Abstract
Purpose
With the advent of replication competent viruses for cancer gene therapy, it has become imperative to monitor the biodistribution, expression and replication of these vectors in living organisms. We evaluated the potential of I-124 positron emission tomography (PET)/computed tomography (CT) imaging in gene therapy animal models utilizing the sodium iodide symporter (NIS) and compared the findings to I-123 gamma camera imaging.
Procedures
CB17 SCID mice were implanted with myeloma cell lines expressing NIS or infected by MV-NIS given systemically. Mice were imaged by both gamma camera (I-123) and PET/CT (I-124 ) and image quality assessed.
Results
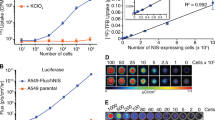
NIS expressing tumors concentrated 7.1% of the injected activity while tumors infected with the control virus had only 0.3% of the activity injected.
Conclusions
I-124 PET/CT in combination with NIS allows the tracking of stably transfected tumors or intravenously transfected tumors. Combined modality imaging using PET/CT allows accurate and non-invasive imaging of the distribution and gene expression of a replicating viral vector in living systems.







Similar content being viewed by others
References
Kirn D (2001) Oncolytic virotherapy for cancer with the adenovirus dl1520 (Onyx-015): Results of phase I and II trials. Expert Opin Biol Ther 1:525–538
Pecora AL, Rizvi N, Cohen GI, et al. (2002) Phase I trial of intravenous administration of PV701, an oncolytic virus, in patients with advanced solid cancers. J Clin Oncol 20:2251–2266
Russell SJ (2002) RNA viruses as virotherapy agents. Cancer Gene Ther 9:961–966
Tjuvajev JG, Stockhammer G, Desai R, et al. (1995) Imaging the expression of transfected genes in vivo. Cancer Res 55:6126–6132
Gambhir SS, Herschman HR, Cherry SR, et al. (2000) Imaging transgene expression with radionuclide imaging technologies. Neoplasia 2:118–138
Herschman HR, MacLaren DC, Iyer M, et al. (2000) Seeing is believing: Non-invasive, quantitative and repetitive imaging of reporter gene expression in living animals, using positron emission tomography. J Neurosci Res 59:699–705
Dingli D, Russell SJ, Morris JC (2003) In vivo imaging and tumor therapy with the sodium iodide symporter. J Cell Biochem 90:1079–1086
Spitzweg C, Morris JC (2001) Approaches to gene therapy with sodium/iodide symporter. Exp Clin Endocrinol Diabetes 109:56–59
Spitzweg C, Dietz AB, O’Connor MK, et al. (2001) In vivo sodium iodide symporter gene therapy of prostate cancer. Gene Ther 8:1524–1531
Dingli D, Diaz RM, Bergert ER, O’Connor MK, Morris JC, Russell SJ (2003) Genetically targeted radiotherapy for multiple myeloma. Blood 20:20
Dingli D, Peng KW, Harvey ME, O’Connor MK, Cattaneo R, Morris JC, Russell SJ (2004) Image-guided radiovirotherapy for multiple myeloma using a recombinant measles virus expressing the thyroidal sodium iodide symporter. Blood 103:1641–1646
Eskandari S, Loo DD, Dai G, Levy O, Wright EM, Carrasco N (1997) Thyroid Na+/Isymporter. Mechanism, stoichiometry, and specificity. J Biol Chem 272:27230–27238
Carlin S, Cunningham SH, Boyd M, McCluskey AG, Mairs RJ (2000) Experimental targeted radioiodide therapy following transfection of the sodium iodide symporter gene: Effect on clonogenicity in both two-and three-dimensional models. Cancer Gene Ther 7:1529–1536
Van Sande J, Massart C, Beauwens R, et al. (2003) Anion selectivity by the sodium iodide symporter. Endocrinology 144:247–252
Cho JY, Shen DH, Yang W, et al. (2002) In vivo imaging and radioiodine therapy following sodium iodide symporter gene transfer in animal model of intracerebral gliomas. Gene Ther 9:1139–1145
Barton KN, Tyson D, Stricker H, Lew YS, Heisey G, Koul S, de la Zerda A, Yin FF, Yan H, Nagaraja TN, Randall KA, Jin GK, Fenstermacher JD, Jhiang S, Kim JH, Freytag SO, Brown SL (2003) GENIS: Gene expression of sodium iodide symporter for noninvasive imaging of gene therapy vectors and quantification of gene expression in vivo. Molec Ther 8:508–518
Groot-Wassink T, Aboagye EO, Glaser M, Lemoine NR, Vassaux G (2002) Adenovirus biodistribution and noninvasive imaging of gene expression in vivo by positron emission tomography using human sodium/iodide symporter as reporter gene. Hum Gene Ther 13:1723–1735
Lardinois D, Weder W, Hany TF, Kamel EM, Korom S, Seifert B, von Schulthess GK, Steinert HC (2003) Staging of non-small-cell lung cancer with integrated positron-emission tomography and computed tomography. N Engl J Med 348:2500–2507
Pentlow KS, Graham MC, Lambrecht RM, Cheung NK, Larson SM (1991) Quantitative imaging of I-124 using positron emission tomography with applications to radioimmunodiagnosis and radioimmunotherapy. Med Phys 18:357–366
Herzog H, Tellmann L, Qaim SM, Spellerberg S, Schmid A, Coenen HH (2002) PET quantitation and imaging of th enon-pure positron-emitting iodine isotope 124I. Appl Radiat Isotopes 56:673–679
Jhiang SM, Cho JY, Ryu KY, et al. (1998) An immunohistochemical study of Na+/Isymporter in human thyroid tissues and salivary gland tissues. Endocrinology 139:4416–4419
Spitzweg C, Joba W, Eisenmenger W, Heufelder AE (1998) Analysis of human sodium iodide symporter gene expression in extrathyroidal tissues and cloning of its complementary deoxyribonucleic acids from salivary gland, mammary gland, and gastric mucosa. J Clin Endocrinol Metab 83:1746–1751
Spitzweg C, Joba W, Schriever K, Goellner JR, Morris JC, Heufelder AE (1999) Analysis of human sodium iodide symporter immunoreactivity in human exocrine glands. J Clin Endocrinol Metab 84:4178–4184
Jhiang SM (2000) Regulation of sodium/iodide symporter. Rev Endocr Metab Disord 1:205–215
Riedel C, Levy O, Carrasco N (2001) Post-transcriptional regulation of the sodium/iodide symporter by thyrotropin. J Biol Chem 276:21458–21463
Radecke F, Spielhofer P, Schneider H, et al. (1995) Rescue of measles viruses from cloned DNA. EMBO J 14:5773–5784
Spitzweg C, O’Connor MK, Bergert ER, Tindall DJ, Young CY, Morris JC (2000) Treatment of prostate cancer by radioiodine therapy after tissue- specific expression of the sodium iodide symporter. Cancer Res 60:6526–6530
Schneider-Schaulies S, Schneider-Schaulies J, Niewiesk S, Ter Meulen V (2002) Measles virus: Immunomodulation and cell tropism as pathogenicity determinants. Med Microbiol Immunol (Berl) 191:83–87
Groot-Wassink T, Aboagye EO, Wang Y, Lemoine NR, Reader AJ, Vassaux G (2004) Quantitative imaging of Na/I symporter transgene expression using positron emission tomography in the living animal. Molec Ther 9:436–442
Acknowledgments
The authors acknowledge the expert technical assistance of Royce Ruter (gamma camera imaging), Mary E. Harvey (veterinary technologist) and Dr Rafael Fonseca, Mayo Clinic, Rochester for the MM1 cell line. This work was supported by grants CA 83181 and CA 91956 from the National Institutes of Health, the Multiple Myeloma Research Foundation (MMRF) and Mayo Foundation.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dingli, D., Kemp, B.J., O’Connor, M.K. et al. Combined I-124 Positron Emission Tomography/Computed Tomography Imaging of NIS Gene Expression in Animal Models of Stably Transfected and Intravenously Transfected Tumor. Mol Imaging Biol 8, 16–23 (2006). https://doi.org/10.1007/s11307-005-0025-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11307-005-0025-0