Y, Furuta T, Noda S, Ogawa T, Iwase H, Nakashima H, Fukahori T, Okumura K, Kai T, Chiba S, Sihver L (2015) Overview of particle and heavy ion transport code system PHITS. Ann Nucl Energy 82:110–115
Sato T, Iwamoto Y, Hashimoto S, Ogawa T, Furuta T, Abe SI, Kai T, Tsai PE, Matsuda N, Iwase H, Shigyo N, Sihver L, Niita K (2018) Features of particle and heavy ion transport code system (PHITS) version 3.02. J Nucl Sci Technol 55:684–690
Iwamoto Y, Sato T, Hashimoto S, Ogawa T, Furuta T, Abe SI, Kai T, Matsuda N, Hosoyamada R, Niita K (2017) Benchmark study of the recent version of the PHITS code. J Nucl Sci Technol 54:617–635
Furuta T, Sato T, Han MC, Yeom YS, Kim CH, Brown JL, Bolch WE (2017) Implementation of tetrahedral-mesh geometry in Monte Carlo radiation transport code PHITS. Phys Med Biol 62:4798–4810
Endo A, Yamaguchi Y (2001) Compilation of new nuclear decay data files used for dose calculation. J Nucl Sci Technol 38:689–696
Alva-Sánchez H, Quintana-Bautista C, Martínez-Dávalos A, Ávila-Rodríguez MA, Rodríguez-Villafuerte M (2016) Positron range in tissue-equivalent materials: experimental microPET studies. Phys Med Biol 61:6307–6321
Wooten AL, Aweda TA, Lewis BC, Gross RB, Lapi SE (2017) Biodistribution and PET imaging of pharmacokinetics of manganese in mice using manganese-52. PLoS One 12:e0174351. https://doi.org/10.1371/journal.pone.0174351
Dogdas B, Stout D, Chatziioannou AF, Leahy RM (2007) Digimouse: a 3D whole body mouse atlas from CT and cryosection data. Phys Med Biol 52:577–587
Si H (2015) TetGen, a Delaunay-based quality tetrahedral mesh generator. ACM Trans Math Softw 41:11:1–11:36. https://doi.org/10.1145/2629697
Disselhorst JA, Boerman OC, Oyen WJG, Slump CH, Visser EP (2010) Spatial resolution of the Inveon small-animal PET scanner for the entire field of view. Nucl Instrum Method A 615:245–248
Visser EP, Disselhorst JA, Brom M, Laverman P, Gotthardt M, Oyen WJ, Boerman OC (2009) Spatial resolution and sensitivity of the Inveon small-animal PET scanner. J Nucl Med 50:139–147
Holland JP, Divilov V, Bander NH, Smith-Jones PM, Larson SM, Lewis JS (2010) 89Zr-DFO-J591 for immunoPET of prostate-specific membrane antigen expression in vivo. J Nucl Med 51:1293–1300
Keinänen O, Fung K, Pourat J, Jallinoja V, Vivier D, Pillarsetty NVK, Airaksinen AJ, Lewis JS, Zeglis BM, Sarparanta M (2017) Pretargeting of internalizing trastuzumab and cetuximab with a 18F-tetrazine tracer in xenograft models. EJNMMI Res 7(95):95. https://doi.org/10.1186/s13550-017-0344-6
Kraeber-Bodéré F, Rousseau C, Bodet-Milin C et al (2015) A pretargeting system for tumor PET imaging and radioimmunotherapy. Front Pharmacol 6(54). https://doi.org/10.3389/fphar.2015.00054
Graves SA, Hernandez R, Fonslet J, England CG, Valdovinos HF, Ellison PA, Barnhart TE, Elema DR, Theuer CP, Cai W, Nickles RJ, Severin GW (2015) Novel preparation methods of (52)Mn for immunoPET imaging. Bioconjug Chem 26:2118–2124
Cox BL, Graves SA, Farhoud M et al (2016) Development of a novel linearly-filled Derenzo microPET phantom. Am J Nucl Med Mol Imaging 6:199–204
DeGraffenreid AJ, Feng Y, Barnes CL et al (2016) Trithiols and their arsenic compounds for potential use in diagnostic and therapeutic radiopharmaceuticals. Nucl Med Biol 43:288–295
Funding
This study is supported by the Radiochemistry and Molecular Imaging Probes Core of MSKCC, which was supported in part by NIH grant P30 CA08748. We thank the DOE Office of Science, Nuclear Physics, Isotope Program under grants DE-SC0015773, DE-SC0016267, and ST5001020 for funding for isotope production at BNL, UAB, and MSKCC, and also acknowledge BNL Project Development grant YN0100000 for this purpose. LMC acknowledges support from the Ruth L. Kirschstein Postdoctoral Fellowship (NIH F32EB025050).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
ESM 1
(PDF 4968 kb)
Rights and permissions
About this article
Cite this article
Carter, L.M., Kesner, A.L., Pratt, E.C. et al. The Impact of Positron Range on PET Resolution, Evaluated with Phantoms and PHITS Monte Carlo Simulations for Conventional and Non-conventional Radionuclides. Mol Imaging Biol 22, 73–84 (2020). https://doi.org/10.1007/s11307-019-01337-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11307-019-01337-2